Class 6 Exam > Class 6 Questions > How will you show that air is essential for b...
Start Learning for Free
How will you show that air is essential for burning fuel. ?answer in detail
Verified Answer
How will you show that air is essential for burning fuel. ?answer in d...
Air is essential for burning fuel , Fire is a chemical reaction that creates light and heat from oxygen and fuel. A lit candle needs to draw oxygen from the air in order to continue burning. If you limit the amount of air available, the candle's flame eventually goes out once it uses up all the oxygen.
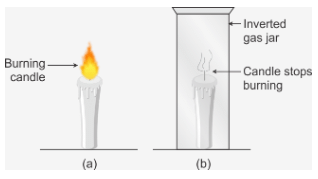
Activity: Take a candle and fix it on a table. The candle is lighted. The candle will continue to burn due to continuously available fresh air providing the required oxygen for combustion.
Now cover the burning candle by putting an inverted gas jar over it. After a short time, the candle stops burning and gets extinguished. When the burning candle is covered with gas jar, then the candle takes away the oxygen necessary for burning from the air enclosed in the gas jar. After some time, when all the oxygen of air inside the gas jar is used up, then the burning candle gets extinguished. This proves that air is necessary for combustion of substances.

 This question is part of UPSC exam. View all Class 6 courses
This question is part of UPSC exam. View all Class 6 courses
Most Upvoted Answer
How will you show that air is essential for burning fuel. ?answer in d...
Air is essential for burning fuel
Burning fuel requires the presence of oxygen, which is abundantly available in the air. The process of burning, also known as combustion, is a chemical reaction that occurs between a fuel source and oxygen. The reaction releases heat and light energy, resulting in the production of carbon dioxide and water vapor.
The role of oxygen in combustion
Oxygen is a key component in the combustion process as it acts as an oxidizer. It combines with the fuel molecules and facilitates their breakdown, releasing energy in the form of heat and light. Without oxygen, the fuel cannot undergo combustion and will not burn.
The composition of air
Air is a mixture of gases, primarily consisting of nitrogen (78%) and oxygen (21%). The remaining 1% comprises trace gases such as carbon dioxide, water vapor, and noble gases. The presence of oxygen in the air makes it an essential component for combustion.
The combustion process
When a fuel source is exposed to heat or a flame, it undergoes a series of chemical reactions. The heat energy breaks down the chemical bonds in the fuel molecules, releasing volatile compounds. These volatile compounds then react with oxygen in the air, resulting in the release of energy in the form of heat and light.
Importance of air in burning fuel
1. Oxygen supply: In the presence of air, oxygen is readily available for the combustion process. It reacts with the fuel, allowing it to burn and release energy.
2. Sustaining the flame: Air provides a continuous supply of oxygen to sustain the flame. Without air, the flame would extinguish due to the lack of oxygen.
3. Complete combustion: Adequate air supply ensures complete combustion of the fuel. This means that the fuel is fully oxidized, resulting in the production of carbon dioxide and water vapor as byproducts. Incomplete combustion, on the other hand, can lead to the formation of harmful pollutants such as carbon monoxide.
Conclusion
Air, specifically the oxygen present in it, is crucial for the burning of fuel. It acts as an oxidizer, facilitating the chemical reactions that release energy during combustion. Without the presence of air, fuel cannot burn, and the combustion process cannot occur. Understanding the role of air in combustion is essential for various applications, such as heating, transportation, and energy production.
Burning fuel requires the presence of oxygen, which is abundantly available in the air. The process of burning, also known as combustion, is a chemical reaction that occurs between a fuel source and oxygen. The reaction releases heat and light energy, resulting in the production of carbon dioxide and water vapor.
The role of oxygen in combustion
Oxygen is a key component in the combustion process as it acts as an oxidizer. It combines with the fuel molecules and facilitates their breakdown, releasing energy in the form of heat and light. Without oxygen, the fuel cannot undergo combustion and will not burn.
The composition of air
Air is a mixture of gases, primarily consisting of nitrogen (78%) and oxygen (21%). The remaining 1% comprises trace gases such as carbon dioxide, water vapor, and noble gases. The presence of oxygen in the air makes it an essential component for combustion.
The combustion process
When a fuel source is exposed to heat or a flame, it undergoes a series of chemical reactions. The heat energy breaks down the chemical bonds in the fuel molecules, releasing volatile compounds. These volatile compounds then react with oxygen in the air, resulting in the release of energy in the form of heat and light.
Importance of air in burning fuel
1. Oxygen supply: In the presence of air, oxygen is readily available for the combustion process. It reacts with the fuel, allowing it to burn and release energy.
2. Sustaining the flame: Air provides a continuous supply of oxygen to sustain the flame. Without air, the flame would extinguish due to the lack of oxygen.
3. Complete combustion: Adequate air supply ensures complete combustion of the fuel. This means that the fuel is fully oxidized, resulting in the production of carbon dioxide and water vapor as byproducts. Incomplete combustion, on the other hand, can lead to the formation of harmful pollutants such as carbon monoxide.
Conclusion
Air, specifically the oxygen present in it, is crucial for the burning of fuel. It acts as an oxidizer, facilitating the chemical reactions that release energy during combustion. Without the presence of air, fuel cannot burn, and the combustion process cannot occur. Understanding the role of air in combustion is essential for various applications, such as heating, transportation, and energy production.
Attention Class 6 Students!
To make sure you are not studying endlessly, EduRev has designed Class 6 study material, with Structured Courses, Videos, & Test Series. Plus get personalized analysis, doubt solving and improvement plans to achieve a great score in Class 6.

|
Explore Courses for Class 6 exam
|

|
Similar Class 6 Doubts
How will you show that air is essential for burning fuel. ?answer in detail
Question Description
How will you show that air is essential for burning fuel. ?answer in detail for Class 6 2024 is part of Class 6 preparation. The Question and answers have been prepared according to the Class 6 exam syllabus. Information about How will you show that air is essential for burning fuel. ?answer in detail covers all topics & solutions for Class 6 2024 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for How will you show that air is essential for burning fuel. ?answer in detail.
How will you show that air is essential for burning fuel. ?answer in detail for Class 6 2024 is part of Class 6 preparation. The Question and answers have been prepared according to the Class 6 exam syllabus. Information about How will you show that air is essential for burning fuel. ?answer in detail covers all topics & solutions for Class 6 2024 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for How will you show that air is essential for burning fuel. ?answer in detail.
Solutions for How will you show that air is essential for burning fuel. ?answer in detail in English & in Hindi are available as part of our courses for Class 6.
Download more important topics, notes, lectures and mock test series for Class 6 Exam by signing up for free.
Here you can find the meaning of How will you show that air is essential for burning fuel. ?answer in detail defined & explained in the simplest way possible. Besides giving the explanation of
How will you show that air is essential for burning fuel. ?answer in detail, a detailed solution for How will you show that air is essential for burning fuel. ?answer in detail has been provided alongside types of How will you show that air is essential for burning fuel. ?answer in detail theory, EduRev gives you an
ample number of questions to practice How will you show that air is essential for burning fuel. ?answer in detail tests, examples and also practice Class 6 tests.

|
Explore Courses for Class 6 exam
|

|
Suggested Free Tests
Signup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.

























