Class 10 Exam > Class 10 Questions > Which of the following is not a saturated hyd... Start Learning for Free
Which of the following is not a saturated hydrocarbon
- a)Cyclohexane
- b)Butane
- c)Isobutane
- d)Benzene
Correct answer is option 'D'. Can you explain this answer?
Most Upvoted Answer
Which of the following is not a saturated hydrocarbona)Cyclohexaneb)Bu...
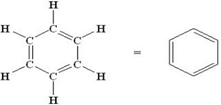
Benzene- C6H6 has alternate carbon-carbon single and double bonds.

Free Test
FREE
| Start Free Test |
Community Answer
Which of the following is not a saturated hydrocarbona)Cyclohexaneb)Bu...
Understanding Saturated Hydrocarbons
Saturated hydrocarbons are organic compounds consisting entirely of hydrogen and carbon, featuring only single bonds between the carbon atoms. This structure leads to the maximum number of hydrogen atoms attached to the carbon skeleton.
Identification of the Options
- Cyclohexane
- Cyclohexane is a saturated hydrocarbon (C6H12) with a ring structure and only single bonds.
- Butane
- Butane (C4H10) is a straight-chain saturated hydrocarbon, containing only single bonds.
- Isobutane
- Isobutane (C4H10) is a branched form of butane, also saturated with only single bonds.
- Benzene
- Benzene (C6H6) is an aromatic compound and is not a saturated hydrocarbon. It contains alternating double bonds, which means it does not have the maximum number of hydrogen atoms.
Conclusion
Thus, the correct answer is option 'D', Benzene, as it does not meet the criteria for a saturated hydrocarbon due to the presence of double bonds in its structure. Understanding the nature of these bonds is crucial in distinguishing between saturated and unsaturated hydrocarbons.
Saturated hydrocarbons are organic compounds consisting entirely of hydrogen and carbon, featuring only single bonds between the carbon atoms. This structure leads to the maximum number of hydrogen atoms attached to the carbon skeleton.
Identification of the Options
- Cyclohexane
- Cyclohexane is a saturated hydrocarbon (C6H12) with a ring structure and only single bonds.
- Butane
- Butane (C4H10) is a straight-chain saturated hydrocarbon, containing only single bonds.
- Isobutane
- Isobutane (C4H10) is a branched form of butane, also saturated with only single bonds.
- Benzene
- Benzene (C6H6) is an aromatic compound and is not a saturated hydrocarbon. It contains alternating double bonds, which means it does not have the maximum number of hydrogen atoms.
Conclusion
Thus, the correct answer is option 'D', Benzene, as it does not meet the criteria for a saturated hydrocarbon due to the presence of double bonds in its structure. Understanding the nature of these bonds is crucial in distinguishing between saturated and unsaturated hydrocarbons.
 | Explore Courses for Class 10 exam |  |
Question Description
Which of the following is not a saturated hydrocarbona)Cyclohexaneb)Butanec)Isobutaned)BenzeneCorrect answer is option 'D'. Can you explain this answer? for Class 10 2026 is part of Class 10 preparation. The Question and answers have been prepared according to the Class 10 exam syllabus. Information about Which of the following is not a saturated hydrocarbona)Cyclohexaneb)Butanec)Isobutaned)BenzeneCorrect answer is option 'D'. Can you explain this answer? covers all topics & solutions for Class 10 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for Which of the following is not a saturated hydrocarbona)Cyclohexaneb)Butanec)Isobutaned)BenzeneCorrect answer is option 'D'. Can you explain this answer?.
Which of the following is not a saturated hydrocarbona)Cyclohexaneb)Butanec)Isobutaned)BenzeneCorrect answer is option 'D'. Can you explain this answer? for Class 10 2026 is part of Class 10 preparation. The Question and answers have been prepared according to the Class 10 exam syllabus. Information about Which of the following is not a saturated hydrocarbona)Cyclohexaneb)Butanec)Isobutaned)BenzeneCorrect answer is option 'D'. Can you explain this answer? covers all topics & solutions for Class 10 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for Which of the following is not a saturated hydrocarbona)Cyclohexaneb)Butanec)Isobutaned)BenzeneCorrect answer is option 'D'. Can you explain this answer?.
Solutions for Which of the following is not a saturated hydrocarbona)Cyclohexaneb)Butanec)Isobutaned)BenzeneCorrect answer is option 'D'. Can you explain this answer? in English & in Hindi are available as part of our courses for Class 10. Download more important topics, notes, lectures and mock test series for Class 10 Exam by signing up for free.
Here you can find the meaning of Which of the following is not a saturated hydrocarbona)Cyclohexaneb)Butanec)Isobutaned)BenzeneCorrect answer is option 'D'. Can you explain this answer? defined & explained in the simplest way possible. Besides giving the explanation of Which of the following is not a saturated hydrocarbona)Cyclohexaneb)Butanec)Isobutaned)BenzeneCorrect answer is option 'D'. Can you explain this answer?, a detailed solution for Which of the following is not a saturated hydrocarbona)Cyclohexaneb)Butanec)Isobutaned)BenzeneCorrect answer is option 'D'. Can you explain this answer? has been provided alongside types of Which of the following is not a saturated hydrocarbona)Cyclohexaneb)Butanec)Isobutaned)BenzeneCorrect answer is option 'D'. Can you explain this answer? theory, EduRev gives you an ample number of questions to practice Which of the following is not a saturated hydrocarbona)Cyclohexaneb)Butanec)Isobutaned)BenzeneCorrect answer is option 'D'. Can you explain this answer? tests, examples and also practice Class 10 tests.
 | Explore Courses for Class 10 exam |  |
Top Courses for Class 10
Explore CoursesSignup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.






