MCAT Exam > MCAT Questions > Ethylene glycol is known to most Americans as... Start Learning for Free
Ethylene glycol is known to most Americans as antifreeze and to some for having a sweet syrupy taste. For scientists, it can be used as a protecting group for carbonyls. Which is the most stable conformer of the compound ethylene glycol depicted below?
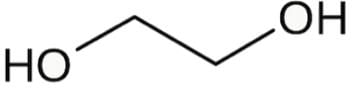

- a)Eclipsed conformer
- b)Gauche conformer
- c)Para conformer
- d)Anti conformer
Correct answer is option 'B'. Can you explain this answer?
Verified Answer
Ethylene glycol is known to most Americans as antifreeze and to some f...
- There is an inverse relationship between energy and stability. The more energy a conformer molecule possesses the more unstable it is, and the less energy the more stable. It is worth noting that there is enough energy to interconvert between the various conformers at room temperature.
- Eclipsed conformations tend to be less stable than the corresponding staggered conformations and to have a higher potential energy due to the interaction of electron clouds of adjacent atoms. Here are two examples of eclipsed conformers, fully eclipsed and eclipsed:
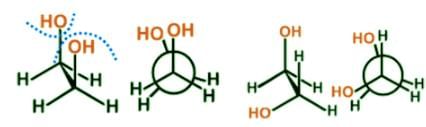
- For butane the gauche conformer is less stable than the anti conformer due to a crowding of the two methyl groups in the gauche structure, and that destabilizing factor is called steric strain or hindrance:
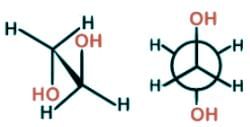
- There is no para conformer. Para is usually used to refer to the 1,4-isomer, where the substituents sit on carbon 1 and 4 of the aromatic ring structure.
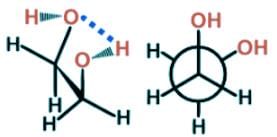
- In 1, 2-ethanediol, the gauche conformer presents an interesting configuration allowing for hydrogen bonding to occur between the two hydroxyl groups leading to a lower energy state and greater stability than the anti conformer.
Most Upvoted Answer
Ethylene glycol is known to most Americans as antifreeze and to some f...
- There is an inverse relationship between energy and stability. The more energy a conformer molecule possesses the more unstable it is, and the less energy the more stable. It is worth noting that there is enough energy to interconvert between the various conformers at room temperature.
- Eclipsed conformations tend to be less stable than the corresponding staggered conformations and to have a higher potential energy due to the interaction of electron clouds of adjacent atoms. Here are two examples of eclipsed conformers, fully eclipsed and eclipsed:

- For butane the gauche conformer is less stable than the anti conformer due to a crowding of the two methyl groups in the gauche structure, and that destabilizing factor is called steric strain or hindrance:

- There is no para conformer. Para is usually used to refer to the 1,4-isomer, where the substituents sit on carbon 1 and 4 of the aromatic ring structure.
- In 1, 2-ethanediol, the gauche conformer presents an interesting configuration allowing for hydrogen bonding to occur between the two hydroxyl groups leading to a lower energy state and greater stability than the anti conformer.

 | Explore Courses for MCAT exam |  |
Question Description
Ethylene glycol is known to most Americans as antifreeze and to some for having a sweet syrupy taste. For scientists, it can be used as a protecting group for carbonyls. Which is the most stable conformer of the compound ethylene glycol depicted below?a)Eclipsed conformerb)Gauche conformerc)Para conformerd)Anti conformerCorrect answer is option 'B'. Can you explain this answer? for MCAT 2026 is part of MCAT preparation. The Question and answers have been prepared according to the MCAT exam syllabus. Information about Ethylene glycol is known to most Americans as antifreeze and to some for having a sweet syrupy taste. For scientists, it can be used as a protecting group for carbonyls. Which is the most stable conformer of the compound ethylene glycol depicted below?a)Eclipsed conformerb)Gauche conformerc)Para conformerd)Anti conformerCorrect answer is option 'B'. Can you explain this answer? covers all topics & solutions for MCAT 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for Ethylene glycol is known to most Americans as antifreeze and to some for having a sweet syrupy taste. For scientists, it can be used as a protecting group for carbonyls. Which is the most stable conformer of the compound ethylene glycol depicted below?a)Eclipsed conformerb)Gauche conformerc)Para conformerd)Anti conformerCorrect answer is option 'B'. Can you explain this answer?.
Ethylene glycol is known to most Americans as antifreeze and to some for having a sweet syrupy taste. For scientists, it can be used as a protecting group for carbonyls. Which is the most stable conformer of the compound ethylene glycol depicted below?a)Eclipsed conformerb)Gauche conformerc)Para conformerd)Anti conformerCorrect answer is option 'B'. Can you explain this answer? for MCAT 2026 is part of MCAT preparation. The Question and answers have been prepared according to the MCAT exam syllabus. Information about Ethylene glycol is known to most Americans as antifreeze and to some for having a sweet syrupy taste. For scientists, it can be used as a protecting group for carbonyls. Which is the most stable conformer of the compound ethylene glycol depicted below?a)Eclipsed conformerb)Gauche conformerc)Para conformerd)Anti conformerCorrect answer is option 'B'. Can you explain this answer? covers all topics & solutions for MCAT 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for Ethylene glycol is known to most Americans as antifreeze and to some for having a sweet syrupy taste. For scientists, it can be used as a protecting group for carbonyls. Which is the most stable conformer of the compound ethylene glycol depicted below?a)Eclipsed conformerb)Gauche conformerc)Para conformerd)Anti conformerCorrect answer is option 'B'. Can you explain this answer?.
Solutions for Ethylene glycol is known to most Americans as antifreeze and to some for having a sweet syrupy taste. For scientists, it can be used as a protecting group for carbonyls. Which is the most stable conformer of the compound ethylene glycol depicted below?a)Eclipsed conformerb)Gauche conformerc)Para conformerd)Anti conformerCorrect answer is option 'B'. Can you explain this answer? in English & in Hindi are available as part of our courses for MCAT. Download more important topics, notes, lectures and mock test series for MCAT Exam by signing up for free.
Here you can find the meaning of Ethylene glycol is known to most Americans as antifreeze and to some for having a sweet syrupy taste. For scientists, it can be used as a protecting group for carbonyls. Which is the most stable conformer of the compound ethylene glycol depicted below?a)Eclipsed conformerb)Gauche conformerc)Para conformerd)Anti conformerCorrect answer is option 'B'. Can you explain this answer? defined & explained in the simplest way possible. Besides giving the explanation of Ethylene glycol is known to most Americans as antifreeze and to some for having a sweet syrupy taste. For scientists, it can be used as a protecting group for carbonyls. Which is the most stable conformer of the compound ethylene glycol depicted below?a)Eclipsed conformerb)Gauche conformerc)Para conformerd)Anti conformerCorrect answer is option 'B'. Can you explain this answer?, a detailed solution for Ethylene glycol is known to most Americans as antifreeze and to some for having a sweet syrupy taste. For scientists, it can be used as a protecting group for carbonyls. Which is the most stable conformer of the compound ethylene glycol depicted below?a)Eclipsed conformerb)Gauche conformerc)Para conformerd)Anti conformerCorrect answer is option 'B'. Can you explain this answer? has been provided alongside types of Ethylene glycol is known to most Americans as antifreeze and to some for having a sweet syrupy taste. For scientists, it can be used as a protecting group for carbonyls. Which is the most stable conformer of the compound ethylene glycol depicted below?a)Eclipsed conformerb)Gauche conformerc)Para conformerd)Anti conformerCorrect answer is option 'B'. Can you explain this answer? theory, EduRev gives you an ample number of questions to practice Ethylene glycol is known to most Americans as antifreeze and to some for having a sweet syrupy taste. For scientists, it can be used as a protecting group for carbonyls. Which is the most stable conformer of the compound ethylene glycol depicted below?a)Eclipsed conformerb)Gauche conformerc)Para conformerd)Anti conformerCorrect answer is option 'B'. Can you explain this answer? tests, examples and also practice MCAT tests.
 | Explore Courses for MCAT exam |  |
Top Courses for MCAT
Explore CoursesSignup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.






