Class 12 Exam > Class 12 Questions > What is the shortest wavelength present in th... Start Learning for Free
What is the shortest wavelength present in the Paschen series of spectral lines?
- a)840 nm
- b)800 nm
- c)860 nm
- d)820 nm
Correct answer is option 'D'. Can you explain this answer?
Verified Answer
What is the shortest wavelength present in the Paschen series of spect...
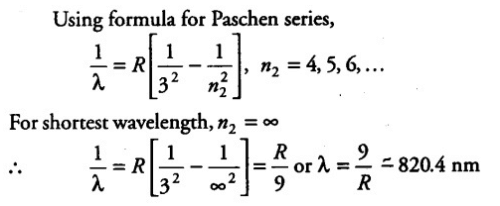
Most Upvoted Answer
What is the shortest wavelength present in the Paschen series of spect...
Paschen Series
The Paschen series is a series of spectral lines in the infrared region of the electromagnetic spectrum. It is named after Friedrich Paschen, who discovered it in 1908. The series corresponds to transitions from higher energy levels to the third energy level of the hydrogen atom.
Wavelength Calculation
The formula for calculating the wavelength of spectral lines in the Paschen series is:
1/λ = R [1/32 - 1/n^2]
where λ is the wavelength, R is the Rydberg constant, and n is an integer representing the energy level.
The shortest wavelength in the Paschen series corresponds to the transition from the fourth energy level (n=4) to the third energy level (n=3). Plugging these values into the formula, we get:
1/λ = R [1/32 - 1/4^2]
1/λ = R [1/32 - 1/16]
1/λ = R [3/256]
λ = 256/3R
Substituting the value of the Rydberg constant (1.097 x 10^7 m^-1), we get:
λ = 820.4 nm
Therefore, the correct answer is option 'D' (820 nm) as it represents the shortest wavelength present in the Paschen series of spectral lines.
The Paschen series is a series of spectral lines in the infrared region of the electromagnetic spectrum. It is named after Friedrich Paschen, who discovered it in 1908. The series corresponds to transitions from higher energy levels to the third energy level of the hydrogen atom.
Wavelength Calculation
The formula for calculating the wavelength of spectral lines in the Paschen series is:
1/λ = R [1/32 - 1/n^2]
where λ is the wavelength, R is the Rydberg constant, and n is an integer representing the energy level.
The shortest wavelength in the Paschen series corresponds to the transition from the fourth energy level (n=4) to the third energy level (n=3). Plugging these values into the formula, we get:
1/λ = R [1/32 - 1/4^2]
1/λ = R [1/32 - 1/16]
1/λ = R [3/256]
λ = 256/3R
Substituting the value of the Rydberg constant (1.097 x 10^7 m^-1), we get:
λ = 820.4 nm
Therefore, the correct answer is option 'D' (820 nm) as it represents the shortest wavelength present in the Paschen series of spectral lines.
Community Answer
What is the shortest wavelength present in the Paschen series of spect...
Thanks bro
 | Explore Courses for Class 12 exam |  |
Question Description
What is the shortest wavelength present in the Paschen series of spectral lines?a)840 nmb)800 nmc)860 nmd)820 nmCorrect answer is option 'D'. Can you explain this answer? for Class 12 2026 is part of Class 12 preparation. The Question and answers have been prepared according to the Class 12 exam syllabus. Information about What is the shortest wavelength present in the Paschen series of spectral lines?a)840 nmb)800 nmc)860 nmd)820 nmCorrect answer is option 'D'. Can you explain this answer? covers all topics & solutions for Class 12 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for What is the shortest wavelength present in the Paschen series of spectral lines?a)840 nmb)800 nmc)860 nmd)820 nmCorrect answer is option 'D'. Can you explain this answer?.
What is the shortest wavelength present in the Paschen series of spectral lines?a)840 nmb)800 nmc)860 nmd)820 nmCorrect answer is option 'D'. Can you explain this answer? for Class 12 2026 is part of Class 12 preparation. The Question and answers have been prepared according to the Class 12 exam syllabus. Information about What is the shortest wavelength present in the Paschen series of spectral lines?a)840 nmb)800 nmc)860 nmd)820 nmCorrect answer is option 'D'. Can you explain this answer? covers all topics & solutions for Class 12 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for What is the shortest wavelength present in the Paschen series of spectral lines?a)840 nmb)800 nmc)860 nmd)820 nmCorrect answer is option 'D'. Can you explain this answer?.
Solutions for What is the shortest wavelength present in the Paschen series of spectral lines?a)840 nmb)800 nmc)860 nmd)820 nmCorrect answer is option 'D'. Can you explain this answer? in English & in Hindi are available as part of our courses for Class 12. Download more important topics, notes, lectures and mock test series for Class 12 Exam by signing up for free.
Here you can find the meaning of What is the shortest wavelength present in the Paschen series of spectral lines?a)840 nmb)800 nmc)860 nmd)820 nmCorrect answer is option 'D'. Can you explain this answer? defined & explained in the simplest way possible. Besides giving the explanation of What is the shortest wavelength present in the Paschen series of spectral lines?a)840 nmb)800 nmc)860 nmd)820 nmCorrect answer is option 'D'. Can you explain this answer?, a detailed solution for What is the shortest wavelength present in the Paschen series of spectral lines?a)840 nmb)800 nmc)860 nmd)820 nmCorrect answer is option 'D'. Can you explain this answer? has been provided alongside types of What is the shortest wavelength present in the Paschen series of spectral lines?a)840 nmb)800 nmc)860 nmd)820 nmCorrect answer is option 'D'. Can you explain this answer? theory, EduRev gives you an ample number of questions to practice What is the shortest wavelength present in the Paschen series of spectral lines?a)840 nmb)800 nmc)860 nmd)820 nmCorrect answer is option 'D'. Can you explain this answer? tests, examples and also practice Class 12 tests.
 | Explore Courses for Class 12 exam |  |
Top Courses for Class 12
Explore CoursesSignup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.






