Class 11 Exam > Class 11 Questions > Passage IIX and Y are two volatile liquids wi...
Start Learning for Free
Passage II
X and Y are two volatile liquids with molar weights of 10 g mol-1 and 40 g mol-1 respectively. Two cotton plugs, one soaked in X and the other soaked in Y, are simultaneously placed at the ends of the tube of length l = 24 cm , as shown in the figure. The tube is filled with an inert gas at 1 atm pressure and temperature of 300 K. Vapours of X and Y react to form a product which is first formed at a distance, d cm from the plug soaked in X. Take X and Y to have equal molecular diameters and assume ideal behaviour for the inert gas and two vapours.
Q. The value of d in cm (shown in the figure), as estimated from Graham’s law is
- a)8
- b)12
- c)16
- d)20
Correct answer is option 'C'. Can you explain this answer?
| FREE This question is part of | Download PDF Attempt this Test |
Verified Answer
Passage IIX and Y are two volatile liquids with molar weights of 10 g ...
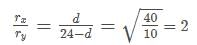
d=48−2d
3d=48
d=16cm
3d=48
d=16cm

|
Explore Courses for Class 11 exam
|

|
Similar Class 11 Doubts
Passage IIX and Y are two volatile liquids with molar weights of 10 g mol-1 and 40 g mol-1 respectively. Two cotton plugs, one soaked in X and the other soaked in Y, are simultaneously placed at the ends of the tube of length l = 24 cm , as shown in the figure. The tube is filled with an inert gas at 1 atm pressure and temperature of 300 K. Vapours of X and Y react to form a product which is first formed at a distance, d cm from the plug soaked in X. Take X and Y to have equal molecular diameters and assume ideal behaviour for the inert gas and two vapours.Q.The value of d in cm (shown in the figure), as estimated from Graham’s law isa)8b)12c)16d)20Correct answer is option 'C'. Can you explain this answer?
Question Description
Passage IIX and Y are two volatile liquids with molar weights of 10 g mol-1 and 40 g mol-1 respectively. Two cotton plugs, one soaked in X and the other soaked in Y, are simultaneously placed at the ends of the tube of length l = 24 cm , as shown in the figure. The tube is filled with an inert gas at 1 atm pressure and temperature of 300 K. Vapours of X and Y react to form a product which is first formed at a distance, d cm from the plug soaked in X. Take X and Y to have equal molecular diameters and assume ideal behaviour for the inert gas and two vapours.Q.The value of d in cm (shown in the figure), as estimated from Graham’s law isa)8b)12c)16d)20Correct answer is option 'C'. Can you explain this answer? for Class 11 2024 is part of Class 11 preparation. The Question and answers have been prepared according to the Class 11 exam syllabus. Information about Passage IIX and Y are two volatile liquids with molar weights of 10 g mol-1 and 40 g mol-1 respectively. Two cotton plugs, one soaked in X and the other soaked in Y, are simultaneously placed at the ends of the tube of length l = 24 cm , as shown in the figure. The tube is filled with an inert gas at 1 atm pressure and temperature of 300 K. Vapours of X and Y react to form a product which is first formed at a distance, d cm from the plug soaked in X. Take X and Y to have equal molecular diameters and assume ideal behaviour for the inert gas and two vapours.Q.The value of d in cm (shown in the figure), as estimated from Graham’s law isa)8b)12c)16d)20Correct answer is option 'C'. Can you explain this answer? covers all topics & solutions for Class 11 2024 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for Passage IIX and Y are two volatile liquids with molar weights of 10 g mol-1 and 40 g mol-1 respectively. Two cotton plugs, one soaked in X and the other soaked in Y, are simultaneously placed at the ends of the tube of length l = 24 cm , as shown in the figure. The tube is filled with an inert gas at 1 atm pressure and temperature of 300 K. Vapours of X and Y react to form a product which is first formed at a distance, d cm from the plug soaked in X. Take X and Y to have equal molecular diameters and assume ideal behaviour for the inert gas and two vapours.Q.The value of d in cm (shown in the figure), as estimated from Graham’s law isa)8b)12c)16d)20Correct answer is option 'C'. Can you explain this answer?.
Passage IIX and Y are two volatile liquids with molar weights of 10 g mol-1 and 40 g mol-1 respectively. Two cotton plugs, one soaked in X and the other soaked in Y, are simultaneously placed at the ends of the tube of length l = 24 cm , as shown in the figure. The tube is filled with an inert gas at 1 atm pressure and temperature of 300 K. Vapours of X and Y react to form a product which is first formed at a distance, d cm from the plug soaked in X. Take X and Y to have equal molecular diameters and assume ideal behaviour for the inert gas and two vapours.Q.The value of d in cm (shown in the figure), as estimated from Graham’s law isa)8b)12c)16d)20Correct answer is option 'C'. Can you explain this answer? for Class 11 2024 is part of Class 11 preparation. The Question and answers have been prepared according to the Class 11 exam syllabus. Information about Passage IIX and Y are two volatile liquids with molar weights of 10 g mol-1 and 40 g mol-1 respectively. Two cotton plugs, one soaked in X and the other soaked in Y, are simultaneously placed at the ends of the tube of length l = 24 cm , as shown in the figure. The tube is filled with an inert gas at 1 atm pressure and temperature of 300 K. Vapours of X and Y react to form a product which is first formed at a distance, d cm from the plug soaked in X. Take X and Y to have equal molecular diameters and assume ideal behaviour for the inert gas and two vapours.Q.The value of d in cm (shown in the figure), as estimated from Graham’s law isa)8b)12c)16d)20Correct answer is option 'C'. Can you explain this answer? covers all topics & solutions for Class 11 2024 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for Passage IIX and Y are two volatile liquids with molar weights of 10 g mol-1 and 40 g mol-1 respectively. Two cotton plugs, one soaked in X and the other soaked in Y, are simultaneously placed at the ends of the tube of length l = 24 cm , as shown in the figure. The tube is filled with an inert gas at 1 atm pressure and temperature of 300 K. Vapours of X and Y react to form a product which is first formed at a distance, d cm from the plug soaked in X. Take X and Y to have equal molecular diameters and assume ideal behaviour for the inert gas and two vapours.Q.The value of d in cm (shown in the figure), as estimated from Graham’s law isa)8b)12c)16d)20Correct answer is option 'C'. Can you explain this answer?.
Solutions for Passage IIX and Y are two volatile liquids with molar weights of 10 g mol-1 and 40 g mol-1 respectively. Two cotton plugs, one soaked in X and the other soaked in Y, are simultaneously placed at the ends of the tube of length l = 24 cm , as shown in the figure. The tube is filled with an inert gas at 1 atm pressure and temperature of 300 K. Vapours of X and Y react to form a product which is first formed at a distance, d cm from the plug soaked in X. Take X and Y to have equal molecular diameters and assume ideal behaviour for the inert gas and two vapours.Q.The value of d in cm (shown in the figure), as estimated from Graham’s law isa)8b)12c)16d)20Correct answer is option 'C'. Can you explain this answer? in English & in Hindi are available as part of our courses for Class 11.
Download more important topics, notes, lectures and mock test series for Class 11 Exam by signing up for free.
Here you can find the meaning of Passage IIX and Y are two volatile liquids with molar weights of 10 g mol-1 and 40 g mol-1 respectively. Two cotton plugs, one soaked in X and the other soaked in Y, are simultaneously placed at the ends of the tube of length l = 24 cm , as shown in the figure. The tube is filled with an inert gas at 1 atm pressure and temperature of 300 K. Vapours of X and Y react to form a product which is first formed at a distance, d cm from the plug soaked in X. Take X and Y to have equal molecular diameters and assume ideal behaviour for the inert gas and two vapours.Q.The value of d in cm (shown in the figure), as estimated from Graham’s law isa)8b)12c)16d)20Correct answer is option 'C'. Can you explain this answer? defined & explained in the simplest way possible. Besides giving the explanation of
Passage IIX and Y are two volatile liquids with molar weights of 10 g mol-1 and 40 g mol-1 respectively. Two cotton plugs, one soaked in X and the other soaked in Y, are simultaneously placed at the ends of the tube of length l = 24 cm , as shown in the figure. The tube is filled with an inert gas at 1 atm pressure and temperature of 300 K. Vapours of X and Y react to form a product which is first formed at a distance, d cm from the plug soaked in X. Take X and Y to have equal molecular diameters and assume ideal behaviour for the inert gas and two vapours.Q.The value of d in cm (shown in the figure), as estimated from Graham’s law isa)8b)12c)16d)20Correct answer is option 'C'. Can you explain this answer?, a detailed solution for Passage IIX and Y are two volatile liquids with molar weights of 10 g mol-1 and 40 g mol-1 respectively. Two cotton plugs, one soaked in X and the other soaked in Y, are simultaneously placed at the ends of the tube of length l = 24 cm , as shown in the figure. The tube is filled with an inert gas at 1 atm pressure and temperature of 300 K. Vapours of X and Y react to form a product which is first formed at a distance, d cm from the plug soaked in X. Take X and Y to have equal molecular diameters and assume ideal behaviour for the inert gas and two vapours.Q.The value of d in cm (shown in the figure), as estimated from Graham’s law isa)8b)12c)16d)20Correct answer is option 'C'. Can you explain this answer? has been provided alongside types of Passage IIX and Y are two volatile liquids with molar weights of 10 g mol-1 and 40 g mol-1 respectively. Two cotton plugs, one soaked in X and the other soaked in Y, are simultaneously placed at the ends of the tube of length l = 24 cm , as shown in the figure. The tube is filled with an inert gas at 1 atm pressure and temperature of 300 K. Vapours of X and Y react to form a product which is first formed at a distance, d cm from the plug soaked in X. Take X and Y to have equal molecular diameters and assume ideal behaviour for the inert gas and two vapours.Q.The value of d in cm (shown in the figure), as estimated from Graham’s law isa)8b)12c)16d)20Correct answer is option 'C'. Can you explain this answer? theory, EduRev gives you an
ample number of questions to practice Passage IIX and Y are two volatile liquids with molar weights of 10 g mol-1 and 40 g mol-1 respectively. Two cotton plugs, one soaked in X and the other soaked in Y, are simultaneously placed at the ends of the tube of length l = 24 cm , as shown in the figure. The tube is filled with an inert gas at 1 atm pressure and temperature of 300 K. Vapours of X and Y react to form a product which is first formed at a distance, d cm from the plug soaked in X. Take X and Y to have equal molecular diameters and assume ideal behaviour for the inert gas and two vapours.Q.The value of d in cm (shown in the figure), as estimated from Graham’s law isa)8b)12c)16d)20Correct answer is option 'C'. Can you explain this answer? tests, examples and also practice Class 11 tests.

|
Explore Courses for Class 11 exam
|

|
Suggested Free Tests
Signup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.























