Class 11 Exam > Class 11 Questions > A pair of enantiomers is possible for _______... Start Learning for Free
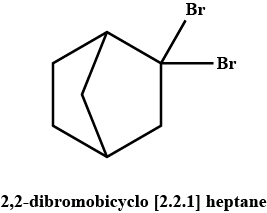
A pair of enantiomers is possible for _______ isomer of 2,2-dibromobicyclo [2.2.1] heptane.
- a)cis
- b)trans
- c)both cis and trans
- d)none of the above
Correct answer is option 'A'. Can you explain this answer?
Verified Answer
A pair of enantiomers is possible for _______ isomer of 2,2-dibromobic...
Only one pair of enantiomers is possible for cis-2,2-dibromobicyclo [2.2.1] heptane. The trans arrangement of one carbon bridge is structurally impossible. Such a molecule would have too much strain.
Most Upvoted Answer
A pair of enantiomers is possible for _______ isomer of 2,2-dibromobic...
Free Test
FREE
| Start Free Test |
Community Answer
A pair of enantiomers is possible for _______ isomer of 2,2-dibromobic...
Understanding Enantiomers in 2,2-Dibromobicyclo[2.2.1]heptane
When discussing the isomers of 2,2-dibromobicyclo[2.2.1]heptane, it’s essential to recognize the structural features that lead to chirality.
Cis vs. Trans Isomerism
- Cis Isomer: In the cis configuration, the bromine atoms are on the same side of the bicyclic structure. This arrangement creates a situation where the molecule lacks a plane of symmetry, leading to the formation of two non-superimposable mirror images, or enantiomers.
- Trans Isomer: In the trans configuration, the bromine atoms are situated on opposite sides. This arrangement allows for a plane of symmetry, resulting in a symmetrical molecule that does not exhibit chirality. As a result, the trans isomer cannot form enantiomers.
Conclusion on Isomer Types
Given the structural characteristics of 2,2-dibromobicyclo[2.2.1]heptane:
- Cis isomer: Can form a pair of enantiomers due to the lack of symmetry.
- Trans isomer: Cannot form enantiomers as it possesses a plane of symmetry.
Thus, the correct answer to the question is option 'A', indicating that a pair of enantiomers is possible for the cis isomer of 2,2-dibromobicyclo[2.2.1]heptane.
This distinction is crucial in stereochemistry and has implications in fields such as pharmaceuticals, where the activity of enantiomers can differ significantly.
When discussing the isomers of 2,2-dibromobicyclo[2.2.1]heptane, it’s essential to recognize the structural features that lead to chirality.
Cis vs. Trans Isomerism
- Cis Isomer: In the cis configuration, the bromine atoms are on the same side of the bicyclic structure. This arrangement creates a situation where the molecule lacks a plane of symmetry, leading to the formation of two non-superimposable mirror images, or enantiomers.
- Trans Isomer: In the trans configuration, the bromine atoms are situated on opposite sides. This arrangement allows for a plane of symmetry, resulting in a symmetrical molecule that does not exhibit chirality. As a result, the trans isomer cannot form enantiomers.
Conclusion on Isomer Types
Given the structural characteristics of 2,2-dibromobicyclo[2.2.1]heptane:
- Cis isomer: Can form a pair of enantiomers due to the lack of symmetry.
- Trans isomer: Cannot form enantiomers as it possesses a plane of symmetry.
Thus, the correct answer to the question is option 'A', indicating that a pair of enantiomers is possible for the cis isomer of 2,2-dibromobicyclo[2.2.1]heptane.
This distinction is crucial in stereochemistry and has implications in fields such as pharmaceuticals, where the activity of enantiomers can differ significantly.
 | Explore Courses for Class 11 exam |  |
Question Description
A pair of enantiomers is possible for _______ isomer of 2,2-dibromobicyclo [2.2.1] heptane.a)cisb)transc)both cis and transd)none of the aboveCorrect answer is option 'A'. Can you explain this answer? for Class 11 2026 is part of Class 11 preparation. The Question and answers have been prepared according to the Class 11 exam syllabus. Information about A pair of enantiomers is possible for _______ isomer of 2,2-dibromobicyclo [2.2.1] heptane.a)cisb)transc)both cis and transd)none of the aboveCorrect answer is option 'A'. Can you explain this answer? covers all topics & solutions for Class 11 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for A pair of enantiomers is possible for _______ isomer of 2,2-dibromobicyclo [2.2.1] heptane.a)cisb)transc)both cis and transd)none of the aboveCorrect answer is option 'A'. Can you explain this answer?.
A pair of enantiomers is possible for _______ isomer of 2,2-dibromobicyclo [2.2.1] heptane.a)cisb)transc)both cis and transd)none of the aboveCorrect answer is option 'A'. Can you explain this answer? for Class 11 2026 is part of Class 11 preparation. The Question and answers have been prepared according to the Class 11 exam syllabus. Information about A pair of enantiomers is possible for _______ isomer of 2,2-dibromobicyclo [2.2.1] heptane.a)cisb)transc)both cis and transd)none of the aboveCorrect answer is option 'A'. Can you explain this answer? covers all topics & solutions for Class 11 2026 Exam. Find important definitions, questions, meanings, examples, exercises and tests below for A pair of enantiomers is possible for _______ isomer of 2,2-dibromobicyclo [2.2.1] heptane.a)cisb)transc)both cis and transd)none of the aboveCorrect answer is option 'A'. Can you explain this answer?.
Solutions for A pair of enantiomers is possible for _______ isomer of 2,2-dibromobicyclo [2.2.1] heptane.a)cisb)transc)both cis and transd)none of the aboveCorrect answer is option 'A'. Can you explain this answer? in English & in Hindi are available as part of our courses for Class 11. Download more important topics, notes, lectures and mock test series for Class 11 Exam by signing up for free.
Here you can find the meaning of A pair of enantiomers is possible for _______ isomer of 2,2-dibromobicyclo [2.2.1] heptane.a)cisb)transc)both cis and transd)none of the aboveCorrect answer is option 'A'. Can you explain this answer? defined & explained in the simplest way possible. Besides giving the explanation of A pair of enantiomers is possible for _______ isomer of 2,2-dibromobicyclo [2.2.1] heptane.a)cisb)transc)both cis and transd)none of the aboveCorrect answer is option 'A'. Can you explain this answer?, a detailed solution for A pair of enantiomers is possible for _______ isomer of 2,2-dibromobicyclo [2.2.1] heptane.a)cisb)transc)both cis and transd)none of the aboveCorrect answer is option 'A'. Can you explain this answer? has been provided alongside types of A pair of enantiomers is possible for _______ isomer of 2,2-dibromobicyclo [2.2.1] heptane.a)cisb)transc)both cis and transd)none of the aboveCorrect answer is option 'A'. Can you explain this answer? theory, EduRev gives you an ample number of questions to practice A pair of enantiomers is possible for _______ isomer of 2,2-dibromobicyclo [2.2.1] heptane.a)cisb)transc)both cis and transd)none of the aboveCorrect answer is option 'A'. Can you explain this answer? tests, examples and also practice Class 11 tests.
 | Explore Courses for Class 11 exam |  |
Top Courses for Class 11
Explore CoursesSignup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.






