Hydrides & Hydrogen Economy
What is Hydrogen Economy?
It is no secret that the world currently is facing an energy crisis.
Non-renewable energy sources such as oil, coal etc are depleting globally. And renewable energy sources are still developing and not able to meet our energy demands fast enough. So scientists from around the world have come up with a vision, to use hydrogen as an energy fuel! This concept is known as Hydrogen Economy.
The term was coined by John Bockrics who worked for General Motors in 1970. It was then understood to be a revolutionary solution to all our energy troubles. Since then a lot of development has taken place in this field. Technological advancements in the last few decades have made this vision very close to possible.
The most primary way to implement hydrogen economy is via transport vehicles. This will be our first step into the future where hydrogen can be used as an efficient energy source. Fuel cell electric vehicles are the main focus of hydrogen economy currently.
Now let us look at a few advantages of hydrogen as a fuel over its conventional counterparts:
- First and foremost advantage is that it is a non-polluting energy source. The only by-product is water.
- Hydrogen, in fact, has a higher efficiency than gasoline, oil, petrol etc
- They operate silently, unlike those engines which work on internal combustion
- Production of hydrogen energy can happen anywhere with water and access to electricity. This helps with grid distribution
- They also have a low heat transmission making them an ideal fuel source
What are Hydrides?
Hydride is the product when hydrogen reacts with any other element, except of course the noble gases. So hydrides are compounds where one atom is hydrogen bonded to another more electropositive element. A hydride is an anion of hydrogen (H-).
The bonding between hydrogen and other elements is always covalent. It just varies in its degrees of covalency.
Types of Hydrides
There are three possible types of hydrides based on their structure and chemical properties. These are:
- Ionic or Saline Hydrides
- Molecular or Covalent Hydrides
- Metallic or Interstitial Hydrides
Ionic or Saline Hydrides
- These are hydrides that form when hydrogen reacts with s-Block elements.
- These are elements found in Group 1 and Group 2.
- These alkali metals and alkaline earth metals are more electropositive than hydrogen.
- The only two exceptions are beryllium hydride and magnesium hydride. Due to the negatively charged ion (H-) they are also known as Ionic Hydride.
- Some examples are Lithium Hydride (LiH), Sodium Hydride (NaH) etc.
Chemical properties
- These saline hydrides are in solid form. They are actually crystals and usually have a whitish hue
- Ionic hydride has both a high melting point and the boiling point as all-metal compounds usually do
- They have high density since these are stable molecules.
- They conduct electricity in a molten state liberating a diatomic molecule of hydrogen gas at the anode. This is also a way to confirm the presence of hydrogen in the compound. Let us take a look at the reaction of Calcium Hydride electrolysis
CaH2 (melt) → Ca2+ + 2H-
At anode: 2H- → H2 + 2e-
At cathode: Ca2+ + 2e- → Ca - One very unique chemical property of Saline hydrides is that they react very vigorously with water and other solvents such as ethanol and ammonia. In the process, they release pure hydrogen gas. This is why they must be kept in moisture-free environments. Here is a reaction of Sodium Hydride reacting with water and ammonia
NaH + H2O → NaOH + H2
NaH + NH3 → NaNH2 + H2
Molecular or Covalent Hydrides
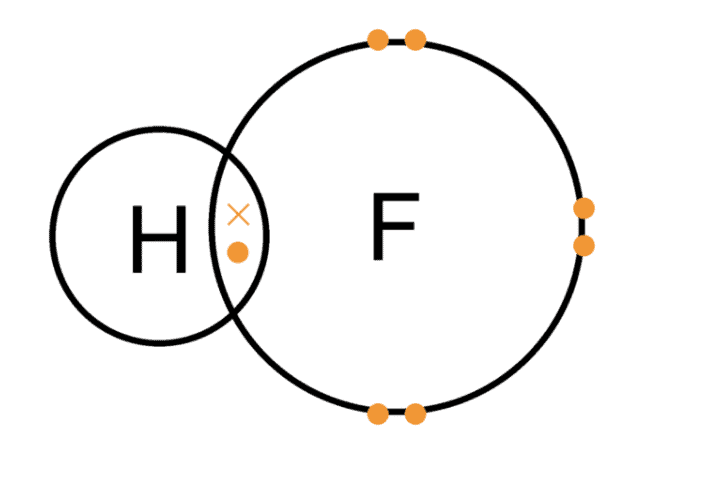
- These are formed by elements of p-block having higher electronegativity than hydrogen.
- These are highly electronegative elements, far more than hydrogen. Hydrogen atoms form a covalent bond with these p-block atoms.
General chemical formula for Covalent Hydrides is XH(8-n), where "n" is the number of electrons the element has in its outermost shell.
Types of Molecular or Covalent Hydrides:
- Electron-deficient hydrides: These are the hydrides that do not have a sufficient number of electrons needed to form normal covalent bonds, e.g., BH3, AlH3, etc.
- Electron precise hydrides: These are the hydrides that have an exact number of electrons needed to form normal covalent bonds. e.g. hydrides of group 14 (CH4, SiH4, etc.)
- Electron-rich hydrides: These are the hydrides that have a greater number of electrons than required to form normal covalent bonds. e.g., hydrides of group 15, 16, 17, (NH3, PH3,H2S, HF, HCl, etc). The excess electrons in these hydrides are present as lone pairs of electrons.
Chemical Characteristics
- These hydrides consist of individual covalent molecules. These covalent bonds are weak and have a weak interparticle force
- Molecular hydrides due to their weak covalent bonding have very low melting and boiling points
- Like most non-metal compounds covalent hydrides are poor conductors of electricity
- They can be in a liquid state if their properties are adapted due to hydrogen bonding. Like in the case of water there is a hydrogen bond, and it slightly alters the chemical properties of these compounds.
- As we move across the periodic table the hydrogen compounds with these non-metals become more acidic
- Hydrogen forms the most number of hydrides with Carbon. These are all covalent hydrides, formed by covalent bonding between hydrogen and carbon.
H2(g)+Cl2(g)→2HCl(g)
3H2(g)+N2(g)→2NH3(g)
Metallic or Interstitial Hydrides
- The transition metals and rare earth metals combine with hydrogen to form interstitial hydrides.
- They exhibit metallic properties and are powerful reducing agents.
- They are non-stoichiometric hydrides and their composition varies with temperature and pressure. For e.g., LaH2.76, TiH1.73.
- Metals of group 7, 8 and 9 do not form hydrides and this region of the Periodic Table is called hydride gap.
Characteristics of Metal Hydrides are:
- They are found in a solid state. They have the dark hues of metals
- Again like all metal compounds, they are good conductors of electricity since they have a high thermal capacity.
- They can decompose into metal and hydrogen gas. but this reaction is easily reversible.
Polymeric Hydrides and Complex Hydrides
Polymeric hydrides are formed by elements having electronegativity in the range of 1.4 to 2.0. e.g., (BeH2)n, (AlH3)n, etc. In complex hydrides, H- acts as a ligand and is attached to a central metal atom, e.g., LiAlH4, LiBH4, etc.
FAQs on Hydrides & Hydrogen Economy
| 1. What is the hydrogen economy? |  |
| 2. What are hydrides? |  |
| 3. What are the types of hydrides? |  |
| 4. How do hydrides relate to the hydrogen economy? |  |
| 5. What are some challenges associated with hydrides in the hydrogen economy? |  |






