Integer Answer Type Questions: Heat & Thermodynamics | JEE Advanced | 35 Years Chapter wise Previous Year Solved Papers for JEE PDF Download
Q.1.A metal rod AB of length 10x has its one end A in ice at 0.°C, and the other end B in water at 100 °C. If a point P onthe rod is maintained at 400 °C, then it is found that equal amounts of water and ice evaporate and melt per unit time. The latent heat of evaporation of water is 540 cal/g and latent heat of melting of ice is 80 cal/g. If the point P is at a distance of λx from the ice end A, find the value λ. [Neglect any heat loss to the surrounding.]
Ans.9
Solution. 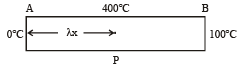
For heat flow from P to 0
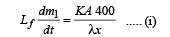
For heat flow from P to B
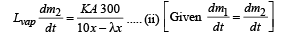
On solving (i) and (ii), we get λ = 9
Q.2. A piece of ice (heat capacity = 2100 J kg–1 °C–1 and latent heat = 3.36 × 105 J kg–1) of mass m grams is at –5°C at atmospheric pressure. It is given 420 J of heat so that the ice starts melting.Finally when the ice-water mixture is in equilibrium, it is found that 1 gm of ice has melted. Assuming there is no other heat exchange in the process, the value of m is
Ans. 8
Solution. Heat supplied = Heat used in converting m grams of ice from –5°C to 0°C + Heat used in converting 1 gram of ice at 0°C to water at 0°
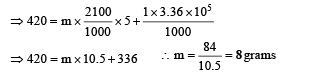
Q.3. Two spherical bodies A (radius 6 cm ) and B(radius 18 cm ) are at temperature T1 and T2, respectively. The maximum intensity in the emission spectrum of A is at 500 nm and in that of B is at 1500 nm. Considering them to be black bodies, what will be the ratio of the rate of total energy radiated by A to that of B?
Ans. 9
Solution. 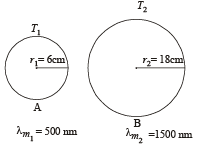
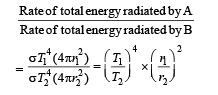
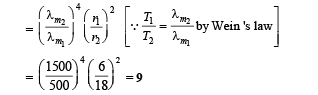
Q.4. A diatomic ideal gas is compressed adiabatically to 1/32 of its initial volume. If the initial temperature of the gas is Ti (in Kelvin) and the final temperature is a Ti, the value of a is
Ans. 4
Solution. For an adiabatic process, the temperature-volume relationship is
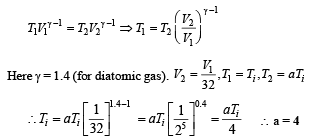
Q.5. Steel wire of lenght ‘L’ at 40°C is suspended from the ceiling and then a mass ‘m’ is hung from its free end. The wire is cooled down from 40°C to 30°C to regain its original length ‘L’. The coefficient of linear thermal expansion of the steel is 10–5 /° C, Young’s modulus of steel is 1011 N/m2 and radius of the wire is 1 mm. Assume that L >>diameter of the wire. Then the value of ‘m’ in kg is nearly
Ans. 3
Solution. We know that
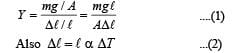
From (1) and (2)
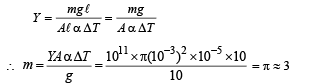
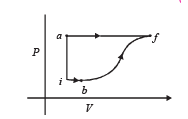
Q.6.A thermodynamic system is taken from an initial state i with internal energy Ui = 100 J to the final state f along two different paths iaf and ibf, as schematically shown in the figure. The work done by the system along the paths af, ib and bf are Waf = 200 J, Wib = 50 J and Wbf =100 J respectively. The heat supplied to the system along the path iaf, ib and bf are Qiaf, Qib and Qbf respectively. If the internal energy of the system in the state b is 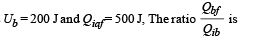
Ans. 2
Solution. Applying first law of thermodynamics to path iaf
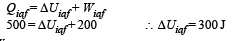
Now,
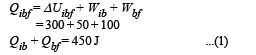
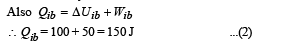
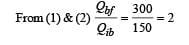
Q.7. Two spherical stars A and B emit blackbody radiation. The radius of A is 400 times that of B and A emits 104 times the power emitted from B. The ratio 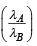 of their wavelengths λA and λB at which the peaks occur in their respective radiation curves is
of their wavelengths λA and λB at which the peaks occur in their respective radiation curves is
Ans.2
Solution. 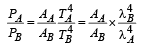
Q.8. A metal is heated in a furnace where a sensor is kept above the metal surface to read the power radiated (P) by the metal.The sensor has a scale that displays log2, (P/P0 ), where P0 is a constant. When the metal surface is at a temperature of 487°C, the sensor shows a value 1. Assume that the emissivity of the metallic surface remains constant. What is the value displayed by the sensor when the temperature of the metal surface is raised to 2767°C?
Ans. 9
Solution. 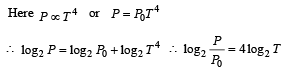
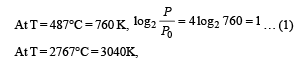
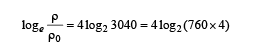
=[4 log 2 760+ log222 ]
= 4log2 760 + 8= 1+ 8= 9
|
347 docs|185 tests
|
|
347 docs|185 tests
|

|
Explore Courses for JEE exam
|

|

















