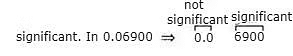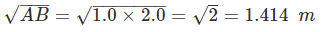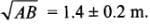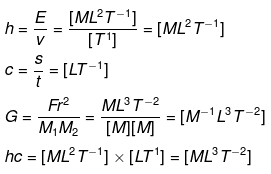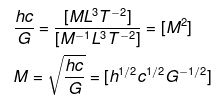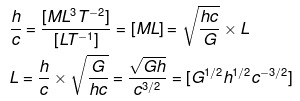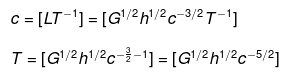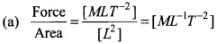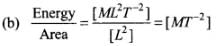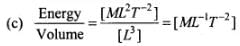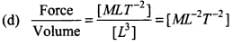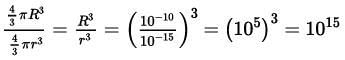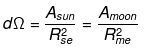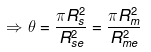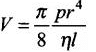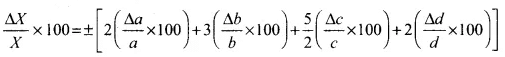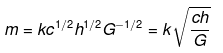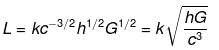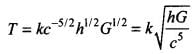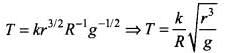NCERT Exemplar: Units & Measurements | Physics Class 11 - NEET PDF Download
| Table of contents |

|
| Multiple Choice Questions |

|
| Very Short Answer Type Questions |

|
| Short Answer Type Questions |

|
| Long Answer Type Questions |

|
Multiple Choice Questions
2.1 The number of significant figures in 0.06900 is
(a) 5
(b) 4
(c) 2
(d) 3
Ans: (b)
Solution:
If the number is less than 1, the zero(s) on the right of the decimal point before the first non-zero digit are not significant. In 0.06900, the zero before the decimal point and the one immediately after the decimal are not significant. Hence, the number of significant figures is four (6900).
Additional: The following rules are observed in counting the number of significant figures in a given measured quantity.
(i) All non-zero digits are significant.
(ii) A zero becomes a significant figure if it appears between two non-zero digits.
(iii) Leading zeros or the zeros placed to the left of the number are never significant.
(iv) Trailing zeros or the zeros placed to the right of the number are significant.
(v) In exponential notation, the numerical portion gives the number of significant figures.
2.2 The sum of the numbers 436.32, 227.2, and 0.301 inappropriate significant figures is
(a) 663.821
(b) 664
(c) 663.8
(d) 663.82
Ans: (c)
Solution:
The result of an addition or subtraction in the number having different precisions should be reported to the same number of decimal places as present in the number having the least number of decimal places.The number with least decimal places is 227.2 is correct to only one decimal place.
436.32
+ 227.2 ← (has only one decimal place)
+ 0.301
------------
663.821 ←(answer should be reported to one decimal place)
------------
The final result should, therefore, be rounded off to one decimal place, i.e. 664.8
2.3 The mass and volume of a body are 4.237 g and 2.5 cm3, respectively. The density of the material of the body in correct significant figures is
(a) 1.6048 g cm–3
(b) 1.69 g cm–3
(c) 1.7 g cm–3
(d) 1.695 g cm–3
Ans: (c)
Solution:
The answer to a multiplication or division is rounded off to the same number of significant figures as possessed by the least precise term used in the calculation. The final result should retain as many significant figures as are there in the original number with the least significant figures. In the given question, density should be reported to two significant figures.
Density = 4.237g/2.5 cm3 = 1.6948 g cm-3
After rounding off the number, we get density = 1.7 g cm-3
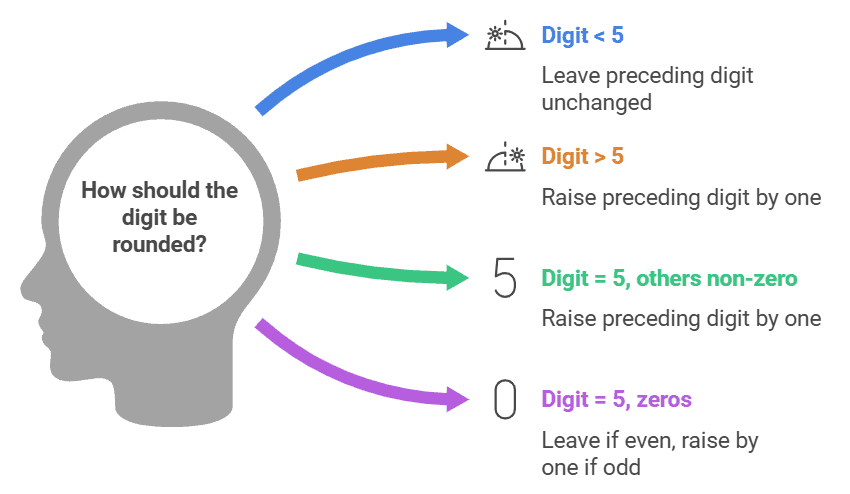
2.4 The numbers 2.745 and 2.735 on rounding off to 3 significant figures will give
(a) 2.75 and 2.74
(b) 2.74 and 2.73
(c) 2.75 and 2.73
(d) 2.74 and 2.74
Ans: (d)
Solution:Let us round off 2.745 to 3 significant figures. Here the digit to be dropped is 5. The preceding digit is 4 which is even so it is left unchanged. Hence on rounding off 2.745, it would be 2.74.
Now consider 2.735, here also the digit to be dropped is 5, then the preceding digit is raised by one, if it is odd. Hence on rounding off 2.735 to 3 significant figures, it would be 2.74. Additionally:
2.5 The length and breadth of a rectangular sheet are 16.2 cm and 10.1 cm, respectively. The area of the sheet with appropriate significant figures and errors is
(a) 164 ± 3 cm2
(b) 163.62 ± 2.6 cm2
(c) 163.6 ± 2.6 cm2
(d) 163.62 ± 3 cm2
Ans: (a)
Solution:
Error in product of quantities: Suppose x = a × b
Let Δa = absolute error in measurement of a,
Δb = absolute error in measurement of b,
Δx = absolute error in the calculation of x, i.e. product of a and b.
The maximum fractional error in x is Δx/x = ± (Δa/a + Δb/b)
Percentage error in the value of x = (Percentage error in value of a) + (Percentage error in value of b)
According to the problem, length l = (16.2 ± 0.1) cm
Breadth b = (10.1 ± 0.1) cm
Area A = l × b= (16.2 cm) × (10.1 cm) = 163.62 cm2
As per the rule area will have only three significant figures and error will have only one significant figure. Rounding off we get, area A = 164 cm2
If ΔA is the error in the area, then the relative error is calculated as ΔA/A.
ΔA/A = Δl/l + Δb/b
= 0.1 cm/16.2 cm + 0.1 cm/10.1 cm
= 1.01 + 1.62/16.2 × 10.1 = 2.63/163.62
⇒ ΔA = A × 2.63/163.62 cm2
= 163.62 × 2.63/163.62 = 2.63cm2
ΔA = 3 cm2 (By rounding off to one significant figure)
Area, A = A ± ΔA = (164 ± 3) cm2
2.6 Which of the following pairs of physical quantities does not have the same dimensional formula?
(a) Work and torque.
(b) Angular momentum and Planck’s constant.
(c) Tension and surface tension.
(d) Impulse and linear momentum.
Ans: (c)
Solution:
(a) Work = Force × displacement = [MLT-2][L]=[ML2T-2]
Torque = force × distance = [MLT-2][L]=[ML2T-2]
Both have the same dimensions.
(b) Angular momentum L = mvr = [M][LT-1][L] = [ML2T-1]
Planck's constant h = F.s/v (∴ E = hv)
= [MLT-2][L]/[T-1] = [ML2T-1]
Dimensions of h and L are equal.
(c) Tension = force = [MLT-2]
Surface tension = Force/i = [MLT-2]/[L] = [ML0T-2]
The dimensions of both are not the same.
(d) Impulse = F × Δt = [MLT-2][T]=[MLT-1]
Momentum = mv = [MLT-1]
Both have the same dimensions.
Additional: One should not be confused with the similar form of tension in both the physical quantities- surface tension and tension. The dimensional formula for both of them is not the same.
2.7 The measure of two quantities along with the precision of the respective measuring instrument is
A = 2.5 m s-1 ± 0.5 m s-1
B = 0.10 s ± 0.01 s
The value of AB will be
(a) (0.25 ± 0.08) m
(b) (0.25 ± 0.5) m
(c) (0.25 ± 0.05) m
(d) (0.25 ± 0.135) m
Ans: (a)
Solution:
A = (2.5 ± 0.5)ms-1
B = (0.10 ± 0.01)s
X = AB = 2.5 × 0.10 = 0.25 m
Δx/x = ΔA/A + ΔB/B
Δx/x = 0.5/2.5 + 0.01./0.10
Δx/x = 0.075/0.25, Δx = 0.007 ≅ 0.08
(Rounding off up to 2 significant figures)
∴ AB = (2.5 ± 0.08) m.
2.8 You measure two quantities as A = 1.0 m ± 0.2 m, B = 2.0 m ± 0.2 m. We should report the correct value for √AB as:
(a) 1.4 m ± 0.4 m
(b) 1.41 m ± 0.15 m
(c) 1.4 m ± 0.3 m
(d) 1.4 m ± 0.2 m
Ans: (d)
Solution:
Quantities A and b are measured quantities so, the number of significant figures in 1.0 m and 2.0 m are two.
rounding off up to minimum numbers of significant figures in 1.0 and 2.0 result must be in 2 significant figures
Δx = 0.2 m (rounding off up to 1 place of decimal)
2.9 Which of the following measurements is most precise?
(a) 5.00 mm
(b) 5.00 cm
(c) 5.00 m
(d) 5.00 km
Ans: (a)
Solution:
Precision is the degree to which several measurements provide answers very close to each other. It is an indicator of the scatter in the data. The lesser the scatter, the higher the precision.
Let us first check the units. In all the options magnitude is the same but the units of measurement are different. As here 5.00 mm has the smallest unit. All given measurements are correct up to two decimal places. However, the absolute error in (a) is 0.01 mm which is the least of all the four. So it is most precise.
2.10 The mean length of an object is 5 cm. Which of the following measurements is most accurate?
(a) 4.9 cm
(b) 4.805 cm
(c) 5.25 cm
(d) 5.4 cm
Ans: (a)
Solution:
Accuracy describes the nearness of a measurement to the standard or true value, i.e. a highly accurate measuring device will provide measurements very close to the standard, true, or known values.
In target shooting, a high score indicates the nearness to the bull’s eye and is a measure of the shooter’s accuracy.
According to the problem, length l = 5 cm
Let us first check the errors in each value by picking options one by one, we get
Δl1 = 5 - 4.9 = 0.1 cm,
Δl2 = 5 - 4.805 = 0.195 cm,
Δl3 = 5.25 - 5 = 0.25 cm and
Δl4 = 5.4 - 5 = 0.4 cm
Error Δl1 is the least.
Hence 4.9 cm is closer to the true value. So, 4.9 is more accurate.
2.11 Young’s modulus of steel is 1.9 × 1011 N/m2. When expressed in CGS units of dynes/cm2, it will be equal to (1 N = 105 dyne, 1 m2 = 104 cm2)
(a) 1.9 × 1010
(b) 1.9 × 1011
(c) 1.9 × 1012
(d) 1.9 × 1013
Ans: (c)
Solution:
According to the problem.
Young's modulus, Y = 1.9 × 1011 N/m2
1 N in SI system of units = 105 dyne in C.G.S system.
Hence, 1.9 × 1011 × 105 dyne/m2
In C.G.S. length is measured in unit ‘cm’, so we should also convert m into cm.
∴ Y = 1.9 × 1011 (105dyne/104cm2) [∵ 1 m = 100 cm]
= 1.9 × 1012 dyne/cm2
2.12 If momentum (P), area (A), and time (T) is taken to be fundamental quantities, then energy has the dimensional formula
(a) (P1 A-1 T1)
(b) (P2 A1 T1)
(c) (P1 A-1/2 T1)
(d) (P1 A1/2 T-1)
Ans: (d)
Solution:
According to the problem, fundamental quantities arc momentum (p), area (A), and time (T) and we have to express energy in these fundamental quantities.
Let energy E,
E ∝ paAATc
⇒ E = kpaAATc
where k is the dimensionless constant of proportionality.
Dimensional formula of energy, [E] = [ML2 T-2] and [p] = [MLT-1]
[A] = [L2], [T] = [T] and [E] = [K][p]a[A]b[T]c
Putting all the dimensions, we get
ML2T-2 = [MLT-1]a[L2]b[T]c
= MaLa+2b T-a+c
According to the principle of homogeneity of dimensions, we get
a = 1 .... (i)
a + 2b = 2 ....(ii)
- a + c = - 2 ... (iii)
By solving these equations (i), (ii) and (iii), we get
a = 1, b = 1/2, c = -1
The dimensional formula for E is [p1 A1/2 T-1].
2.13 On the basis of dimensions, decide which of the following relations for the displacement of a particle undergoing simple harmonic motion is not correct:
(a) y = a sin 2πt/T
(b) y = a sin vt.
(c) 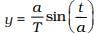
(d) 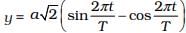
Ans: (b, c)
Solution:
The argument of trigonometric functions (sin, cos, etc.) should be dimensionless. y is displacement and according to the principle of homogeneity of dimensions LHS and RHS.
[Y] = [L], [a] = [L]
[2πt/T] = [T/T]
[vt] = [v] [t] = [LT-1] [T] = [L]
[a/T]/[a]/[T] = [L]/[T] = [LT-1]
[t/a] = [LT-1]
[LHS] ≠ [RHS]
Hence, (c) is not the correct option.
⇒ LHS ≠ RHS.
So, option (b) is also not correct.
2.14 If P, Q, and R are physical quantities, having different dimensions, which of the following combinations can never be a meaningful quantity?
(a) (P – Q)/R
(b) PQ – R
(c) PQ/R
(d) (PR – Q2)/R
(e) (R + Q)/P
Ans: (a, e)
Solution:
Principle of Homogeneity of Dimensions: It states that in a correct equation, the dimensions of each term added or subtracted must be the same. Every correct equation must have the same dimensions on both sides of the equation.
According to the problem P, Q, and R have different dimensions, since, the sum and difference of physical dimensions, are meaningless, i.e., (P – Q) and (R + Q) are not meaningful.
So in options (b) and (c), PQ may have the same dimensions as those of R, and in options (d) PR and Q2 may have the same dimensions as those of R.
Hence, they cannot be added or subtracted, so we can say that (a) and (e) are not meaningful.
2.15 Photon is a quantum of radiation with energy E = hν where ν is frequency and h is Planck’s constant. The dimensions of h are the same as that of
(a) Linear impulse
(b) Angular impulse
(c) Linear momentum
(d) Angular momentum
Ans: (b, d)
Solution:
∴ E = hv
h = E/v = [ML2T-2]/[T-1] = [ML2T-1]
Linear impulse = F.t = dp/dt.dt = dp
= mv = [MLT-1]
Angular impulse =τ.dt = dL/dt.dt = dL = mvr
= [M][LT-1][L] = [ML2T-1]
Linear momentum = mv = [MLT-1]
Angular momentum L = mvr = [ML2T-1]
So the dimensional formulae of h, Angular impulse, and Angular momentum are the same.
2.16 If Planck’s constant (h) and speed of light in vacuum (c) are taken as two fundamental quantities, which one of the following can, in addition, be taken to express length, mass and time in terms of the three chosen fundamental quantities?
(a) Mass of electron (me)
(b) Universal gravitational constant (G)
(c) Charge of electron (e)
(d) Mass of proton (mp)
Ans: (a, b, d)
Solution:
We know that the dimensions of
Hence, physical quantities (a, b, and d) can be used to represent L, M, T in terms of the chosen fundamental quantities.
2.17 Which of the following ratios express pressure?
(a) Force/Area
(b) Energy/Volume
(c) Energy/Area
(d) Force/Volume
Ans: (a, b)
Solution:
Let us first express the relation of pressure with other physical quantities one by one with the help of dimensional analysis.
We know that pressure
So, this ratio expresses pressure (In fact this ratio actually represents pressure).
Dimensions of this ratio are not the same as pressure, so this ratio does not express pressure.
Dimensions of this ratio are the same as pressure, so this ratio also expresses pressure.
Dimensions of this ratio are not the same as pressure, so this ratio does not express pressure.
2.18 Which of the following is not a unit of time?
(a) Second
(b) Parsec
(c) Year
(d) Light year
Ans: (b, d)
Solution:
Parsec and light year are those practical units which are used to measure large distances. For example, the distance between sun and earth or other celestial bodies. So they are the units of length not time. Here, second and year represent time.
Additional: 1 light year (distance that light travels in 1 year with speed = 3 × 108 m/s.) = 9.46 × 1011 m and 1 par see = 3.08 × 1016 m
Very Short Answer Type Questions
2.19 Why do we have different units for the same physical quantity?
Ans: The magnitude of any given physical quantity may vary over a wide range, therefore, different units of the same physical quantity are required.
Example:
(a) Mass ranges from 10-30 kg (for an electron) to 1053 kg (for the known universe). We need different units to measure them like milligrams, grams, kilograms, etc.
(b) The length of a pen can be easily measured in cm, the height of a tree can be measured in meters, the distance between two cities can be measured in kilometers and the distance between two heavenly bodies can be measured in a light year.
2.20 The radius of an atom is of the order of 1 Å and the radius of a nucleus is of the order of fermi. How many magnitudes higher is the volume of the atom as compared to the volume of the nucleus?
Ans: Radius (R) of atom = 1 = 10-10 m
Radius (r) of nucleus = 1 fermi =10-15 m
The ratio of the volume of an atom to the nucleus
2.21 Name the device used for measuring the mass of atoms and molecules.
Ans: A mass spectrograph is a device that is used for measuring the mass of atoms and molecules.
2.22 Express unified atomic mass unit in kg.
Ans: The unified atomic mass unit is the standard unit that is used for indicating mass on an atomic or molecular scale (atomic mass). One unified atomic mass unit is approximately the mass of one nucleon (either a single proton or neutron) and is numerically equivalent to 1 g/mol. It is defined as one-twelfth of the mass of an unbound neutral atom of carbon-12 in its nuclear and electronic ground state.
2.23 A function f (θ) is defined as: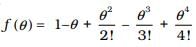
Why is it necessary for f(θ) to be a dimensionless quantity?
Ans: θ is represented by an angle which is equal to
so angle θ is a dimensionless physical quantity. The first term is 1 which is dimensionless, next term contains only powers of θ, as θ is dimensionless so their powers will also be dimensionless. Hence, each term in the R.H.S. expression is dimensionless so the left-hand side f(θ) must be dimensionless.
2.24 Why length, mass, and time are chosen as base quantities in mechanics?
Ans: Normally each physical quantity requires a unit or standard for its specification, so it appears that there must be as many units as there are physical quantities. However, it is not so. It has been found that if in mechanics we choose arbitrarily units of any three physical quantities we can express the units of all other physical quantities in mechanics in terms of these. So, length, mass, and time are chosen as base quantities in mechanics because
(a) Length, mass, and time cannot be derived from one another, that is these quantities are independent.
(b) All other quantities in mechanics can be expressed in terms of length, mass, and time.
Short Answer Type Questions
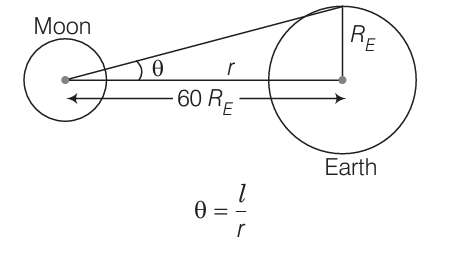
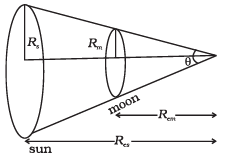
2.25 (a) The earth-moon distance is about 60 earth radius. What will be the diameter of the earth (approximately in degrees) as seen from the moon?
(b) The moon is seen to be off (½)° diameter from the earth. What must be the relative size compared to the earth?
(c) From parallax measurement, the sun is found to be at a distance of about 400 times the earth-moon distance. Estimate the ratio of sun-earth diameters.
Ans:
(a) As the distance between the moon and the earth is greater than the radius of the earth, then the radius of the earth can be treated as an arc.
According to the problem,
RE = length of arc
Distance between moon and earth = 60RE
So, the angle subtended at distance r due to an arc of length l is
θE = l/r = RE/60RE = 1/60 rad
= (1/60 x 180°/π)degree
= (3°/3.14)degree ≈1°
Hence, the angle subtended by the diameter of the earth 2θ = 2°.
(b) According to the problem, the moon is seen as (1/2)° diameter from Earth, and the earth is seen as 2° diameter from the moon.
As θ is proportional to the diameter. Hence,(c) From parallax measurement given that the Sun is at a distance of about 400 times the earth-moon distance, hence, rsun/rmoon = 400
(Suppose, here r stands for distance and D for diameter) The sun and moon both appear to be of the same angular diameter as seen from the Earth.
∴ Dsun/rsun = Dmoon/rmoon
∴ Dsun/Dmoon = 400
But Dearth/Dmoon = 4
∴ Dsun/Dearth = 100.
2.26 Which of the following time-measuring devices is most precise?
(a) A wall clock.
(b) A stopwatch.
(c) A digital watch.
(d) An atomic clock.
Give a reason for your answer.
Ans: (d)
Solution:
The least count of a wall clock, stopwatch, digital Watch, and atomic clock are 1 sec, 1/10 sec, 1/100 sec, and 1/1013 sec respectively. So atomic clock is the most precise.
2.27 The distance of a galaxy is of the order of 1025 m. Calculate the order of magnitude of time taken by light to reach us from the galaxy.
Ans. According to the problem, the distance of the galaxy = 1025m.
Speed of light = 3 × 108 m/s
Hence, the time taken by light to reach us from the galaxy is
t = Distance/Speed
= (1025 m/3 × 108 m/s) ≈ 1/3 × 1017
= 10/3 × 1016 = 3.33 × 1016s
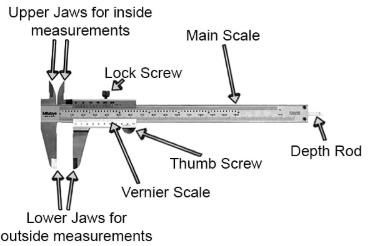
2.28 The vernier scale of a travelling microscope has 50 divisions which coincide with 49 main scale divisions. If each main scale division is 0.5 mm, calculate the minimum inaccuracy in the measurement of distance.
Ans. A diagram of the Vernier caliper is shown below.
According to the problem, 50 divisions of the Vernier scale coincide with 49 main scale divisions.
50 VSD = 49 MSD
⇒ 1 MSD = 50/49 VSD or 1 VSD
= 49/50 MSD
where MSD = Main scale division and VSD = Vernier scale division.
We know that
Minimum inaccuracy = Vernier constant
= 1 MSD - 1 VSD
= 1 MSD - (49/50) MSD
= (1/50) MSD
It is given in the problem that 1 MSD = 0.5 mm
Hence, minimum inaccuracy = 1/50 × 0.5 mm = 1/100 = 0.01 mm
2.29 During a total solar eclipse the moon almost entirely covers the sphere of the sun. Write the relation between the distances and sizes of the sun and moon.
Ans. The Diagram given below shows that the moon almost entirely covers the sphere of the sun.
Rme = Distance of moon from earth
Rse = Distance of sun from earth
Let the solid angle made by the sun and moon be dΩ, we can write
Here, Asun = Area of the sun
Amoon = Area of the moon
⇒ (Rs/Rse)2 = (Rm/Rme)2
⇒ Rs/Rse = Rm/Rme or Rs/Rm = Rse/Rme
(Here, the radius of the sun and moon represent their sizes respectively)Additional: In geometry, a solid angle (symbol: Ω or w) is the two dimensional angle in three-dimensional space that an object subtends at a point. It is a measure of how large the object appears to an observer looking from that point. In the International System of Units (SI), a solid angle is expressed in a dimensionless unit called a steradian (symbol: sr).
A small object nearby may subtend the same solid angle as a larger object farther away. For example, although the Moon is much smaller than the Sun, it is also much closer to Earth.
2.30 If the unit of force is 100 N, the unit of length is 10 m and the unit of time is 100 s, what is the unit of mass in this system of units?
Ans. First, write the dimensions of each quantity and then relate them.
Force [F] = [MLT-2] = 100 N ...(i)
Length [L] = [L] = 10 m ...(ii)
Time [t] = [T] = 100 s ...(iii)
Substituting values of L and T from Eqs. (ii) and (iii) in Eq. (i), we get
⇒ M × 10 × (100)-2 = 100
⇒ M × 10/100 × 100 = 100
⇒ M = 100 × 1000 kg = 105 kg
2.31 Give an example of
(a) A physical quantity that has a unit but no dimensions.
(b) A physical quantity that has neither unit nor dimensions.
(c) A constant that has a unit.
(d) A constant that has no unit.
Ans. (a) Solid angle Ω = A/r2 steradian and a Plane angle θ = L/r radian. Both are dimensionless but have units.
(b) Specific density = density of medium/density of water at 4°C
(c) It is a ratio of two quantities. So, it is a unitless and dimensionless constant.
(c) Gravitational constant (G) = 6.67 × 10-11 N-m2/kg2
(d) Reynold’s number is a constant that has no unit.
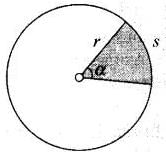
2.32 Calculate the length of the arc of a circle of radius 31.0 cm which subtends an angle of π/6 at the centre.
Ans. Plance Angle α = Arc length/Radius = s/r
According to the problem, θ = π/6 = s/31 cm
Hence, length of arc = s = 31 × (π/6)cm = (31 × 3.14)/6 = 16.22 cm
Rounding off to three significant figures it would be 16.2 cm.
2.33 Calculate the solid angle subtended by the periphery of an area of 1 cm2 at a point situated symmetrically at a distance of 5 cm from the area.
Ans. Solid angle, Ω = Area/(Radial distance)2
= ((1 cm2)/(5 cm)2) = 1/25 = 4 × 10-2 steradian
(∵ Area = 1 cm2, distance = 5 cm)
Important point: Please keep in mind that a solid angle is for 3-D figures like spheres, cones, etc and a plane angle is for plane objects or 2-D figures like circles, arcs, etc.
2.34 The displacement of a progressive wave is represented by y = A sin(wt – kx), where x is distance and t is time. Write the dimensional formula of (i) ω and (ii) k.
Ans. We have to apply the principle of homogeneity to solve this problem. The principle of homogeneity states that in a correct equation, the dimensions of each term added or subtracted must be the same, i.e., the dimensions of LHS and RHS should be equal.
According to the problem
y = A sin(ωt - kx)
Here y = [L] hence
A sin(ωt - kx) = [L]
Here A = [L], which peak value of y
So, ωt - kx should be dimensionless,
(i) [ωt] = constant
⇒ [ω] = [T-1]
(ii) [kx] = constant
⇒ m = [L-1]
2.35 Time for 20 oscillations of a pendulum is measured as t1 = 39.6 s; t2= 39.9 s; t3 = 39.5 s. What is the precision of the measurements? What is the accuracy of the measurement?
Ans. According to the problem, the time for 20 oscillations of a pendulum,
t1 = 39.6 s, t2 = 39.9 s and t3 = 39.5 s
It is quite obvious from these observations that the least count of the watch is 0.1 s. As measurements have only one decimal place. Precision in the measurement = Least count of the measuring instrument= 0.1 s
Precision in 20 oscillations = 0.1
Therefore, Precision in 1 oscillation = 0.1/20 = 0.005
(ii) Mean value of time for 20 oscillations is given by
t = (t1 + t2 + t3)/3
= (39.6 + 39.9 + 39.5)/3 = 39.66 s
Mean time period of the second pendulum = 39.66/20 ≈ 1.98 s
Rounding off the time period of the second pendulum = 2 s
Measured time period of the second pendulum = 2 - 0.005 = 1.995 s
Accuracy of measurement is the maximum observed error and is given by = 1.995 - 1.980 = 0.015 s
Long Answer Type Questions
2.36 A new system of units is proposed in which the unit of mass is α kg, the unit of length β m, and the unit of time γ s. How much will 5 J measure in this new system?
Ans. Let the physical quantity be Q = n1u1 = n2u2
Let M1, L1, T1, and M2, L2, T2 are units of mass, length, and time in the given two systems.
So, n2 = n1 [M1/M2]a x [L1/L2]b x [T1/T2]c
We know that the dimension of energy
[U] = [ML2T-2]
According to the problem, M1 = 1 kg, L1 = 1 m, T1 = 1 s
M2 = α kg, L2 = β m, T2 = γs
Substituting the values, we get
n2 = 5[M1/M2] × [L1/L2]2 × [T1/T2]-2
= 5[(1/α)kg] × [(1/β)m]2 × [(1/γ)s]-2
= 5 × (1/α) × (1/β2) × (1/γ)-2 = 5γ2/αβ2 J
This is the required value of energy in the new system of units.
2.37 The volume of a liquid flowing out per second of a pipe of length l and radius r is written by a student as
where P is the pressure difference between the two ends of the pipe and η is the coefficient of viscosity of the liquid having dimensional formula ML–1 T–1.
Check whether the equation is dimensionally correct.
Ans. If the dimensions of LHS of an equation are equal to the dimensions of RHS, then the equation is said to be dimensionally correct.
According to the problem, the volume of a liquid flowing out per second of a pipe is given by
(where, V = rate of volume of liquid per unit time)
Dimension of given physical quantities,
[V] = Dimension of volume/Dimension of time = [L3]/[T] = [L3T-1],[p]=[ML-1T-2],
[η] = [ML-1T-1], [l] = [L],[r] = [L]
LHS = [V] = [L3]/[T] = [L3T-1]
RHS = [ML-1T-2] × [L4]/[ML-1T-1] × [L] = [L3T-1]
Dimensionally, L.H.S. = R.H.S.
Therefore, the equation is correct dimensionally.
2.38 A physical quantity X is related to four measurable quantities a, b, c, and d as follows: X = a2 b3 c5/2 d–2. The percentage error in the measurement of a, b, c, and d are 1%, 2%, 3%, and 4%, respectively. What is the percentage error in quantity X? If the value of X calculated on the basis of the above relation is 2.763, to what value should you round off the result?
Ans. Percentage error in quantity X is given by, (Δx/x) x 100
According to the problem, physical quantity is X = a2 b3 c5/2 d-2
percentage error in a = ((Δa/a) × 100) = 1%
percentage error in b = ((Δb/b) × 100) = 2%
percentage error in c = ((Δc/c) × 100) = 3%
percentage error in d = ((Δd/d) × 100) = 4%
The maximum percentage error in X is
= ±[2(1) + 3(2) + (5/2)3 + 2(4)]%
= ±[2 + 6 + (15/2) + 8] = ± 23.5%
∴ Percentage error in quantity X = ±23.5%
Mean absolute error in X = ±0.235 = ±0.24 (rounding-off upto two significant digits)
On the basis of these values, the value of X should have two significant digits only.
∴ X = 2.8
2.39. In the expression P = El2 m–5 G–2, E, m, l, and G denote energy, mass, angular momentum, and gravitational constant, respectively. Show that P is a dimensionless quantity.
Ans. According to the problem, the expression is P = El2 m–5 G–2 where E is energy [E] = [ML2T-2], m is mass [w] = [M], L is angular momentum [L] = [ML2 T-1], G is gravitational constant [G] = [M-1 L2T-2]
Substituting dimensions of each physical quantity in the given expression
[P] = [ML2T-2] × [ML2Y-1]2 × [M]-5n × [M-1L3T-2]-2
= [M1+2-5+2 L2+4-6 T-2-2+4]
=[M0L0T0]
This shows that P is a dimesionless quantity.
2.40 If the velocity of light c, Planck’s constant h, and gravitational constant G are taken as fundamental quantities then express mass, length, and time in terms of dimensions of these quantities.
Ans. We have to apply the principle of homogeneity to solve this problem. The principle of homogeneity states that in a correct equation, the dimensions of each term added or subtracted must be the same, i.e., dimensions of LHS and RHS should be equal,
We know that, the dimensions of
[h] = [ML2T-1], [c] = [LT-1], [G] = [M-1 L3 T-2]
(i) Let m ∝ cxhyGz
⇒ m = kcahbGc ....(i)
where k is a dimensionless constant of proportionality.
Substituting the dimensions of each term in Eq. (i), we get
[ML0T0] = [LT-1]a × [ML2T-1]b[M-1L3T2]c
Comparing powers of the same terms on both sides, we get
b - c = 1 ...(ii)
a + 2b + 3c = 0 .....(iii)
- a - b - 2c = 0 ....(iv)
Adding Eqs. (ii), (iii) and (iv). we get
⇒ 2b = 1
⇒ b = 1/2
Substituting the value of b in Eq. (ii). we get
c = -1/2
From Eq. (iv)
a = - b - 2c
Substituting values of b and c, we get
a = -(1/2)-2(-1/2)=1/2
Putting values of a, b, and c in Eq. (i), we get
(ii) Let L ∝ cahbGc
⇒ L = kcahbGc ....(v)
where k is a dimensionless constant.
Substituting the dimensions of each term in Eq. (v), we get
[M0LT0] = [LT-1]a x [ML2T-1]b x [M-1L3T-2]c
=[Mb-c La+2b+3c T-a-b-2c]
On comparing powers of the same terms, we get
b - c = 0 ....(vi)
a + 2b + 3c = 1 .....(vii)
- a - b - 2 c = 0 ......(viii)
Adding Eqs. (vi), (vii) and (viii), we get
2b = 1 ⇒ b = 1/2
Substituting the value of b in Eq. (vi), we get
c = 1/2
From Eq. (viii), a = -b - 2c
Substituting values of b and c, we get
a = -(1/2)-2(1/2) = -3/2
Putting values of a, b and c in Eq. (v), we get
(iii) Let T ∝ cahbGc
⇒ T = cahbGc ....(ix)
where k is a dimensionless constant.
Substituting the dimensions of each term in Eq. (ix), we get
[M0L0T1] = [LT-1]a x [ML2T-1]b x [M-1L3T-2]c
=[Mb-c La+2b+3c T-a-b-2c]
On comparing powers of the same terms, we get
b - c = 0 ....(x)
a + 2b + 3c = 1 ....(xi)
-a - b - 2c = 1 ....(xii)
Adding Eqs. (x), (xi), and (xii), we get
2b = 1 ⇒ b = 1/2
Substituting the value of b in Eq. (x), we get
c = b = 1/2
From Eq. (xii),
a = -b - 2c - 1
Substituting values of b and c, we get
a = -(1/2)-2(1/2)-1 = -5/2
Putting values of a, b, and c in Eq. (ix), we get
2.41 An artificial satellite is revolving around a planet of mass M and radius R, in a circular orbit of radius r. From Kepler’s Third law about the period of a satellite around a common central body, the square of the period of revolution T is proportional to the cube of the radius of the orbit r. Show using dimensional analysis, that 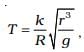
where k is a dimensionless constant and g is the acceleration due to gravity.
Ans. According to Kepler’s third law, T2 ∝ a3 i.e., the square of time period (T2) of a satellite revolving around a planet, is proportional to the cube of the radius of the orbit (a3).
We have to apply Kepler’s third law,
T2 ∝ r3
⇒ T ∝ r3/2
Also, T depends on R and g.
Let T ∝ r3/2gaRb
⇒ T = kr3/2Ragb ....(i)
where A: is a dimensionless constant of proportionality.
Writing the dimensions of various quantities on both sides, we get
[M0L0T] = [L]3/2[LT-2]a[L]b
= [M0La+b+3/2T-2a]
On comparing the dimensions of both sides, we get
a + b + (3/2) = 0
-2a = 1 ⇒ a = -1/2 ....(iii)
From Eq. (ii), we get
b - (1/2) + (3/2) = 0
⇒ b = -1
Substituting the values of a and b in Eq. (i), we get
2.42 In an experiment to estimate the size of a molecule of oleic acid 1 mL of oleic acid is dissolved in 19 mL of alcohol. Then 1 mL of this solution is diluted to 20 mL by adding alcohol. Now 1 drop of this diluted solution is placed on water in a shallow trough. The solution spreads over the surface of water forming one molecule thick layer. Now, lycopodium powder is sprinkled evenly over the film and its diameter is measured. Knowing the volume of the drop and the area of the film we can calculate the thickness of the film which will give us the size of the oleic acid molecule.
Read the passage carefully and answer the following questions:
(a) Why do we dissolve oleic acid in alcohol?
(b) What is the role of lycopodium powder?
(c) What would be the volume of oleic acid in each mL of solution prepared?
(d) How will you calculate the volume of n drops of this solution of oleic acid?
(e) What will be the volume of oleic acid in one drop of this solution?
Ans. (a) Since Oleic acid does not dissolve in water, hence it is dissolved in alcohol
(b) Lycopodium powder spreads on the entire surface of water when it is sprinkled evenly. When a drop of prepared solution of oleic acid and alcohol is dropped into water, oleic acid does not dissolve in water. Instead, it spreads on the water surface pushing the lycopodium powder away to clear a circular area where the drop falls. We can thus be able to measure the area over which oleic acid spreads.
(c) Since 20 mL (1 mL oleic acid + 19 mL alcohol) contains 1 mL of oleic acid, oleic acid in each mL of the solution =1/20 mL. Further, this 1 mL is diluted to 20 mL by adding alcohol. In each mL of the solution prepared, the volume of oleic acid = 1/20 mL x 1/20 = 1/400 mL
(d) The volume of n drops of this solution of oleic acid can be calculated by means of a burette (used to make solution in the form of countable drops) and measuring cylinder and measuring the number of drops.
(e) As 1 mL of solution contains n number of drops, then the volume of oleic acid in one drop will be = 1/(400)n mL
2.43 (a) How many astronomical units (A.U.) make 1 parsec?
(b) Consider a sun-like star at a distance of 2 parsecs. When it is seen through a telescope with 100 magnification, what should be the angular size of the star? Sun appears to be (1/2)° from the earth. Due to atmospheric fluctuations, the eye can’t resolve objects smaller than 1 arc minute.
(c) Mars has approximately half of the Earth’s diameter. When it is closest to the earth it is at about 1/2 A.U. from the earth. Calculate what size it will appear when seen through the same telescope.
Ans. (a) By definition of parsec
∴ 1 parsec = (1A.U./1arc sec)
1 deg = 3600 arc sec
∴ 1 arcsec = π/(3600 x 180) radians
∴ 1 parsec = ((3600 x 180)/π )A.U.
= 206265 A.U. ≈ 2 x 105 A.U.
(b) At 1 A.U. distance, the sun is (1/2°) in diameter.
Therefore, at 1 parsec, the star is (1/2)/(2x105) degree in diameter = 15 × 10-5 arcmin. With 100 magnification, it should look 15 × 10-3 arcmin. However, due to atmospheric fluctuations, it will still look at about 1 arcmin. It can’t be magnified using a telescope.
(c) From parallax measurement given that the Sun is at a distance of about 400 times the earth-moon distance, hence, rsun/rmoon = 400
(Suppose, here r stands for distance and D for diameter) The sun and moon both appear to be of the same angular diameter as seen from the Earth.
∴ Dsun/rsun = Dmoon/rmoon
Dmars/Dearth = 1/2, Dearth/Dsun = 1/400
∴ Dmars/Dsun = 1/800.
At 1 A.U. sun is seen as 1/2 degree in diameter, and Mars will be seen as 1/1600 degree in diameter.
At 1/2 A.U., mars will be seen as 1/800 degree in diameter. With 100 magnification mars will be seen as 1/8 degree = 60/8 = 7.5 arcmin.
This is larger than the resolution limit due to atmospheric fluctuations. Hence, it looks magnified.
2.44 Einstein’s mass-energy relation emerging out of his famous theory of relativity relates mass (m) to energy (E) as E = mc2, where c is the speed of light in a vacuum. At the nuclear level, the magnitudes of energy are very small. The energy at the nuclear level is usually measured in MeV, where 1 MeV = 1.6×10–13J; the masses are measured in the unified atomic mass unit (u) where 1u = 1.67 × 10–27 kg.
(a) Show that the energy equivalent of 1 u is 931.5 MeV.
(b) A student writes the relation as 1 u = 931.5 MeV. The teacher points out that the relation is dimensionally incorrect. Write the correct relation.
Ans. We can apply Einstein’s mass-energy relation in this problem, E = mc2, to calculate the energy equivalent of the given mass.
Here
1 amu = 1 u = 1.67 × 10-27 kg
Applying E = mc2
Energy E = (1.67 × 10-27)(3 × 108)2J
E = 1.67 × 9 × 10-11 J
E = (1.67 × 9 × 10-11/1.6 × 10-13) MeV
E = 939.4 MeV ≈ 931.5 MeV
(b) As E = mc2
⇒ m = E/c2
According to this, 1u = 931.5 Mev/c2
Hence the dimensionally correct relation 1 amu * c2 = 1 u * c2 = 931.5 MeV
|
96 videos|367 docs|98 tests
|
FAQs on NCERT Exemplar: Units & Measurements - Physics Class 11 - NEET
| 1. What are the SI units of length, mass, and time? |  |
| 2. What is the difference between precision and accuracy in measurements? |  |
| 3. How can the dimensions of a physical quantity be determined? |  |
| 4. What is the significance of significant figures in measurements? |  |
| 5. Can the magnitude of a physical quantity be negative? |  |