NCERT Exemplar: Dual Nature of Radiation and Matter | Physics for Airmen Group X - Airforce X Y / Indian Navy SSR PDF Download
MULTIPLE CHOICE QUESTIONS
Q.1. A particle is dropped from a height H. The de Broglie wavelength of the particle as a function of height is proportional to
(a) H
(b) H1/2
(c) H0
(d) H–1/2
Ans. (d)
Solution.
According to de-Broglie a moving material particle sometimes acts as a wave and sometimes as a particle.
The wave associated with a moving particle is called matter wave or de-Broglie wave and it propagates in the form of wave packets with group velocity. According to de-Broglie theory, the wavelength of de-Broglie wave is given by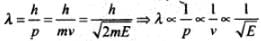
Where h = Planck's constant, m = Mass of the particle, v = Speed of the particle, E = Energy of the particle.
For a body falling freely under gravity from a height H, final velocity of v is obtained by applying kinematic equations
v2 - u2 = 2aH ⇒ v2 - 02 = 2gH.
or, 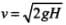
We define de-broglie wavelength as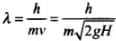
⇒ 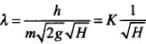
Where 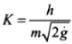 = Constant
= Constant
⇒ λ ∝ 1/√H. Hence λ ∝ H-1/2 .
Q.2. The wavelength of a photon needed to remove a proton from a nucleus which is bound to the nucleus with 1 MeV energy is nearly
(a) 1.2 nm
(b) 1.2 × 10–3 nm
(c) 1.2 × 10–6 nm
(d) 1.2 × 10 nm
Ans. (b)
Solution.
According to Einstein’s quantum theory light propagates in the bundles (packets or quanta) of energy, each bundle being called a photon and possessing energy.
Energy of photon is given by
E = hv = hc/λ; where c = Speed of light, h = Planck's constant = 6.6 x 10-34 J-sec, v = Frequency in Hz, λ = the minimum wavelength of the photon required to eject the proton from nucleus.
In electron volt, 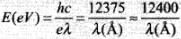
According to the problem,
Energy of a photon, E = 1 MeV or 106 eV
Now, hc = 1240 eV nm
Now, E = hc/λ
⇒ 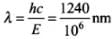
= 1.24 x 10-3 nm
Q.3. Consider a beam of electrons (each electron with energy E0) incident on a metal surface kept in an evacuated chamber. Then
(a) No electrons will be emitted as only photons can emit electrons.
(b) Electrons can be emitted but all with an energy, E0.
(c) Electrons can be emitted with any energy, with a maximum of E0 – φ (φ is the work function).
(d) Electrons can be emitted with any energy, with a maximum of E0.
Ans. (d)
Solution.
If a beam of electrons of having energy E0 is incident on a metal surface kept in an evacuated chamber.
The electrons can be emitted with maximum energy E0 (due to elastic collision) and With any energy less than E0, when part of incident energy of electron is used in liberating the electrons from the surface of metal.
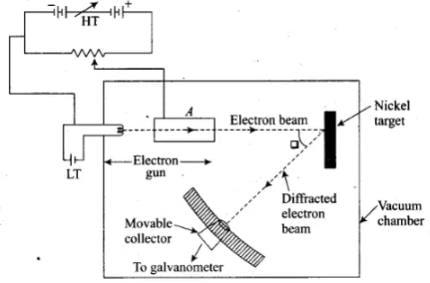
Q.4. Consider Fig. in the NCERT text book of physics for Class XII. Suppose the voltage applied to A is increased. The diffracted beam will have the maximum at a value of θ that
(a) Will be larger than the earlier value.
(b) Will be the same as the earlier value.
(c) Will be less than the earlier value.
(d) Will depend on the target.
Ans. (c)
Solution.
Davison and Germer Experiment:
1. It is used to study the scattering of electron from a solid or to verify the wave nature of electron. A beam of electrons emitted by an . electron gun is made to fall on nickel crystal cut along cubical axis at a particular angle. Ni crystal behaves like a three dimensional diffraction grating and it diffracts the electron beam obtained from electron gun.
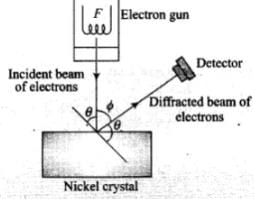
2. The diffracted beam of electrons is received by the detector which can be positioned at any angle by rotating it .about the point of incidence. The energy of the incident beam of electrons can also be varied by changing the applied voltage to the electron gun.
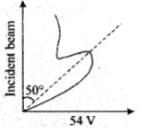
According to classical physics, the intensity of scattered beam of electrons at all scattering angle will be same but Davisson and Germer found that the intensity of scattered beam of electrons was not the same but different at different angles of scattering. It is maximum for diffracting angle 50° at 54 volt potential difference.
3. If the de-Broglie waves exist for electrons then these should be diffracted as X-rays. Using the Bragg’s formula 2d sinθ = nλ, we can determine the wavelength of these waves.
The de-Broglie wavelength associated with electron is where V is the applied voltage.
Using the Bragg’s formula we can determine the wavelength of these waves. If there is a maxima of the, diffracted electrons at an angle θ, then
2d sin θ = A (ii)
From Eq. (i), we note that if V is inversely proportional to the wavelength λ. i.e., V will increase with the decrease, in λ.
From Eq. (ii), we note that wavelength λ is directly proportional to sinθ and hence θ.
So, with the decrease in λ , θ will also decrease.
Thus, when the voltage applied to A is increased. The diffracted beam will have the maximum at a value of θ that will be less than the earlier value.
4. A proton, a neutron, an electron and an a-particle have same energy. Then, their de-Broglie wavelengths compare as
The de-Broglie wavelength associated with electron is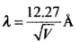 ...(i)
...(i)
where V is the applied voltage.
Using the Bragg's formula we can determine the wavelength of these waves. If there is a maxima of the diffracted electrons at an angle θ, then
2d sin θ = λ ...(ii)
From Eq.(i), we note that if V is inversely proportional to the wavelength λ. i.e., V will increase with the decrease in λ.
From Eq. (ii), we note that wavelength λ is directly proportional to sin θ and hence θ.
So, with the decrease in λ, θ will also decrease.
Thus, when the voltage applied to A is increased. The diffracted beam will have the maximum at a value of θ that will be less than the earlier value.
Q.5. A proton, a neutron, an electron and an α-particle have same energy. Then their de Broglie wavelengths compare as
(a) λp = λn > λe > λα
(b) λα < λp = λn > λe
(c) λe < λp = λn > λα
(d) λe = λp = λn = λα
Ans. (b)
Solution.
de - Broglie wavelength λd = h/p
EP = En = Ee = E∝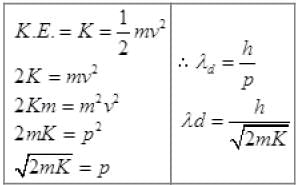
Or λd = 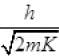 [as h and E (K.E) is constt.]
[as h and E (K.E) is constt.]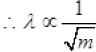
Mα > mp = mn > me
∴ λα < λp = λn < λe
Q.6. An electron is moving with an initial velocity v = v0î and is in a magnetic field 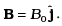 . Then it’s de Broglie wavelength
. Then it’s de Broglie wavelength
(a) Remains constant.
(b) Increases with time.
(c) Decreases with time.
(d) Increases and decreases periodically.
Ans. (a)
Solution.
If a particle is carrying a positive charge q and moving with a velocity v enters a magnetic field 5 then it experiences a force F which is given by the expression
F = q(v x B)=$ F = qvB sin θ. As this force is perpendicular to v and B , so the magnitude of v will not change, i.e. momentum (p = mv) will remain constant in magnitude. Hence,
According to the problem, 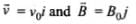
Magnetic force on moving electron = -e[v0i x B0j] ⇒ = -ev0B0k
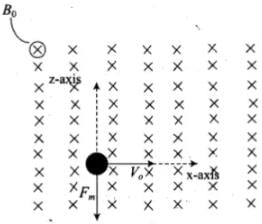
As this force is perpendicular to 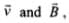 so the magnitude of v will not change, i.e., momentum (p = mv) will remain constant in magnitude. Hence, de-Broglie wavelength λ = h/mv remains constant.
so the magnitude of v will not change, i.e., momentum (p = mv) will remain constant in magnitude. Hence, de-Broglie wavelength λ = h/mv remains constant.
Q.7. An electron (mass m ) with an initial velocity v = v0î (v0> 0) is in an electric field E = – E0î(E0 = constant > 0). It’s de Broglie wavelength at time t is given by
(a) 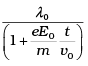
(b) 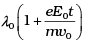
(c) λ0
(d) λ0t
Ans. (a)
Solution.
The wave associated with moving particle is called matter wave or de-Broglie wave and it propagates in the form of wave packets with group velocity. According to de-Broglie theory, the wavelength of de- Broglie wave is given by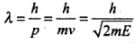
As initial velocity of the electron is v0î, the initial de-Broglie wavelength of electron,
λ0 = h/mv0 ...(i)
Electrostatic force on electron in electric field is,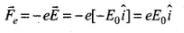
Acceleration of electron, 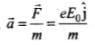
Velocity of the electron after time t,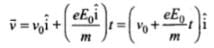
⇒ 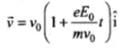
de-Broglie wavelength associated with electron at time t is λ = h/mv
⇒ 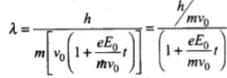
⇒ 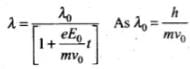
Q.8. An electron (mass m ) with an initial velocity v = v0î is in an electric field 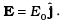 If λ0 = h/mv0, it’s de Broglie wavelength at time t is given by
If λ0 = h/mv0, it’s de Broglie wavelength at time t is given by
(a) λ0
(b) 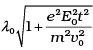
(c) 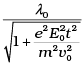
(d) 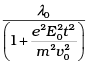
Ans. (c)
Solution.
According to the problem de-Broglie wavelength of electron at time t=0.
is λ0 = h/mv0.
Electrostatic force on electron in electric field is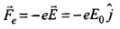
The acceleration of electron, 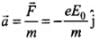
It is acting along negative y-axis.
The initial velocity of electron along x-axis, vx0 = v0î.
This component of velocity will remain constant as there is no force on electron in this direction.
Now considering y-direction. Initial velocity of electron along y-axis, vy0 = 0.
Velocity of electron after time t along y-axis,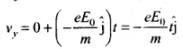
Magnitude of velocity of electron after time t is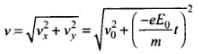
⇒ 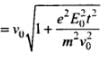
de-Broglie wavelength, λ' = h/mv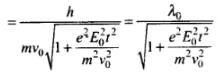
⇒ 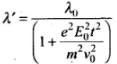
Q.9. Relativistic corrections become necessary when the expression for the kinetic energy 1/2 mv2, becomes comparable with mc2, where m is the mass of the particle. At what de Broglie wavelength will relativistic corrections become important for an electron?
(a) λ = 10 nm
(b) λ = 10–1 nm
(c) λ = 10–4 nm
(d) λ = 10–6 nm
Ans. (c, d)
Solution.
de-Broglie wavelength λ = h/mv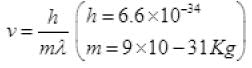
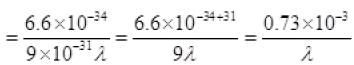
For option
(a) λ = 10nm =10 × 10-9m = 10-8 m
∴ v = 7.3 × 10-4 × 10+8 = 7.3 × 104 < 3 × 108 (speed of light)
(b) λ=10-1nm = 10-1 × 10-9 m = 10-10m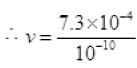 = 7.3 x 10-4+10 = 7.3 x 106 ≈ 107 < 108 (speed of light)
= 7.3 x 10-4+10 = 7.3 x 106 ≈ 107 < 108 (speed of light)
(c) λ = 10-4nm = 10-4 x 10-9 m = 10-13 m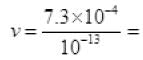 7.3 x 10-4+13 = 7.3 x 109 ≈ 1010 > 108 (speed of light)
7.3 x 10-4+13 = 7.3 x 109 ≈ 1010 > 108 (speed of light)
(d) λ = 10-6nm = 10-15m
v= 7.3 × 10-4 × 1015 = 7.3 × 1011 ≈ 1012 > 108 (Speed of light)
So the velocity of electron is more for option (c) and (d) where the relativistic correction become necessary although the speed of electron is 7.3 x 106 m/s is comparable with (c) speed of light, there must be relativistic correction.
Q.10. Two particles A1 and A2 of masses m1, m2 (m1 > m2) have the same de Broglie wavelength. Then
(a) Their momenta are the same.
(b) Their energies are the same.
(c) Energy of A1 is less than the energy of A2.
(d) Energy of A1 is more than the energy of A2.
Ans. (a, c)
Solution.
We know that de-Broglie wavelength λ = h/mv
where, mv = p(momentum) of the particle
⇒ But we can express wavelength 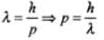
Here, h is Planck constant.
Hence, 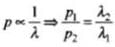
But particles have the same de-Broglie wavelength. (λ1 - λ2) = λ.
Then, 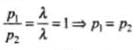
Thus, their momenta is same.
Also, 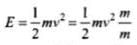
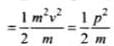
As p is constant, E ∝ 1/m
∴ 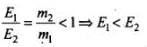
Important points:
Some important characteristics of Matter Waves:
1. Matter wave represents the probability of finding a particle in space.
2. Matter waves are not electromagnetic in nature.
3. De - Brogile or matter wave is independent of the charge on the material particle. It means, matter wave of de-Broglie wave is associated with every moving particle (whether charged or uncharged).
4. Practical observation of matter waves is possible only when the de-Broglie wavelength is of the order of the size of the particles.
5. Electron microscope works on the basis of de-Broglie waves.
6. The phase velocity of the matter waves can be greater than the speed of the light.
7. Matter waves can propagate in vacuum, hence they are not mechanical waves.
8. The number of de-Broglie waves associated with nth orbital electron is n.
9. Only those circular orbits around the nucleus are stable whose circumference is integral multiple of de-Broglie wavelength associated with the orbital electron.
Q.11. The de-Broglie wavelength of a photon is twice the de-Broglie wavelength of an electron. The speed of the electron is ve = c/100. Then
(a) 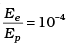
(b) 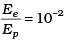
(c) 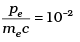
(d) 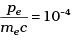
Ans. (b, c)
Solution.
de-Broglie wavelength λ = h/p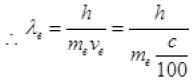
= 100h/mec ...(I)
Now K.E. = 1/2 mv2 ⇒ E = m2v2/2m ...(II)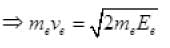
Now 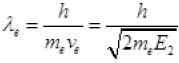 [from (II)]
[from (II)]
⇒ 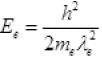 ...(III)
...(III)
For proton λp = 2λe [given]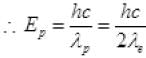
Now 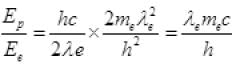 [from (III)]
[from (III)]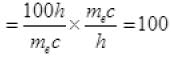
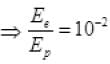 [verifies Ans. (b)]
[verifies Ans. (b)]
Now 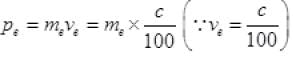
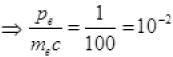 [verifies Ans. (c)]
[verifies Ans. (c)]
Q.12. Photons absorbed in matter are converted to heat. A source emitting n photon/sec of frequency ν is used to convert 1kg of ice at 0°C to water at 0°C. Then, the time T taken for the conversion
(a) Decreases with increasing n, with ν fixed.
(b) Decreases with n fixed, ν increasing
(c) Remains constant with n and ν changing such that nν = constant.
(d) Increases when the product nν increases.
Ans. (a, b, c)
Solution.
We know that energy spent to convert ice into water
Eabsorb = mass x latent heat
∴ E = mL = (1000 g) x (80 cal/g)
E = 80000 cal
Energy of photons used = nT x E = nT x hv [∵ E = hv]
So, nThv = mL ⇒ T = mL/nhv
∴ T ∝ 1/n. when v is constant.
Thus, time taken for conversion decreases with increasing v, with n kept constant.
⇒ T ∝ 1/nv
Thus, time taken for conversion decreases with increase in product of nv and T is constant, If nv is constant.
Q.13. A particle moves in a closed orbit around the origin, due to a force which is directed towards the origin. The de Broglie wavelength of the particle varies cyclically between two values λ1, λ2 with λ1 > λ2. Which of the following statement are true?
(a) The particle could be moving in a circular orbit with origin as centre
(b) The particle could be moving in an elliptic orbit with origin as its focus.
(c) When the de Broglie wave length is λ1, the particle is nearer the origin than when its value is λ2.
(d) When the de Broglie wavelength is λ2, the particle is nearer the origin than when its value is λ1.
Ans. (b, d)
Solution.
According to the question, here given that the de-Broglie wavelength of the particle can be varying cyclically between two values λ1 and λ2, it is possible if particle is moving in an elliptical orbit with origin as its one focus.
As shown in the figure given alongside,
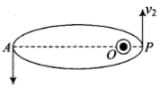
Let v1 and v2 be the speed of particle at A and B respectively and origin is at for us O. If λ1 and λ2 are the de-Broglie wavelengths associated with particle while moving at A and B respectively, then
λ1 = h/mv1
and λ2 = h/mv2
∴ 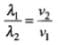
Since λ1 > λ2
∴ v2 > v1
By law of conservation of angular momentum, the particle moves faster when it is closer to focus.
From figure, we note that origin O is closed to P than A.
VERY SHORT ANSWER TYPE QUESTIONS
Q.14. A proton and an α-particle are accelerated, using the same potential difference. How are the deBroglie wavelengths λp and λa related to each other?
Ans. Hence de-Broglie wavelength: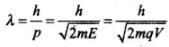
In this problem since both proton and α-particle are accelerated through same potential difference,
We know that, 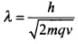
∴ 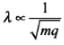
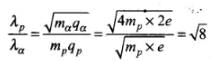
∴ λp = √8 λα
i.e., wavelength of proton is √8 times wavelength of α-particle.
Important point:
De-Broglie wavelength associated with the charged particles : The energy of a charged particle accelerated through potential difference V is E = 1/2 mv2 = qV
Hence de-Broglie wavelength 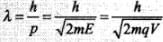
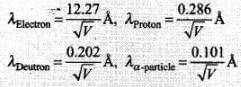
Q.15. (i) In the explanation of photo electric effect, we assume one photon of frequency ν collides with an electron and transfers its energy. This leads to the equation for the maximum energy Emax of the emitted electron as Emax = hν - Φ0
where Φ0 is the work function of the metal. If an electron absorbs 2 photons (each of frequency ν ) what will be the maximum energy for the emitted electron?
(ii) Why is this fact (two photon absorption) not taken into consideration in our discussion of the stopping potential?
Ans. (i) Here, 2 photons transfer its energy to one electron as E = hv
∴ Ee = Ep
Hve = 2hv
∴ ve = 2v
Maximum energy of emitted electron is
Emax = hve- Φ0 = h(2v) - Φ0 =2hv - Φ0
(ii) The probability of absorbing 2 photons by electron is very low due to their mass difference. So possibilities of such emission of electrons is negligible.
Q.16. There are materials which absorb photons of shorter wavelength and emit photons of longer wavelength. Can there be stable substances which absorb photons of larger wavelength and emit light of shorter wavelength.
Ans. In the first case, when the materials which absorb photons of shorter wavelength has the energy of the incident photon on the material is high and the energy of emitted photon is low when it has a longer wavelength or in short we can say that energy given out is less than the energy supplied.
But in second case, the energy of the incident photon is low for the substances which has to absorb photons of larger wavelength and energy of emitted photon is high to emit light of shorter wavelength. This means in this statement material has to supply the energy for the emission of photons.
But this is not possible for a stable substances.
Q.17. Do all the electrons that absorb a photon come out as photo-electrons?
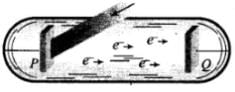
Ans. Photo-Electric Effect:
The photo-electric effect is the emission of electrons (called photo-electrons when light strikes a surface. To escape from the surface, the electron must absorb enough energy from the incident radiation to overcome the attraction of positive ions in the material of the surface.
The photoelectric effect is based on the principle of conservation of energy.
1. Two conducting electrodes, the anode (Q) and cathode (P) are enclosed in an evacuated glass tube as shown on next page.
2. The battery or other source of potential difference creates an electric field in the direction from anode to cathode.
3. Light of certain wavelength or frequency falling on the surface of cathode causes a current in the external circuit called photoelectric current.
4. As potential difference increases, photoelectric current also increases till saturation is reached.
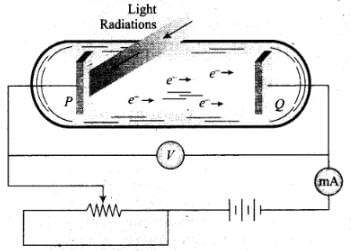
5. When polarity of battery is reversed (i.e., plate Q is at negative potential w.r.r. plate P) electrons start moving back towards the cathode.
6. At a particular negative potential of plate Q no electron will reach the plate Q and the current will become zero. This negative potential is called stopping potential denoted by V0. Maximum kinetic energy of photo electrons in terms of stopping potential will therefore be Kmax = (|V0|) eV
So we conclude that in photoelectric effect, we can observe that most electrons get scattered into the metal by absorbing a photon.
Therefore, all the electrons that absorb a photon doesn't come out as photoelectron. Only a few come out of metal whose energy becomes greater than the work function of metal.
Q.18. There are two sources of light, each emitting with a power of 100 W. One emits X-rays of wavelength 1 nm and the other visible light at 500 nm. Find the ratio of number of photons of X-rays to the photons of visible light of the given wavelength?
Ans. X-Rays:
1. X-rays were discovered by scientist Roentgen that is why they are also called Roentgen rays.
2. Roentgen discovered that when pressure inside a discharge tube is kept 10-3 mm of Hg and potential difference is kept 25 kV, then some unknown radiations (X-rays) are emitted by anode.
3. There are three essential requirements for the production of X-rays.
(i) A source of electron
(ii) An arrangement to accelerate the electrons
(iii) A target of suitable material of high atomic weight and high melting point on which these high speed electrons strike.
Here in this problem total energy will be constant.
Let us assume wavelength of X-rays is λ1 and the wavelength of visible light is λ2.
Given, P = 100 W
λ1 = 1 nm
and λ2 = 500 nm
Let n1 and n2 be the number of photons of X-rays and visible light emitted from the two sources per sec.
So, 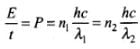
⇒ 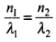
⇒ 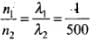
Important Point: The wavelength of characteristic X-ray doesn't depend on accelerating voltage. It depends on the atomic number (Z) of the target material. In characteristic X-ray spectrum λKα < λLα < λMα and vKα < vLα < λMα. Also λKα < λKβ < λKγ
SHORT ANSWER TYPE QUESTIONS
Q.19. Consider Fig. for photo-emission.
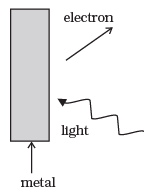
How would you reconcile with momentum-conservation? Note light (photons) have momentum in a different direction than the emitted electrons.
Ans. The momentum of incident photon is transferred to the metal ,during photo electric emission.
At microscopic level ,atoms of a metal absorb the photon and its momentum is transferred mainly to the nucleus and electrons.The excited electron is emitted. Therefore, the conservation of momentum is to be considered as the momentum of incident photon transferred to the nucleus and electron.
Q.20. Consider a metal exposed to light of wavelength 600 nm. The maximum energy of the electron doubles when light of wavelength 400 nm is used. Find the work function in eV.
Ans. Let the maximum energies of emitted electrons are K1 and K2 when 600 nm and 400 nm visible light are used according to question
k2 = 2K1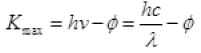
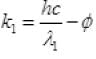
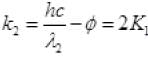
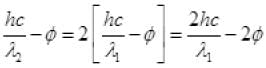
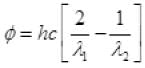 ( ∴ hc = 1240 ev nm)
( ∴ hc = 1240 ev nm)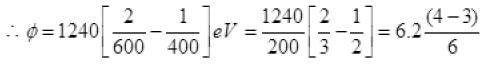
Work function φ = 6.2/6 = 1.03 eV.
Q.21. Assuming an electron is confined to a 1nm wide region , find the uncertainty in momentum using Heisenberg Uncertainty principle (Ref Eq 11.12 of NCERT Textbook). You can assume the uncertainty in position ∆x as 1nm. Assuming p = Δp, find the energy of the electron in electron volts.
Ans. In this problem, Δx = 1 nm = 10-9 m, we have to find Δp. As we know Δx. Δp = h
Therefore, 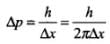
⇒ 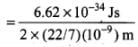
= 1.05 x 10-25 kg m/s
Energy, 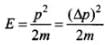 [∵ p = Δp]
[∵ p = Δp]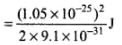
⇒ 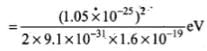
= 3.8 x 10-2 eV
Q.22. Two monochromatic beams A and B of equal intensity I, hit a screen. The number of photons hitting the screen by beam A is twice that by beam B. Then what inference can you make about their frequencies?
Ans. IA = IB [Given]
nAhvA = nBhvB
∵ nA = 2nB [Given]
∴ 2nB vA = nB vB
2vA = vB
So the frequency of source B is twice the frequency of source A.
Q.23. Two particles A and B of de Broglie wavelengths λ1 and λ2 combine to form a particle C. The process conserves momentum. Find the de Broglie wavelength of the particle C. (The motion is one dimensional).
Ans. By the law of conservation of momentum,
|pC| = |pA| + |pB|
Let us first take the case I when both pA and pB are positive, then 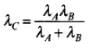
In second case when both pA and pB are negative, then 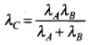
In case III when pA > 0, pB < 0 i.e., pA is negative and pB is positive.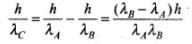
⇒ 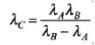
And in case IV when pA<0, pB>0, i.e., pA is negative and pB is positive.
∴ 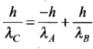
⇒ 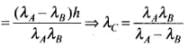
Q.24. A neutron beam of energy E scatters from atoms on a surface with a spacing d = 0.1nm. The first maximum of intensity in the reflected beam occurs at θ = 30°. What is the kinetic energy E of the beam in eV?
Ans. By Bragg’s law of diffraction, condition for nth maxima is
2d sinθ = nλ
n = 1 so λ = 2d sinθ [ θ = 30°(Given)]
= 2 × 0.1 × 10-9 sin 30° (∵ d = 0.1 nm)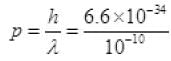 = 6.6 x 10-24 kg m/s
= 6.6 x 10-24 kg m/s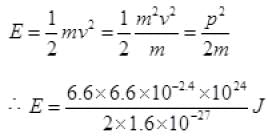
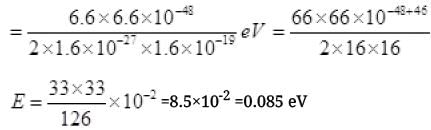
LONG ANSWER TYPE QUESTIONS
Q.25. Consider a thin target (10–2m square, 10–3m thickness) of sodium, which produces a photocurrent of 100µA when a light of intensity 100W/m2 (λ = 660nm) falls on it. Find the probability that a photoelectron is produced when a photons strikes a sodium atom. [Take density of Na = 0.97 kg/m3].
Ans. Area of square sheet = (A) = 10-2×10-2 = 10-4 m2
Thickness (d) = 10-3 m
Current (i) = 100 μA = 10-4 A
Intensity (I) = 100 W/m2
Mass of target (m) = Vol. × density
m = Area of sheet × thickness × density
= (10-4 × 10-3) × 0.97kg = 0.97 × 10-7kg
M = 0.97 ×10-4 gm
∴ No. of Na Atoms in target = 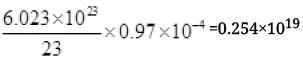
Number of Na atoms in Na target = 2.54 × 1018 Atoms
Total energy falling per second on target = nhv
Intensity × Area = n x h x = c/λ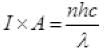
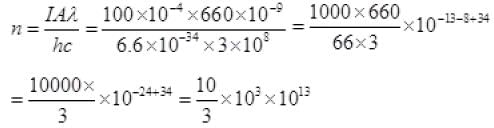
Number of photons (n) incident per second on Na-metal
∴ n = 3.3 × 1016
Let P is the probability of emission of photo-electrons per atom per photon.
Number of photo-electrons emitted per second
N = P·n. (No. of sodium atom)
N = P × 3.3 × 1016 × 2.54 × 1018 ...(I)
i = 100μA = 10-4 A = Ne
⇒ N = i/e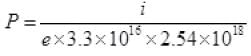
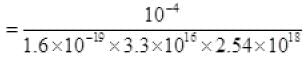
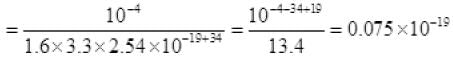
P = 7.5 × 10-21
Q.26. Consider an electron in front of metallic surface at a distance d (treated as an infinite plane surface). Assume the force of attraction by the plate is given as 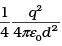
Calculate work in taking the charge to an infinite distance from the plate. Taking d = 0.1nm, find the work done in electron volts. [Such a force law is not valid for d < 0.1 nm].
Ans. Consider the figure in which an electron is displaced slowly by a distance x by the means of an external force which is,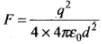
where, d = 0.1 nm = 10-10 m
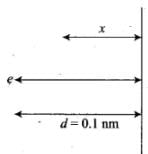
Let the electron be at distance x from metallic surface. Then, force of attraction on it is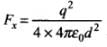
Work done by external agency in taking the electron from distance d to infinity is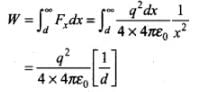
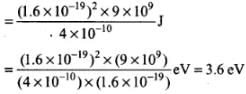
Q.27. A student performs an experiment on photoelectric effect, using two materials A and B. A plot of Vstop vs ν is given in Fig.
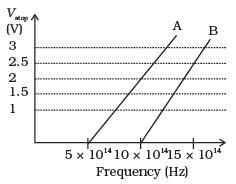
(i) Which material A or B has a higher work function?
(ii) Given the electric charge of an electron = 1.6 × 10–19 C, find the value of h obtained from the experiment for both A and B.
Comment on whether it is consistent with Einstein’s theory:
Ans. Threshold frequency (n0) : The minimum frequency of incident radiation, required to eject the electron from metal surface is defined as threshold frequency.
If incident frequency n < n0 ⇒ No photoelectron emission
For most metals the threshold frequency is in the ultraviolet (corresponding to wavelengths between 200 and 300 nm), but for potassium and cesium oxides it is in the visible spectrum (λ between 400 and 700 nm).
Here we are given threshold frequency of A
vOA = 5 x 1014 Hz and
For B, vOB = 10 x 1014 Hz
We know that
Work function, 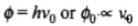
⇒ Ф0 ∝ v0
So, 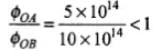
⇒ ФOA < ФOB
Thus, work function of B is higher than A.
(ii) For metal A, slope = h/e = 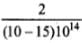
or 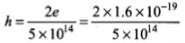
= 6.4 x 10-34 Js
For metal B, slope = h/e = 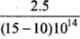
Or 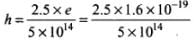
= 8 x 10-34 Js
Since, the vale of h from experiment for metals A and B is different. Hence, experiment is not consistent with theory.
Q.28. A particle A with a mass mA is moving with a velocity v and hits a particle B (mass mB) at rest (one dimensional motion). Find the change in the de-Broglic wavelength of the particle A. Treat the collision as elastic.
Ans. The laws of conservation of momentum and kinetic energy are obeyed because the collision is elastic.
By the law of conservation of momentum,
mAv + mB0 = mAv1 + mBv2
⇒ mA(v - v1) = mBv2
And the law of conservation of energy,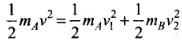 ...(i)
...(i)
⇒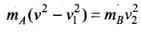
⇒ 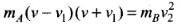 ...(ii)
...(ii)
Dividing Eq. (ii) by Eq. (i),
we get v + v1 = v2 or v = v2 - v1 ...(iii)
Solving Eqs. (i) and (iii), we get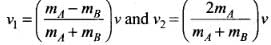
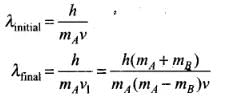
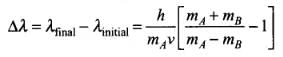
Q.29. Consider a 20 W bulb emitting light of wavelength 5000 Å and shining on a metal surface kept at a distance 2m. Assume that the metal surface has work function of 2 eV and that each atom on the metal surface can be treated as a circular disk of radius 1.5 Å.
(i) Estimate no. of photons emitted by the bulb per second. [Assume no other losses]
(ii) Will there be photoelectric emission?
(iii) How much time would be required by the atomic disk to receive energy equal to work function (2 eV)?
(iv) How many photons would atomic disk receive within time duration calculated in (iii) above?
(v) Can you explain how photoelectric effect was observed instantaneously?
[Hint: Time calculated in part (iii) is from classical consideration and you may further take the target of surface area say 1cm2 and estimate what would happen?]
Ans. (i) P = 20W, λ = 5000 Å = 5000 × 10-10 m, d = 2m, φ = 2 eV,
r = 1.5 Å = 1.5 × 10-10 m (Atomic radius)
Let number of photons emitted by bulb per second is n1 then Power P is
P = n1hv or P = 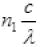
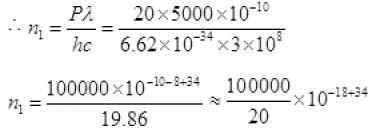
n1 = 5 × 1016 + 3 = 5 × 1019 no. of photons per sec.
Number of photons emitted by bulb per second
n1 = 5 × 1019
(ii) Energy of incident photon E = hv = hc/λ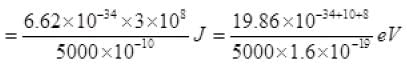
= 2.48 eV
Energy of photon = hv = hc/λ =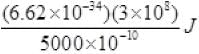
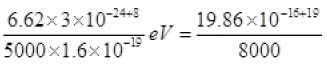
Energy of photon 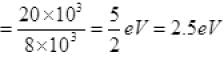
As the energy of an incident photon is more than 2 eV i.e., the work function of metal surface hence the photoelectric emission takes place.
(iii) Let ΔT time spent in getting the energy φ (work function of metal) Energy received by atomic disk in
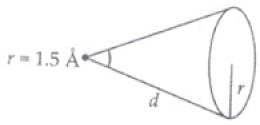
Δt time E = P × A· Δt
E = P × πr2·Δt
Energy transferred by bulb in full solid angle 4πd2 to atoms = 4πd2φ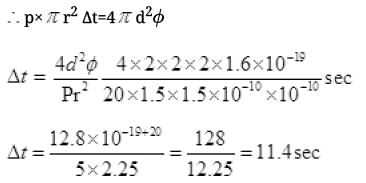
(iv) Number of photons received by one atomic dick in time Δt is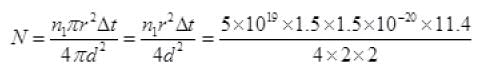
[n1 from part (i) and Δt from part (iii)]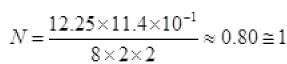 photon per atom
photon per atom
N = 1 photon per atom.
(v) Time of emission of electrons is 11.4 sec. So the photoelectric emission is not instantaneous in the problem. It takes about 11.4 sec.
In photoelectric emission there is a collision between incident photon and free electron and nucleus, which lasts last for very - very short interval of time (10-9 sec) hence we say photoelectric emission is instantaneous.
|
201 videos|410 docs|280 tests
|
FAQs on NCERT Exemplar: Dual Nature of Radiation and Matter - Physics for Airmen Group X - Airforce X Y / Indian Navy SSR
| 1. What is the dual nature of radiation and matter? |  |
| 2. What is the photoelectric effect? |  |
| 3. How does the photoelectric effect support the particle nature of light? |  |
| 4. What is the de Broglie wavelength? |  |
| 5. How is the wave-particle duality demonstrated by the electron diffraction experiment? |  |





















