NCERT Solutions for Class 9 Science Chapter 3 - Atoms and Molecules
| Table of contents |

|
| Page No. 27 |

|
| Page No. 30 |

|
| Page No. 34 |

|
| Page No. 35 |

|
| Page No. 36 |

|
Page No. 27
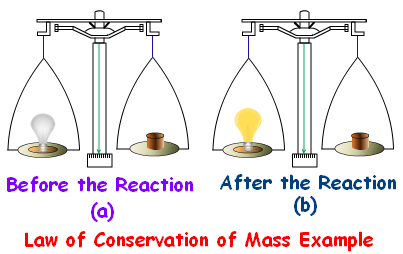
Q1. In a reaction, 5.3 g of sodium carbonate reacted with 6 g of ethanoic acid. The products were 2.2 g of carbon dioxide, 0.9 g water and 8.2 g of sodium ethanoate. Show that these observations are in agreement with the law of conservation of mass.
Sodium carbonate ethanoic acid → sodium ethanoate carbon dioxide water
Ans: 
Mass of reactants = 5.3 g + 6 g = 11.3 g
Mass of products = 2.2 g + 0.9 g + 8.2 g = 11.3 g
Mass of reactants = Mass of products
Therefore, the law of conservation of mass is proven.
Q2. Hydrogen and oxygen combine in the ratio of 1 : 8 by mass to form water. What mass of oxygen gas would be required to react completely with 3g of hydrogen gas?
Ans: Since hydrogen and oxygen combine in the ratio of 1:8 by mass, 3g of hydrogen gas will react completely with 24 g of oxygen gas.
Q3. Which postulate of Dalton's atomic theory is the result of the law of conservation of mass?
Ans: Dalton’s postulate that “atoms can neither be created nor destroyed,” is a result of the law of conservation of mass.
Q4. Which postulate of Dalton's atomic theory can explain the law of definite proportions?
Ans: Atoms combine in a fixed ratio to form compounds, which can explain the law of definite proportions.
Page No. 30
Q1. Define atomic mass unit.
Ans: It is defined as equal to 1/12th of the mass of 1 atom of C-12. It is called unified mass denoted by 'u’ these days.
Q2. Why is it not possible to see an atom with naked eyes?
Ans: The size of an atom is so small that it is not possible to see it with naked eyes. Also, the atom of an element does not exist independently.
Page No. 34
Q1. Write down the formulae of
(a) sodium oxide
(b) aluminium chloride
(c) sodium sulphide
(d) magnesium hydroxide
Ans:
(a) Formula of Sodium Oxide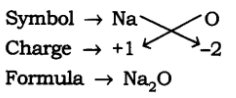
(b) Formula of Aluminium Chloride
(c) Formula of Sodium Sulphide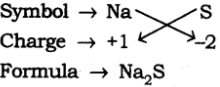
(d) Formula of Magnesium Hydroxide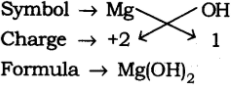
Q2. Write down the names of compounds represented by the following formulae:
(a) Al2(SO4)3
(b) CaCl2
(c) K2SO4
(d) KNO3
(e) CaCO3
Ans:
(a) Aluminium sulphate
(b) Calcium chloride
(c) Potassium sulphate
(d) Potassium nitrate
(e) Calcium carbonate
Q3. What is meant by the term chemical formula?
Ans: The chemical formula of a compound is a symbolic representation of its composition.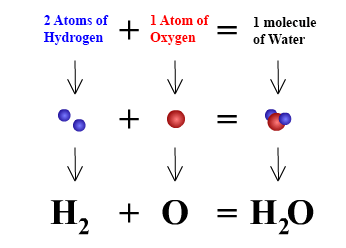 Chemical Formula of Water
Chemical Formula of Water
Q4. How many atoms are present in
(a) H2S molecule and
(b) PO43- ion?
Ans:
(i) H2S molecule has 2 atoms of hydrogen and 1 atom of sulphur hence 3 atoms in totality.
(ii) PO43- ion has 1 atom of phosphorus and 4 atoms of oxygen hence 5 atoms in totality.
Page No. 35
Q1. Calculate the molecular masses of H2, O2, Cl2, CO2, CH4, C2H6, C2H4, NH3, CH3OH.
Ans:
Molecular mass of H2 = 2 × Atomic mass of H
= 2 × 1
= 2 u
Molecular mass of O2 = 2 × Atomic mass of O
= 2 × 16
= 32 u
Molecular mass of Cl2 = 2 × Atomic mass of Cl
= 2 × 35.5
= 71 u
Molecular mass of CO2 = Atomic mass of C2 × Atomic mass of O
= 12+ (2+16) = (12 + 32)u
= 44 u
Molecular mass of CH4= Atomic mass of C4 × Atomic mass of H
= 12+ (4 x 1)u = (12 + 4)u
= 16 u
Molecular mass of C2H6 = 2× Atomic mass of C6× Atomic mass of H
= (2 x 12 + 6 x 1)u = (24 + 6)u
= 30 u
Molecular mass of C2H4 = 2 x Atomic mass of C4 × Atomic mass of H
= (2 x 12 + 4 x 1)u = (24 + 4)u
= 28 u
Molecular mass of NH3 = Atomic mass of N3 × Atomic mass of H
= (14 + 3 x 1)u = (14 + 3)u
= 17 u
Molecular mass of CH3OH = Atomic mass of C3 × Atomic mass of H Atomic mass of O Atomic mass of H
= (12 + 3 x 1 + 16 + 1)u = (12 + 3 + 17)u
= 32 u
Q2. Calculate the formula unit masses of ZnO, Na2O, K2CO3, given atomic masses of Zn = 65 u, Na = 23 u, K = 39 u, C = 12 u, and O = 16 u.
Ans:
(i) Formula unit mass of ZnO
= 65 + 16 = 81 u
(ii) Formula unit mass of Na2O
= 2 x 23 + 16 = 46 + 16 = 62 u
(iii) Formula unit mass of K2CO3
= 2 x 39 + 12 + 3 x 16
= 78 + 12 + 48 = 138 u
Page No. 36
Q1. A 0.24 g sample of a compound of oxygen and boron was found by analysis to contain 0.096 g of boron and 0.144 g of oxygen.
Calculate the percentage composition of the compound by weight.
Ans: Percentage of boron = (mass of boron / mass of the compound) x 100
= (0.096g / 0.24g) x 100
= 40%
Percentage of oxygen = 100 – percentage of boron
= 100 – 40
= 60%
Q2. When 3.0 g of carbon is burnt in 8.00 g oxygen, 11.00 g of carbon dioxide is produced. What mass of carbon dioxide will be formed when 3.00 g of carbon is burnt in 50.00 g of oxygen? Which law of chemical combination will govern your answer?
Ans: When 3.0 g of carbon is burnt in 8.00 g of oxygen, 11.00 g of carbon dioxide is produced.
Given that
3.0 g of carbon combines with 8.0 g of oxygen to give 11.0 of carbon dioxide.
Find out
We need to find out the mass of carbon dioxide that will be formed when 3.00 g of carbon is burnt in 50.00 g of oxygen.
Solution
First, let us write the reaction taking place here.
C + O2 → CO2
As per the given condition, when 3.0 g of carbon is burnt in 8.00 g of oxygen, 11.00 g of carbon dioxide is produced.
3g + 8g →11 g ( from the above reaction)
The total mass of reactants = mass of carbon + mass of oxygen
= 3g+8g
= 11g
The total mass of reactants = Total mass of products
Therefore, the law of conservation of mass is proved.
Then, it also depicts that carbon dioxide contains carbon and oxygen in a fixed ratio by mass, which is 3:8.
Thus, it further proves the law of constant proportions.
3 g of carbon must also combine with 8 g of oxygen only.
This means that (50−8) = 42g of oxygen will remain unreacted.
The remaining 42 g of oxygen will be left un-reactive. In this case, too, only 11 g of carbon dioxide will be formed.
The above answer is governed by the law of constant proportions.
Q3. What are polyatomic ions? Give examples.
Ans: Polyatomic ions are ions that contain more than one atom, but they behave as a single unit.
Example: CO32-, H2PO4–
Q4. Write the chemical formulae of the following.
(a) Magnesium chloride
(b) Calcium oxide
(c) Copper nitrate
(d) Aluminium chloride
(e) Calcium carbonate.
Ans: The following are the chemical formula of the above-mentioned list:
(a) Magnesium chloride – MgCl2
(b) Calcium oxide – CaO
(c) Copper nitrate – Cu(NO3)2
(d) Aluminium chloride – AlCl3
(e) Calcium carbonate – CaCO3
Q5. Give the names of the elements present in the following compounds.
(a) Quick lime
(b) Hydrogen bromide
(c) Baking powder
(d) Potassium sulphate.
Ans: The following are the names of the elements present in the following compounds:
(a) Quick lime – Calcium and oxygen (CaO)
(b) Hydrogen bromide – Hydrogen and bromine (HBr)
(c) Baking powder – Sodium, Carbon, Hydrogen, Oxygen (NaHCO3)
(d) Potassium sulphate – Sulphur, Oxygen, Potassium (K2SO4)
Q6. Calculate the molar mass of the following substances.
(a) Ethyne, C2H2
(b) Sulphur molecule, S8
(c) Phosphorus molecule, P4 (Atomic mass of phosphorus =31)
(d) Hydrochloric acid, HCl
(e) Nitric acid, HNO3
Ans: Listed below is the molar mass of the following substances:
(a) Molar mass of Ethyne C2H2= 2 x Mass of C+2 x Mass of H = (2×12)+(2×1)=24+2=26g
(b) Molar mass of Sulphur molecule S8 = 8 x Mass of S = 8 x 32 = 256g
(c) Molar mass of Phosphorus molecule, P4 = 4 x Mass of P = 4 x 31 = 124g
(d) Molar mass of Hydrochloric acid, HCl = Mass of H+ Mass of Cl = 1+35.5 = 36.5g
(e) Molar mass of Nitric acid, HNO3 =Mass of H+ Mass of Nitrogen + 3 x Mass of O = 1 + 14+
3×16 = 63g
|
84 videos|478 docs|60 tests
|
FAQs on NCERT Solutions for Class 9 Science Chapter 3 - Atoms and Molecules
| 1. What are atoms and molecules? |  |
| 2. How are atoms and molecules related to chemistry? |  |
| 3. What is the difference between an element and a compound? |  |
| 4. What is the mole concept and why is it important in chemistry? |  |
| 5. How do we balance chemical equations? |  |
















