Practice Questions with Solutions: Atoms and Molecules
Multiple Choice Questions
Q1: A sample of pure water, irrespective of its source contains 11.1% hydrogen and 88.9% oxygen. The data supports
(a) law of constant proportions
(b) law of conservation of mass
(c) law of reciprocal proportions
(d) law of multiple proportions
Ans: (a)
Explanation: Water obtained from any source contains hydrogen and oxygen in the same fixed proportion by mass (about 1 : 8 by mass of H : O). This constant mass ratio supports the law of constant proportions, which states that a chemical compound always contains the same elements in the same fixed proportion by mass.
Q2: Identify the correct statements.
1. In a compound such as water, the ratio of the mass of hydrogen to the mass of oxygen is always 8 : 1.
2. If 9 g of water is decomposed, 1 g of hydrogen and 8 g of oxygen are always obtained.
3. In ammonia, nitrogen and hydrogen are always present in the ratio 3 : 14 by mass.
4. Many compounds are composed of two or more elements and each such compound has the same elements in the same proportions.
(a) 1 and 3
(b) 1, 2 and 3
(c) 2 and 4
(d) All of these
Ans: (c)
Explanation: Statement 2 is correct because 9 g of water will yield 1 g H and 8 g O (mass ratio H : O = 1 : 8). Statement 4 is correct as it restates the law of definite proportions. Statement 1 is incorrect because it reverses the ratio (it should be 1 : 8, not 8 : 1). Statement 3 is incorrect because in ammonia (NH3) the mass ratio N : H is 14 : 3, not 3 : 14. Hence only statements 2 and 4 are correct.
(c)
In a compound such as water, the ratio of the mass of hydrogen to the mass of oxygen is always 1 : 8. In ammonia, nitrogen and hydrogen are always present in the ratio 14 : 3 by mass.
Q3: The atomic mass of calcium (Ca) is 40 g. The number of moles in 60 g of calcium are
(a) 0.5 mol
(b) 2.0 mol
(c) 1.5 mol
(d) 0.75 mol
Ans: (c)
Sol: Number of moles = mass / atomic (molar) mass = 60 g / 40 g mol-1 = 1.5 mol.
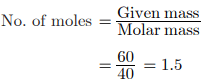
Q4: What mass of carbon-di-oxide (CO2) will contain 3.011 × 1023 molecules?
(a) 11.0 g
(b) 22.0 g
(c) 4.4 g
(d) 44.0 g
Ans: (b)
Sol: One mole (6.022 × 1023 molecules) of CO2 has mass 44 g. Number of moles = 3.011 × 1023 / 6.022 × 1023 = 0.5 mol. Mass = 0.5 × 44 g = 22.0 g.
Q5: The valency of nitrogen in ammonia (NH3) is
(a) 2
(b) 0
(c) 3
(d) 4
Ans: (c)
Explanation: In NH3, nitrogen forms three covalent bonds with three hydrogen atoms. Therefore, the valency of nitrogen in ammonia is 3.
Q6: The molecular formula P2O5 means that
(a) a molecule contains 2 atoms of P and 5 atoms of O
(b) the ratio of the mass of P to the mass of O in the molecule is 2 : 5
(c) there are twice as many P atoms in the molecule as there are O atoms
(d) the ratio of the mass of P to the mass of O in the molecule is 5 : 2.
Ans : (a)
Explanation: A molecular formula shows the actual number of atoms of each element in one molecule. P2O5 means each molecule contains 2 phosphorus atoms and 5 oxygen atoms.
Q7: Which of the following represents a polyatomic ion?
(a) Sulphide
(b) Chloride
(c) Sulphate
(d) Nitride
Ans : (c)
Explanation: Sulphate (SO42-) is a group of atoms bonded together carrying a net charge, so it is a polyatomic ion. Sulphide (S2-), chloride (Cl-) and nitride (N3-) are monoatomic ions.
 ion consists of group of atoms. Sulphate is a polyatomic ion.
ion consists of group of atoms. Sulphate is a polyatomic ion.Q8: How many elements are present in one formula unit of Al(OH)3?
(a) 3
(b) 4
(c) 5
(d) 6
Ans: (a)
Explanation: One formula unit of Al(OH)3 contains three different elements: aluminium (Al), oxygen (O) and hydrogen (H).
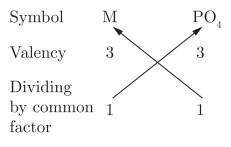
Q9: The formula of chloride of a metal M is MCl3, then the formula of the phosphate of metal M will be (a) MPO4
(b) M2PO4
(c) M3PO4
(d) M2(PO4)3
Ans: (a)
Explanation: MCl3 shows metal M has a charge of +3. The phosphate ion PO43- has charge -3. One M3+ will balance one PO43-, giving formula MPO4.
Q10: Which of the following is a triatomic molecule?
(a) Carbon-di-oxide
(b) Ammonia
(c) Helium
(d) Sugar
Ans: (a)
Explanation: A triatomic molecule contains three atoms. Carbon-di-oxide (CO2) has one carbon and two oxygen atoms (total 3 atoms). Ammonia (NH3) has four atoms (one N and three H), helium is a single atom, and sugar molecules are large molecules with many atoms.
Q11: All samples of carbon-di-oxide contain carbon and oxygen in the mass ratio 3 : 8. This is in agreement with the law of
(a) conservation of mass
(b) constant proportions
(c) multiple proportions
(d) gaseous volumes
Ans: (b)
Explanation: The law of constant proportions (law of definite proportions) states that a chemical compound always contains the same elements combined in the same fixed proportion by mass. The fixed 3 : 8 ratio of C : O in CO2 illustrates this law.
Q12: How many grams of H2SO4 are present in 0.25 mole of H2SO4 ?
(a) 2.45
(b) 24.5
(c) 0.245
(d) 0.25
Ans: (b)
Sol: Molar mass of H2SO4 = 2×1 + 32 + 4×16 = 98 g mol-1. Mass = number of moles × molar mass = 0.25 × 98 g = 24.5 g.
Q13: Select the incorrect match.
1. N2O4 -Dinitrogen tetroxide
2. HCl-Hydrogen chloride
3. CO-Carbon dioxide
4. PCl5 -Phosphorus trichloride
(a) 1 and 2
(b) 3 and 4
(c) 1 and 3
(d) 2 and 4
Ans: (b)
Explanation: Statements 3 and 4 are incorrect matches. CO is carbon monoxide, not carbon dioxide. PCl5 is phosphorus pentachloride, not phosphorus trichloride.
Q14: Atomicity of sulphur is
(a) 8
(b) 4
(c) 2
(d) 1
Ans : (a)
Explanation: Elemental sulphur commonly exists as S8 molecules, so its atomicity is 8.
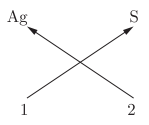
Q15: Valency of silver in Ag2S is
(a) 1
(b) 2
(c) 0
(d) 3
Ans: (a)
Explanation: In Ag2S, sulphur has valency 2 (S2-). Two silver atoms balance this charge, so each silver atom has valency 1 (Ag+).
Fill in the blanks.
Q16: In ionic compounds, the charge on each ion is used to determine the ......... of the compound.
Ans: chemical formula
In ionic compounds, the charge on each ion determines the chemical formula so that the total positive and negative charges balance and the compound is neutral. The numbers of ions are chosen to achieve this charge balance.
Q17: The Avogadro constant ......... is defined as the number of atoms in exactly ......... of carbon-12.
Ans: 6.022 × 1023, 12 g
The Avogadro constant is 6.022 × 1023 and it is defined as the number of atoms (or elementary entities) in exactly 12 g of carbon-12. This constant links the microscopic scale (atoms) to macroscopic amounts (grams).
Q18: The abbreviation used for lengthy names of elements are termed as their .........
Ans: symbol.
The short abbreviations for element names are called their symbols, for example H for hydrogen, O for oxygen and C for carbon.
Q19: Mole is link between the .......... and .........
Ans: mass of atoms & number of atoms.
The mole links the mass of atoms (or molecules) to the number of atoms (or molecules). One mole of a substance contains 6.022 × 1023 particles and has a mass equal to its molar mass in grams.
Q20: The valency of an ion is ......... to the charge on the ion.
Ans: equal.
The valency of an ion is equal to the magnitude of the charge on the ion (for example, Na+ has valency 1, Mg2+ has valency 2).
True/False
Q21: In a pure chemical compound, elements are always present in a definite proportion by mass.
Ans: True
Explanation: This statement is true. It states the law of definite proportions, which says a chemical compound always contains the same elements combined in a fixed proportion by mass.
Q22: Mass of 1 mole of a substance is called its formula mass.
Ans: False
Explanation: This statement is false. The mass of 1 mole of a substance is called its molar mass (expressed in g mol-1). Formula mass or formula unit mass is usually expressed in atomic mass units (amu) for ionic compounds or empirical formulas.
Q23: Water is an atom.
Ans: False
Explanation: This is false. Water (H2O) is a molecule made of two hydrogen atoms and one oxygen atom bonded together; it is not a single atom.
Q24: Formula mass of Na2O is 62 amu.
Ans: True
Explanation: This is true. Formula mass = 2×(atomic mass of Na) + (atomic mass of O) = 2×23 + 16 = 62 amu.
Q25: Those particles which have more or less electrons than the normal atoms are called ions.
Ans: True
Explanation: True. Particles that have gained or lost electrons compared with the neutral atom carry a net charge and are called ions (anions if negative, cations if positive).
Matching Questions
Direction: In the section, each question has two matching lists. Choices for the correct combination of elements from List-I and List-II are given as options (a), (b), (c) and (d) out of which one is correct.
Q26:
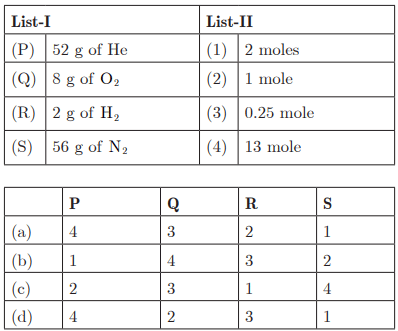
Ans: (a) P - 4, Q - 3, R - 2, S - 1
Q27:
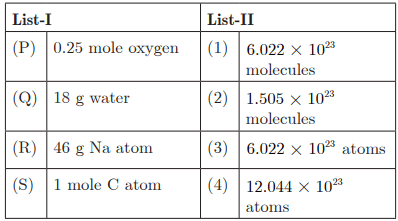
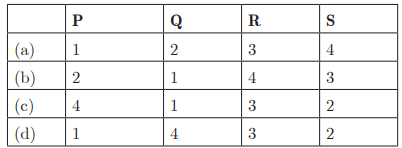
Q28:
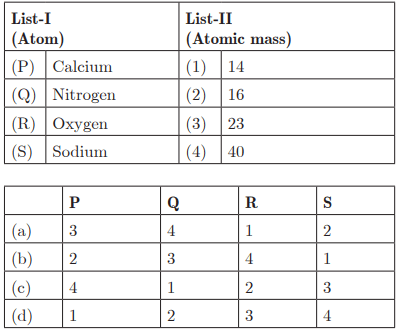
Ans: (c) P - 4, Q - 1, R - 2, S - 3
Q29:
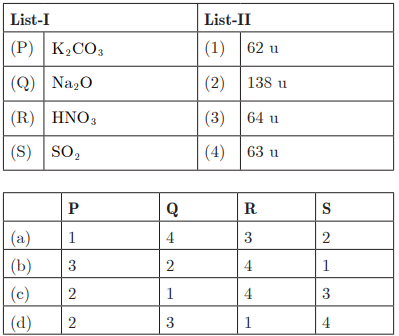
Ans : (c) P - 2, Q - 1, R - 4, S - 3
Q30:
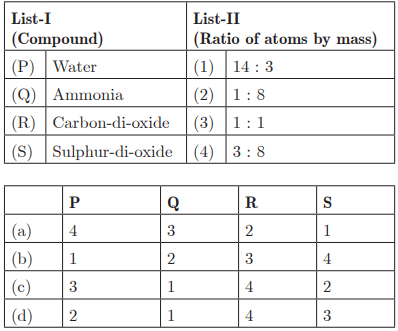
Ans : (d) P - 2, Q - 1, R - 4, S - 3
FAQs on Practice Questions with Solutions: Atoms and Molecules
| 1. What are atoms and how do they differ from molecules? |  |
| 2. What is the significance of the atomic number in relation to elements? |  |
| 3. How are isotopes defined and what is an example? |  |
| 4. What role do chemical bonds play in the formation of molecules? |  |
| 5. Can you explain the difference between a compound and a mixture? |  |





