Guide for Chemistry NEET Preparation | NEET Mock Test Series - Updated 2026 Pattern PDF Download

- NEET Chemistry is the highest scoring section out of these three sections. This subject can increase the selection chances if prepared well. The NEET Chemistry syllabus comprises an equal distribution of questions from both Class 11 and 12. Preparation for NEET Chemistry should, therefore, be started as early as Class 11.
- The NEET Chemistry section contains 45 questions mounting to a total of 180 marks. It is essential that candidates obtain a good score in NEET Chemistry section, in order to secure a good rank in the NEET UG examination. Candidates preparing for NEET Chemistry must have a good command over its various sections. The diversity of the syllabus requires candidates preparing for NEET Chemistry to be thorough with each topic.
- Chemistry is the most scoring subject in NEET! This is because the level of questions asked in NEET Chemistry is easy to moderate. The chances of mistakes and errors in this subject are minimum than of NEET Physics and Biology. With a little effort, you can extract maximum marks in this subject.
- Chemistry is divided into three sections i.e. Physical, Organic & Inorganic Chemistry. The questions asked from Inorganic are unambiguous. If you learned the chapters appropriately, then you can answer those questions with confidence.
- If you want to score well in Physical & Organic Chemistry for NEET then start practicing on a regular basis for these two sections. Regular and diligent practice is required in Physical and Organic Chemistry. Some students have been scoring full marks in Chemistry in the previous years and majority of the students have answered at least 35 correct answers out of 45. All this is possible if you structure your study plan properly.
Here are some Preparation tips for studying Chemistry for NEET 2022:
1. Analyse the Syllabus and Plan accordingly
The initial step in the process of preparation for NEET Chemistry is to analyse the entire syllabus for chemistry and plan your schedule for studying every topic accordingly. Take a good read of the syllabus before starting your preparation.
2. Keep it Simple
- The next thing to keep in mind for NEET is to keep in simple and do those simple things perfectly and as fast as you can. NEET questions are not the toughest in nature and neither are they designed to choose the one with the most IQ score.
- NEET demands a variety of skills and applications. For chemistry, it is absolutely important to first go through the past years question papers and notice the pattern of questions.
3. Important Topics should be Highlighted
Candidates preparing for NEET Chemistry must have proper knowledge about the important topics in order to have a head start. The NEET Chemistry syllabus is vast, and thus, highlighting the topics that carry more weightage in the examination will help the candidates to prepare accordingly. Based on the analysis of previous year question papers, the important topics of NEET Chemistry can be outlined.
Comprehensive Courses: Chemistry Class 11 & Chemistry Class 12
4. Make a Good Study Plan
- Time management and its distribution is the most crucial step in the preparation for chemistry and for that, one needs to prepare a proper schedule wherein every topic is allotted a particular amount of time. This will help you concentrate better on each topic and make time for every topic and chapter.
- Try not to study difficult topics one after another. This will result in mixing up of concepts in your head and may create difficulty in recalling the concepts later. Divide your time for all the major topics of all the subjects properly. Manage your timetable according to your school studies.
5. Do Regular Practice
- Only reading the formulas in physical chemistry or the reaction mechanisms in organic chemistry will leave you stranded in a test, without actual practice of the questions. After learning a formula in physical or a mechanism in organic chemistry, try to practice a lot of problems on those concepts so that you get the application right.
- It is almost impossible to apply any concept and law in the heat of the exam without having the practice of that type of question before.Learn your formulas thoroughly. Do not leave anything out. With regular revision, formulas should soon be at the top of your head.
- Aspirants while preparing for NEET Chemistry should develop the practice of writing everything down. This goes a long way during the time of revision and also helps in remembering the details.
- You must prepare topics and simultaneously solve MCQs from different comprehensive books. Practice is the only master key to crack the NEET exam.
Here are some really good mocks for you to practice
6. Study Inorganic Chemistry Regularly
- It is a major misconception that inorganic chemistry is all about mugging up and it must be done only at the last moment before the examination. Well, this is not applicable to many.
- You must try to learn them by heart, little at a time, throughout the year. It will become instilled in you, thus making it difficult for you to forget the exam anxiety even. It will also help you focus on other things more evenly before exams.
7. Attempt Mock and Previous Years Question Papers
- With NTA introducing online NEET 2022 exams, it is necessary for candidates to solve mock tests online so as to be able to manage the allotted time and get familiar with the real day exam scenario.
- By taking as many tests as you can before the final exam, you will gain confidence and have a good grasp over various important formulas and topics.
- You must attempt online test series for at least one month. This will greatly help you in evaluating your preparation level and will help you identify your weak areas.
EduRev Course: NEET Mock Tests Series
8. Revise Regularly and Effectively
- You must revise and recall everything that you study on a regular basis. This will help you remember all the concepts for a longer period.
- The more you learn the more you tend to forget. Revision is the only key to help prevent this aspect. Schedule revisions after each topic has been dealt with. Aspirants in Class 12 should continuously keep revising the syllabus of Class 11 to ensure that every topic is still fresh in mind. The more you manage to revise in the last week, the number of things will be fresh in your mind and you will be able to answer more easily.
NEET Chemistry Syllabus
- NEET Chemistry comprises three major sections of organic, inorganic and Physical Chemistry. Candidates dwelling on the prospect of how to prepare for NEET Chemistry should ideally start with a proper analysis of all the three sections.
- These three sections have been divided between the curriculum of Class 11 and 12. To be familiar with the topics, a detailed syllabus of NEET Chemistry has been provided below for reference:
NEET 2022 Chemistry Topicwise Syllabus
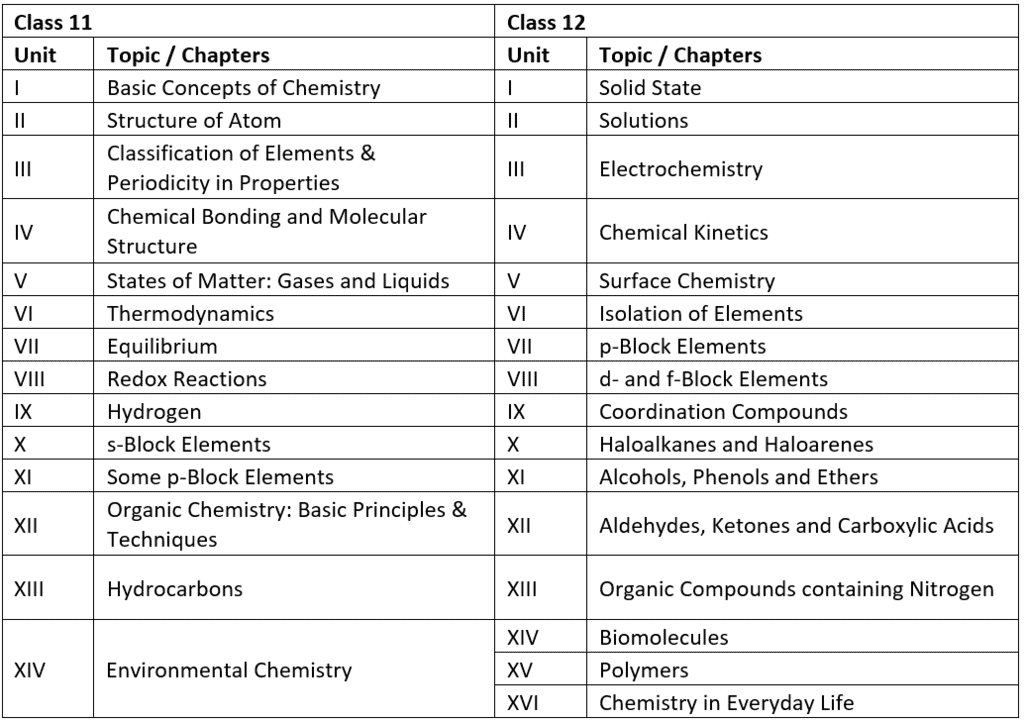
Important Topics of NEET 2022 Chemistry
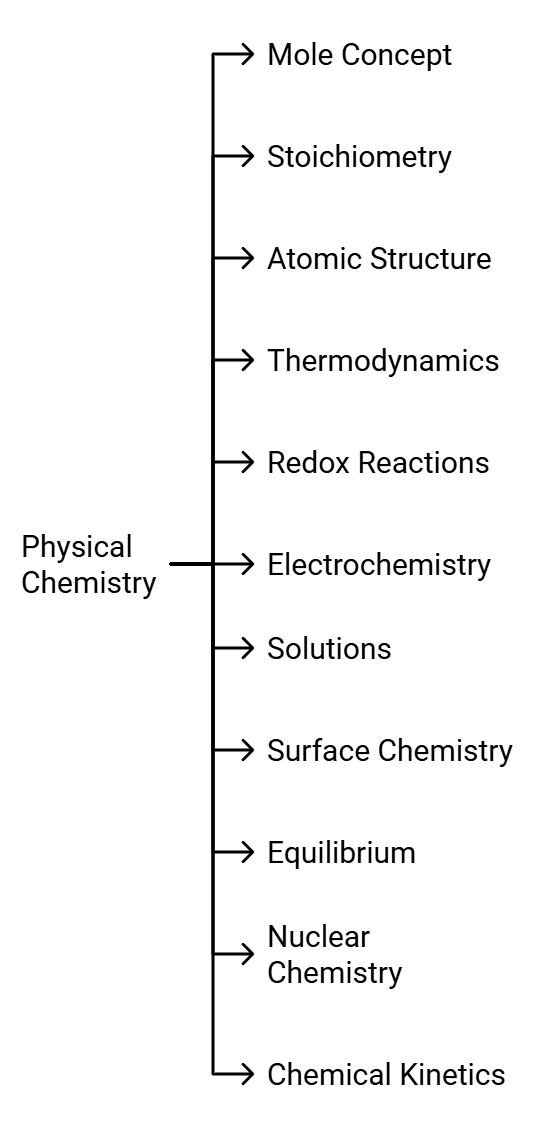
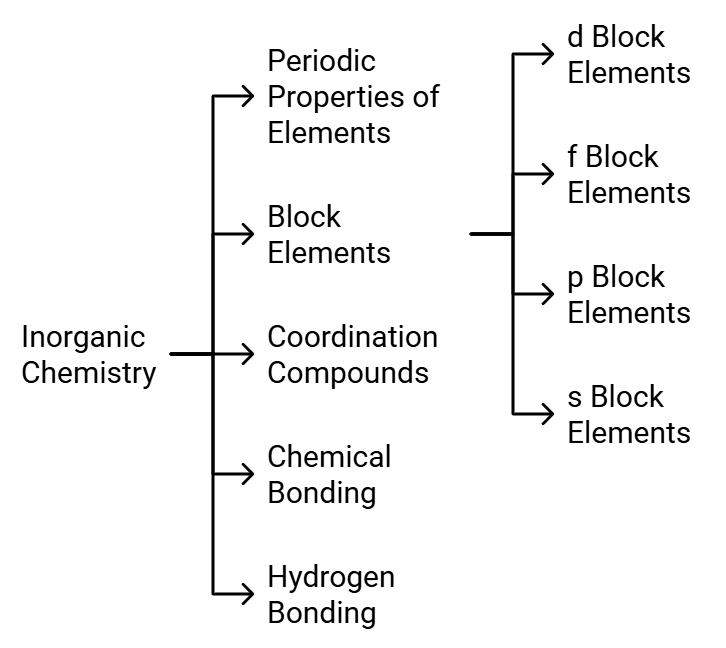
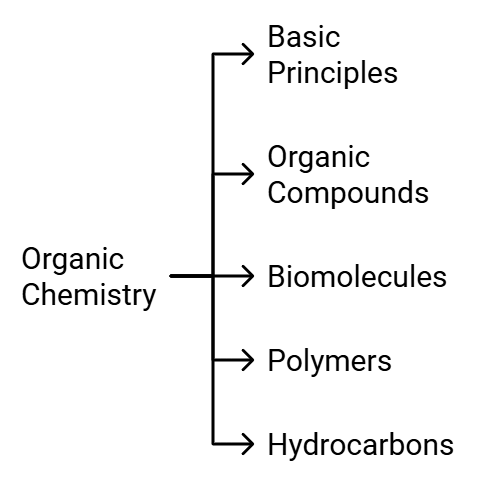
NEET 2022 Chemistry – Important Topics with their Weightage
Many aspirants are not able to figure out the important topics of Chemistry from the examination point of view. As per the analysis of previous years’ NEET Chemistry Questions.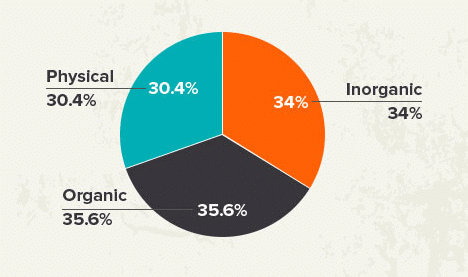
Here we are highlighting the section wise important topics with their weightage in the table below. 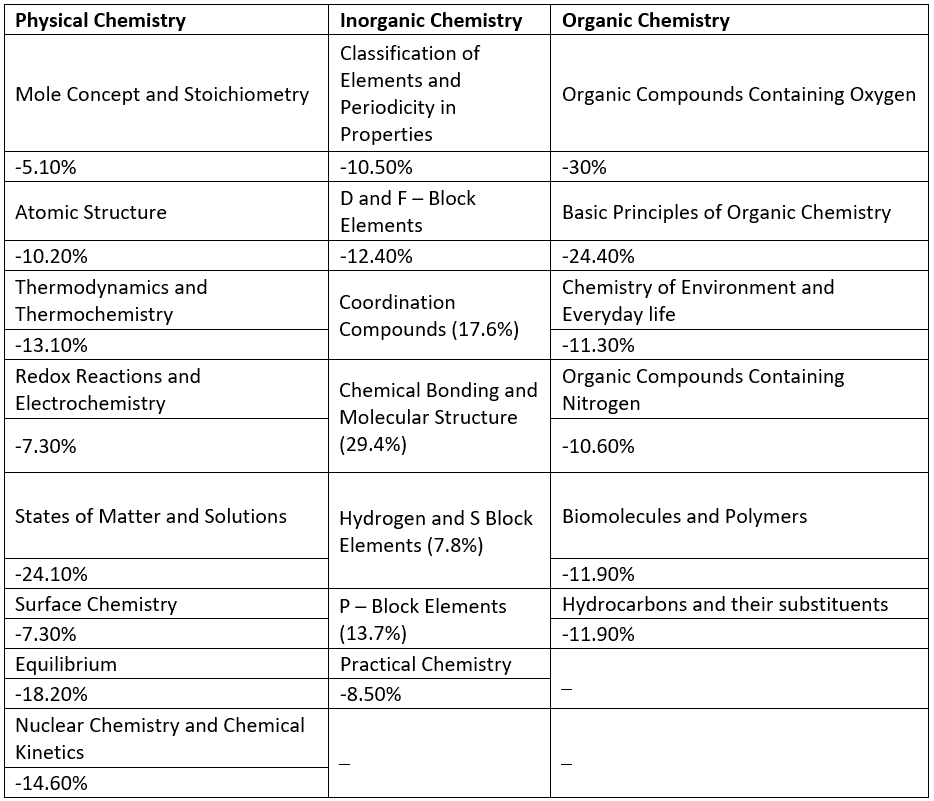
Chemistry Books for NEET
Having a precise study material is necessary for perfect preparation. Top-notch NEET Chemistry Books are the keys to open the gateway for MBBS & BDS Colleges.
Here are some of the best books for NEET Chemistry Preparation: 
Three things to keep in mind for the NEET exam
- Prepare a Strategy:
Make sure you are answering each question within the ideal time. Identify your weak concepts and keep practicing them to improve your score. - Time Management:
You have 180 minutes to answer 180 questions, but not necessarily 1 minute per question because each question carries a different difficulty level. So, managing tour time accordingly and don't spend too much time on one question. Move on if you are unable to find the answer to one. - Selection of Questions:
Each question carries 4 marks and 1 negative marking. Keeping this in mind, question selection is crucial. Attempt your strongest concepts first and leave the ones that will take a longer time to complete for last.
Similarly, Prepare for Physics and Chemistry for NEET with tips and tricks provided here to guide you:
|
1 videos|19 docs|71 tests
|
FAQs on Guide for Chemistry NEET Preparation - NEET Mock Test Series - Updated 2026 Pattern
| 1. What is the NEET Chemistry syllabus for Class 11 and Class 12? |  |
| 2. Which are the best books for NEET Chemistry preparation? |  |
| 3. How can I effectively prepare for the NEET Chemistry exam? |  |
| 4. What is the importance of NCERT books in NEET Chemistry preparation? |  |
| 5. Are there any online resources available for NEET Chemistry preparation? |  |






















