NEET Previous Year Questions (2014-2025): The Solid State (Old NCERT) | Physical Chemistry for NEET PDF Download
Q.1. A compound is formed by two elements A and B. The element B forms cubic close packed structure and atoms of A occupy 1/3 of tetrahedral voids. If the formula of the compound is AxBy, then the value of x + y is in option (2023)
A: 5
B: 4
C: 3
D: 2
Ans: A
Number of atoms of element B is N
So, the number of atoms of element A is 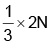
∴ The formula of the compound is 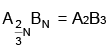
So, x = 2
y = 3
∴ x + y = 5
Q.2. What fraction of one edge centred octahedral void lies in one unit cell of fcc? (2023)
A: 1/2
B: 1/3
C: 1/4
D: 1/12
Ans: C
The total number of octahedral voids in FCC are four
Octahedral voids in FCC = Edge centres + Body centre
The contribution of edge centre = 1/4
∴ Fraction of one edge centred octahedral void in one unit cell of FCC = 1/4
Q.3. Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R). (2022)
Assertion (A): In a particular point defect, an ionic solid is electrically neutral, even if few of its cations are missing from its unit cells.
Reason (R): In an ionic solid, Frenkel defect arises due to dislocation of cation from its lattice site to interstitial site, maintaining overall electrical neutrality.
In the light of the above statements, choose the most appropriate answer from the options given below:
A: (A) is correct but (R) is not correct
B: (A) is not correct but (R) is correct
C: Both (A) and (R) are correct and (R) is the correct explanation of (A)
D: Both (A) and (R) are correct but (R) is not the correct explanation (A)
Ans: D
In particular point defect, ionic solid remains electrically neutral.
Q.4. Copper crystallises in fcc unit cell with cell edge length of 3.608 × 10–8 cm. The density of copper is 8.92 g cm–3. Calculate the atomic mass of copper. (2022)
A: 60 u
B: 65 u
C: 63.1 u
D: 31.55 u
Ans: C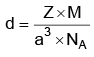
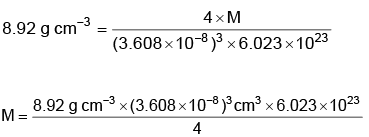
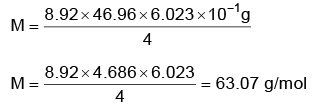
Atomic mass of Cu = 63.07 u
≈ 63.1 u
Q.5. Right option for the number of tetrahedral and octahedral voids in hexagonal primitive unit cell are: (2021)
A: 2, 1
B: 12, 6
C: 8, 4
D: 6, 12
Ans: B
No. of atoms in Hexagonal primitive unit cell = 6
No. of Tetrahedral voids = 2 × No. of atoms per unit cell
= 2 × 6 = 12
No. of Octahedral voids = No. of atoms per unit cell = 6
Q.6. The correct option for the number of body centred unit cells in all 14 types of Bravais lattice unit cells is : (2021)
A: 2
B: 3
C: 7
D: 5
Ans: B
The number of Body centred unit cells in all 14 types of Bravais lattice unit cells is 3.
Q.7. An element has a body centered cubic (bcc) structure with a cell edge of 288 pm. The atomic radius is: (2020)
A: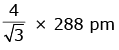
B: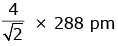
C: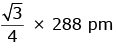
D: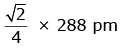
Ans: C
For bcc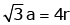
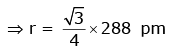
Q.8. A compound is formed by cation C and anion A. The anions form hexagonal close packed (hcp) lattice and the cations occupy 75% of octahedral voids. The formula of the compound is : (2019)
A: C2A3
B: C3A2
C: C3A4
D: C4A3
Ans: C
Anions(A) are in hcp, so number of anions (A) = 6
Cations(C) are in 75% O.V., so number of cations (C)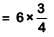
= 18/4
= 9/2
So formula of compound will be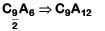
C9A12 ⇒ C3A4
Q.9. Iron exhibits bcc structure at room temperature. Above 900°C, it transforms to fcc structure. The ratio of density of iron at room temperature to that at 900°C (assuming molar mass and atomic radii of iron remains constant with temperature) is (2018)
A: 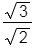
B: 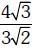
C: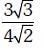
D: 1/2
Ans: C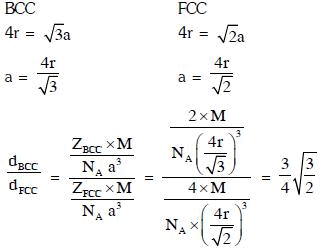
Q.10. Which is the incorrect statement? (2017)
A: Density decreases in case of crystals with Schottky's defect
B: NaCl(s) is insulator, silicon is semi conductor,silver is conductor, quartz is piezo electric crystal
C: Frenkel defect is favoured in those ionic compounds in which sizes of cation and an ions are almost equal
D: FeO0.98 has non stoichiometric metal deficiency defect
Ans: C
In frenkel defect the radius of cation must be very less than anion.
Q.11. Lithium has bcc structure. Its density is 530 kg m-3 and its atomic mass is 6.94 g mol-1. Calculate the edge length of a unit cell of Lithium metal. (NA = 6.02 x 1023 mol-1) (2016)
A: 264 pm
B: 154 pm
C: 352 pm
D: 527 pm
Ans: C
Given, Li has a bcc structure
Density (ρ) =530 kg-m-3
Atomic mass (M) = 6.94 g mol-1
Avogadro's number of atoms per unit cell in bcc (Z) = 2.
∴ We have the formula for density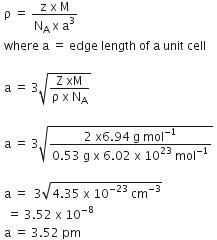
Q.12. The ionic radii of A+ and B- ions are 0.98 x 10-10 m and 1.81 x10-10 m. The coordination number of each ions in AB is (2016)
A: 2
B: 6
C: 4
D: 8
Ans: B
Given, ionic radius of cation (A+) =0.98 x 10-10 m
Ionic radius of anion (B-)=1.81 x 10-10 m
Now we have,
If radius ratio range is in between 0.441-0.732, ion would have an octahedral structure. Therefore, the coordination number of each ion in AB is 6.
Q.13. A given metal crystallizes out with a cubic structure having edge length of 361 pm. If there are four metal atoms in one unit cell, what is the radius of one atom? (2015)
A: 108 pm
B: 40 pm
C: 127 pm
D: 80 pm
Ans: C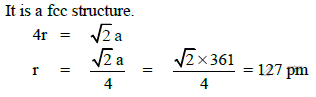
Q.14. If 'a' is the length of the side of a cube, the distance between the body centered atom and one corner atom in the cube will be : (2014)
A: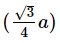
B: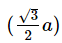
C: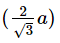
D: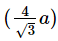
Ans: B
The distance between the body centered atom and corner atom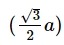
|
117 videos|226 docs|237 tests
|

















