NCERT Exemplar: The d & f-Block Elements
MULTIPLE CHOICE QUESTIONS (TYPE - I)
Q.1. Electronic configuration of a transition element X in +3 oxidation state is [Ar]3d5. What is its atomic number?
(i) 25
(ii) 26
(iii) 27
(iv) 24
Ans: (ii)
Explanation: Electronic configuration of X3+ is [Ar] 3d5. The [Ar] core accounts for 18 electrons. The 3d5 indicates five electrons in the d-shell and the +3 charge means three electrons have been removed from the neutral atom. Therefore, atomic number of X = 18 (Ar) + 5 (d-electrons) + 3 (electrons removed) = 26. Hence option (ii) is correct.
Q.2. The electronic configuration of Cu(II) is 3d9 whereas that of Cu(I) is 3d10. Which of the following is correct?
(i) Cu(II) is more stable
(ii) Cu(II) is less stable
(iii) Cu(I) and Cu(II) are equally stable
(iv) Stability of Cu(I) and Cu(II) depends on nature of copper salts
Ans: (i)
Explanation: Although Cu(I) has a stable 3d10 electronic configuration, in aqueous solution Cu+ tends to undergo disproportionation: 2Cu+ → Cu2+ + Cu(s). This shows that Cu(II) is the more stable oxidation state in many common conditions (particularly in aqueous media). Factors such as hydration energy and tendency to undergo redox processes make Cu(II) relatively more stable than Cu(I) in solution; hence option (i) is correct.
Q.3. Metallic radii of some transition elements are given below. Which of these elements will have highest density?
| Element | Fe | Co | Ni | Cu |
| Metallic radii/pm | 126 | 125 | 125 | 128 |
(i) Fe
(ii) Ni
(iii) Co
(iv) Cu
Ans: (iv)
Explanation: Density depends on mass per unit volume. Across a period, metallic radii generally decrease while atomic mass increases. A decrease in radius together with a relatively higher atomic mass increases density. Among the given elements, copper lies to the right and has the largest atomic mass with comparable metallic radius. The density of copper is about 8.96 g cm-3, which is the highest among the four given elements, so option (iv) is correct.
Q.4. Generally transition elements form coloured salts due to the presence of unpaired electrons. Which of the following compounds will be coloured in solid state?
(i) Ag2SO4
(ii) CuF2
(iii) ZnF2
(iv) Cu2Cl2
Ans: (ii)
Explanation: Colour in transition-metal compounds arises from d-d transitions or charge-transfer transitions involving unpaired d-electrons. In CuF2, copper is in the +2 oxidation state (Cu2+), which has one unpaired d-electron (3d9), so CuF2 is coloured. Ag2SO4 and ZnF2 contain d10 or closed-shell ions and are usually colourless; Cu2Cl2 contains Cu(I) (d10) and is typically white or colourless. Hence option (ii) is correct.
Q.5. On addition of small amount of KMnO4 to concentrated H2SO4, a green oily compound is obtained which is highly explosive in nature. Identify the compound from the following.
(i) Mn2O7
(ii) MnO2
(iii) MnSO4
(iv) Mn2O3
Ans: (i)
Explanation: Permanganate (MnO4-) reacts with concentrated H2SO4 to give the anhydride Mn2O7, a dark green oily compound that is a powerful and dangerous oxidiser and is explosively reactive. The reaction is:
2KMnO4 + 2H2SO4 (conc.) → Mn2O7 + 2KHSO4 + H2O.
This identifies Mn2O7 as the product, so option (i) is correct.
Q.6. The magnetic nature of elements depends on the presence of unpaired electrons. Identify the configuration of transition element, which shows highest magnetic moment.
(i) 3d7
(ii) 3d5
(iii) 3d8
(iv) 3d2
Ans: (ii)
Explanation: The magnetic moment (spin-only) increases with the number of unpaired electrons. The 3d5 configuration has five unpaired electrons (one in each d orbital), which is the maximum possible for first-row d-orbitals. Using the spin-only formula
μ = √[n(n+2)] BM,
where n is the number of unpaired electrons, for n = 5 we get
μ ≈ √[5×7] = √35 ≈ 5.92 B.M. Thus 3d5 shows the highest magnetic moment among the options given.
Q.7. Which of the following oxidation state is common for all lanthanoids?
(i) +2
(ii) +3
(iii) +4
(iv) +5
Ans: (ii)
Explanation: Lanthanoids show common oxidation state of +3. Some of the lanthanoids also show + 2 and +4 stable oxidation state along with +3 oxidation state. These are shown by those elements which by losing 2 or 4 electrons acquire a stable configuration of f0, f7 or f14,e.g.,
Eu2+ is [Xe] 4f7, Yb2+ is [Xe]4f14, Ce4+ is [Xe]4f° and Tb4f is [Xe]4f7
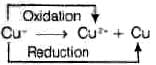
Q.8. Which of the following reactions are disproportionation reactions?
(a) Cu+ → Cu2+ + Cu
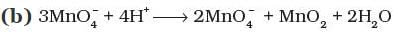
(c) 2KMnO4 → K2MnO4 + MnO2 + O2
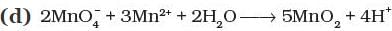
(i) a
(ii) a, b, c
(iii) b, c, d
(iv) a, d
Ans: (i)
Explanation: A disproportionation reaction is one in which the same element is simultaneously oxidised and reduced. Reaction (a) shows Cu+ being oxidised to Cu2+ and reduced to Cu(0) at the same time, which is classic disproportionation. The other reactions (b), (c), (d) must be examined individually, but here only (a) fits the definition as listed; hence option (i) is correct.
Q.9. When KMnO4 solution is added to oxalic acid solution, the decolourisation is slow in the beginning but becomes instantaneous after some time because
(i) CO2 is formed as the product.
(ii) Reaction is exothermic.
(iii) 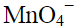 catalyses the reaction.
catalyses the reaction.
(iv) Mn2+ acts as autocatalyst.
Ans: (iv)
Explanation: The Mn2+ produced in the early stages of the reaction acts as an autocatalyst and accelerates the rate of reduction of MnO4- by oxalic acid. Initially, when Mn2+ concentration is low, the reaction is slow; as Mn2+ builds up it catalyses the reaction and the decolourisation becomes rapid. The overall redox process involves oxidation of oxalic acid to CO2 and reduction of MnO4- to Mn2+.
Reduction half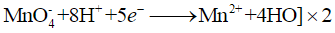
Oxidation half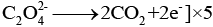
Overall equation
End point of this reaction Colourless to light pink.
Q.10. There are 14 elements in actinoid series. Which of the following elements does not belong to this series?
(i) U
(ii) Np
(iii) Tm
(iv) Fm
Ans: (iii)
Explanation: The actinoid (actinide) series comprises elements with atomic numbers 90 to 103. Thulium (Tm) has atomic number 69 and belongs to the lanthanoid (4f) series, not the actinoids. Uranium (U), Neptunium (Np) and Fermium (Fm) are actinoids. Hence option (iii) is correct.
Q.11. KMnO4 acts as an oxidising agent in acidic medium. The number of moles of KMnO4 that will be needed to react with one mole of sulphide ions in acidic solution is
(i) 2/5
(ii) 3/5
(iii) 4/5
(iv) 1/5
Ans: (i)
Explanation: In acidic medium the net oxidising oxygen equivalent produced by KMnO4 comes from the reaction:
2KMnO4 + 3H2SO4 → K2SO4 + 2MnSO4 + 3H2O + 5[O].
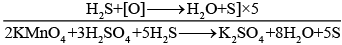
Here 2 moles of KMnO4 provide 5 atoms of nascent oxygen [O], each capable of oxidising one S2- to higher oxidation states. Therefore 5 moles of S2- react with 2 moles of KMnO4, so 1 mole of S2- requires 2/5 mole of KMnO4. Hence option (i) is correct.
Q.12. Which of the following is amphoteric oxide?
Mn2O7, CrO3, Cr2O3, CrO, V2O5, V2O4
(i) V2O5, Cr2O3
(ii) Mn2O7, CrO3
(iii) CrO, V2O5
(iv) V2O5, V2O4
Ans: (i)
Explanation: Amphoteric oxides react both with acids and bases. Cr2O3 and V2O5 exhibit amphoteric behaviour - they can dissolve in acids as well as in alkalis to form corresponding anions or complex salts. Other oxides listed are typically acidic (Mn2O7, CrO3) or have different behaviour.
Q.13. Gadolinium belongs to 4f series. It's atomic number is 64. Which of the following is the correct electronic configuration of gadolinium?
(i) [Xe] 4f75d16s2
(ii) [Xe] 4f65d26s2
(iii) [Xe] 4f86d2
(iv) [Xe] 4f95s1
Ans: (i)
Explanation: Gadolinium (Z = 64) has electronic configuration [Xe]4f75d16s2. The half-filled 4f7 subshell provides extra stability, and one electron occupies the 5d orbital in the ground-state configuration. Hence option (i) is correct.
Q.14. Interstitial compounds are formed when small atoms are trapped inside the crystal lattice of metals. Which of the following is not the characteristic property of interstitial compounds?
(i) They have high melting points in comparison to pure metals.
(ii) They are very hard.
(iii) They retain metallic conductivity.
(iv) They are chemically very reactive.
Ans: (iv)
Explanation: Some of the important characteristics of interstitial compounds are as follows:
(i) They are very hard and rigid.
(ii) They have high melting point which are higher than those of the pure metals.
(iii) They show conductivity like that of the pure metal.
(iv) They acquire chemical inertness.
Q.15. The magnetic moment is associated with its spin angular momentum and orbital angular momentum. Spin only magnetic moment value of Cr3+ ion is ___________.
(i) 2.87 B.M.
(ii) 3.87 B.M.
(iii) 3.47 B.M.
(iv) 3.57 B.M.
Ans: (ii)
Explanation: Cr3+ has electronic configuration 3d3, so number of unpaired electrons n = 3. Using the spin-only formula μ = √[n(n+2)] BM, we get μ = √[3×5] = √15 ≈ 3.87 B.M. Hence option (ii) is correct.
Q.16. KMnO4 acts as an oxidising agent in alkaline medium. When alkaline KMnO4 is treated with KI, iodide ion is oxidised to _________.
(i) I2
(ii) IO-
(iii)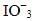
(iv)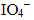
Ans: (iii)
Explanation: In alkaline medium permanganate oxidises iodide to iodate (IO3-) or higher oxyanions depending on conditions. The given reaction leads to formation of KIO3 as the oxidised iodine species. For example:
2KMnO4 + H2O + KI → 2MnO4- + 2KOH + KIO3 (showing iodide oxidation to iodate under alkaline conditions).

Q.17. Which of the following statements is not correct?
(i) Copper liberates hydrogen from acids.
(ii) In its higher oxidation states, manganese forms stable compounds with oxygen and fluorine.
(iii) Mn3+ and Co3+ are oxidising agents in aqueous solution.
(iv) Ti2+ and Cr2+ are reducing agents in aqueous solution.
Ans: (i)
Explanation: Copper lies below hydrogen in the electrochemical series (its standard reduction potential is positive), so it does not displace hydrogen from acids under normal conditions. Therefore statement (i) is incorrect. The other statements (ii), (iii) and (iv) are correct: higher oxidation states of manganese form stable oxides/fluorides; Mn3+ and Co3+ can act as oxidising agents in aqueous solution; Ti2+ and Cr2+ are reducing agents.
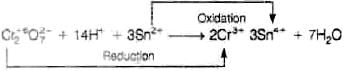
Q.18. When acidified K2Cr2O7 solution is added to Sn2+ salts then Sn2+ changes to
(i) Sn
(ii) Sn3+
(iii) Sn4+
(iv) Sn+
Ans: (iii)
Explanation: Acidified dichromate (Cr2O72-/Cr6+) is a strong oxidising agent and oxidises Sn2+ to Sn4+. Thus Sn2+ is converted to Sn4+ in the reaction. The redox equation can be shown with the appropriate half-reactions; the image placeholder shows the detailed ionic changes.
Q.19. Highest oxidation state of manganese in fluoride is +4 (MnF4) but highest oxidation state in oxides is +7 (Mn2O7) because ____________.
(i) Fluorine is more electronegative than oxygen.
(ii) Fluorine does not possess d-orbitals.
(iii) Fluorine stabilises lower oxidation state.
(iv) In covalent compounds fluorine can form single bond only while oxygen forms double bond.
Ans: (iv)
Explanation: Oxygen can form multiple bonds (double bonds) with metals, allowing stabilisation of higher oxidation states through stronger covalent bonding (for example Mn-O double bonds in oxides). Fluorine forms only single bonds (and prefers to remain as F-), which limits the highest oxidation state stabilised in fluorides. Hence manganese attains higher oxidation state (+7) in oxides but reaches lower maximum in fluorides; therefore option (iv) is correct.
Q.20. Although Zirconium belongs to 4d transition series and Hafnium to 5d transition series even then they show similar physical and chemical properties because __________.
(i) Both belong to d-block.
(ii) Both have same number of electrons.
(iii) Both have similar atomic radius.
(iv) Both belong to the same group of the periodic table.
Ans: (iii)
Explanation: Due to the lanthanoid contraction, the 5d element hafnium (Hf) has an atomic radius very similar to that of 4d zirconium (Zr). This near-identity of atomic and ionic radii (Zr ≈ 160 pm, Hf ≈ 159 pm; Zr4+ ≈ 79 pm, Hf4+ ≈ 78 pm) leads to very similar chemical and physical properties, so option (iii) is the correct reason.
Q.21. Why is HCl not used to make the medium acidic in oxidation reactions of KMnO4 in acidic medium?
(i) Both HCl and KMnO4 act as oxidising agents.
(ii) KMnO4 oxidises HCl into Cl2 which is also an oxidising agent.
(iii) KMnO4 is a weaker oxidising agent than HCl.
(iv) KMnO4 acts as a reducing agent in the presence of HCl.
Ans: (ii)
Explanation: If HCl is used to make the medium acidic, permanganate will oxidise chloride ions to chlorine gas: MnO4- + 8H+ + 5Cl- → Mn2+ + 4H2O + 5/2 Cl2 (overall). The chlorine produced is itself an oxidising agent and can interfere with the intended redox reaction by oxidising the reducing agent. Therefore HCl is avoided and acids such as H2SO4 are preferred; option (ii) is correct.
MULTIPLE CHOICE QUESTIONS (TYPE - II)
Q.22. Generally transition elements and their salts are coloured due to the presence of unpaired electrons in metal ions. Which of the following compounds are coloured?
(i) KMnO4
(ii) Ce (SO4)2
(iii) TiCl4
(iv) Cu2Cl2
Ans: (i, ii)
Explanation: KMnO4 is intensely coloured (violet) mainly due to ligand-to-metal charge transfer transitions rather than simple d-d transitions, because Mn in KMnO4 is in a high oxidation state. Ce(SO4)2 contains Ce(IV) with f0 configuration; its colour (often yellow) arises from charge transfer transitions (Ce(IV) ↔ ligand) rather than f-f transitions. TiCl4 is a colourless molecular compound and Cu2Cl2 contains Cu(I) (d10) and is typically white or colourless. Thus (i) and (ii) are the coloured compounds.
Q.23. Transition elements show magnetic moment due to spin and orbital motion of electrons. Which of the following metallic ions have almost same spin only magnetic moment?
(i) Co2+
(ii) Cr2+
(iii) Mn2+
(iv) Cr3+
Ans: (i, iv)
Explanation: Count the number of unpaired electrons for each ion (in high-spin states typical for many complexes):
Co2+ (3d7) → 3 unpaired electrons;
Cr2+ (3d4) → 4 unpaired electrons;
Mn2+ (3d5) → 5 unpaired electrons;
Cr3+ (3d3) → 3 unpaired electrons.
Thus Co2+ and Cr3+ both have three unpaired electrons and therefore almost the same spin-only magnetic moment (≈ √[3×5] ≈ 3.87 B.M.). Hence (i) and (iv) are correct.
Q.24. In the form of dichromate, Cr (VI) is a strong oxidising agent in acidic medium but Mo (VI) in MoO3 and W (VI) in WO3 are not because ___________.
(i) Cr (VI) is more stable than Mo(VI) and W(VI).
(ii) Mo(VI) and W(VI) are more stable than Cr(VI).
(iii) Higher oxidation states of heavier members of group-6 of transition series are more stable.
(iv) Lower oxidation states of heavier members of group-6 of transition series are more stable.
Ans: (ii, iii)
Explanation: For the heavier members of group 6 (Mo and W), the +6 oxidation state is relatively more stable due to better stabilisation by oxygen and greater availability of d-orbitals, so Mo(VI) and W(VI) are more stable than Cr(VI). Consequently, Cr(VI) (as dichromate) is a stronger oxidising agent because it is less stable and more readily reduced. Thus (ii) and (iii) together explain the behaviour.
Q.25. Which of the following actinoids show oxidation states upto +7?
(i) Am
(ii) Pu
(iii) U
(iv) Np
Ans: (ii, iv)
Explanation: The oxidation states of the given actinoids are as follows:
(i) Americium (Z = 95); Electronic configuration = [Rn] 5f76d07s2
Oxidation states shown by Am = + 3, + 4, + 5, + 6.
(ii) Plutonium (Z = 94); Electronic configuration = [Rn] 5f66d07s2
Oxidation states shown by Pu = + 3, + 4, + 5, + 6, + 7.
(iii) Uranium (Z = 92); Electronic configuration = [Rn] 5f36d17s2
Oxidation states shown by U = + 3, + 4, + 5, + 6.
(iv) Neptunium (Z = 93); Electronic configuration = [Rn] 5f46d17s2
Oxidation states shown by Np = + 3, + 4, + 5. + 6, + 7.
Q.26. General electronic configuration of actionoids is (n-2)f1-14 (n-1)d0-2ns2. Which of the following actinoids have one electron in 6d orbital?
(i) U (Atomic number. 92)
(ii) Np (Atomic number.93)
(iii) Pu (Atomic number. 94)
(iv) Am (Atomic number. 95)
Ans: (i, ii)
Explanation: General electronic configuration of actinoids is (n-1)f1-14(n-1)d0-2ns2. U and Np each have one electron in 6d orbital.
(i) Uranium (Z = 92); Electronic configuration = [Rn] 5f36d17s2
(ii) Neptunium (Z = 93); Electronic configuration = [Rn] 5f46d17s2
(iii) Plutonium (Z = 94); Electronic configuration = [Rn] 5f66d07s2
(iv) Americium (Z = 95); Electronic configuration = [Rn] 5f76d07s2
Q.27. Which of the following lanthanoids show +2 oxidation state besides the characteristic oxidation state +3 of lanthanoids?
(i) Ce
(ii) Eu
(iii) Yb
(iv) Ho
Ans: (ii, iii)
Explanation: (i) Cerium (Z = 57) ⇒ Electronic configuration = [Xe]4f55d°6s2
Oxidation state of Ce = +3, + 4
(ii) Europium (Z = 63) ⇒ Electronic configuration = [Xe]4f75d°6s2
Oxidation state of Eu = +2, + 3
(iii) Ytterbium (Z = 70) ⇒ Electronic configuration = [Xe]4f145d°6s2
Oxidation state of Yb = + 2, + 3
(iv) Holmium (Z = 67) Þ Electronic configuration = [Xe]4f115d°6s2
Oxidation state of Ho = +3
Q.28. Which of the following ions show higher spin only magnetic moment value?
(i) Ti3+
(ii) Mn2+
(iii) Fe2+
(iv) Co3+
Ans: (ii, iii)
Explanation: Compare unpaired electrons (assuming high-spin configurations typical for many complexes): Ti3+ (3d1) → 1 unpaired; Mn2+ (3d5) → 5 unpaired; Fe2+ (3d6, high-spin) → 4 unpaired; Co3+ (3d6, often low-spin) generally has paired electrons and low spin contribution. Thus Mn2+ and Fe2+ show higher spin-only magnetic moments compared with Ti3+ and Co3+ (which is frequently low-spin). Hence (ii) and (iii) are correct.
Q.29. Transition elements form binary compounds with halogens. Which of the following elements will form MF3 type compounds?
(i) Cr
(ii) Co
(iii) Cu
(iv) Ni
Ans: (i, ii)
Explanation: Chromium and cobalt commonly form stable +3 halides such as CrF3 and CoF3. Copper normally forms +1 and +2 halides (CuF and CuF2), and nickel typically forms +2 halides (NiF2), so they do not commonly form MF3. Therefore options (i) and (ii) are correct.
Q.30. Which of the following will not act as oxidising agents?
(i) CrO3
(ii) MoO3
(iii) WO3
(iv)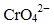
Ans: (ii, iii)
Explanation: A species acts as an oxidising agent when it can be readily reduced, i.e., when a high oxidation state is less stable and can accept electrons. For heavier group-6 elements (Mo and W), the +6 oxidation state (as MoO3, WO3) is relatively stable, so they are less powerful oxidisers under many conditions. Chromium in the +6 state (CrO3 / dichromate) is a strong oxidising agent because Cr(VI) is more easily reduced. Hence MoO3 and WO3 generally do not act as strong oxidising agents in the same way, so (ii) and (iii) are correct.
Q.31. Although +3 is the characteristic oxidation state for lanthanoids but cerium also shows +4 oxidation state because ________.
(i) It has variable ionisation enthalpy
(ii) It has a tendency to attain noble gas configuration
(iii) It has a tendency to attain f0 configuration
(iv) It resembles Pb4+
Ans: (ii, iii)
Explanation: Neutral cerium has configuration [Xe]4f15d16s2 (often written as 4f2 in some representations). On losing four electrons to form Ce4+ it attains the noble-gas configuration [Xe] (i.e., 4f0), which is particularly stable. Thus cerium can exist in both +3 and +4 states; the tendency to achieve a noble-gas (f0) configuration explains the +4 state. Therefore (ii) and (iii) are correct.
SHORT ANSWER TYPE QUESTIONS
Q.32. Why does copper not replace hydrogen from acids?
Ans: Copper does not replace hydrogen from acids because its standard reduction potential is positive (Eº for Cu2+/Cu is +0.34 V), indicating it is less reactive than hydrogen. Metals with negative standard potentials can displace H2 from acids; copper cannot, so it does not liberate hydrogen from acids.
Q.33. Why Eº values for Mn, Ni and Zn are more negative than expected?
Ans: The more negative Eº values for these metals reflect particular electronic stabilities and hydration effects:
- Mn2+ has a half-filled stability influence (arising from favourable electronic structure of 3d configuration), making the metal more reducing;
- Zn2+ corresponds to a fully filled d-subshell (3d10) which is electronically stable, so Zn tends to lose electrons less readily in standard conditions;
- Ni2+ shows a large negative Eº partly due to a highly negative enthalpy of hydration of Ni2+.
These effects make their standard potentials more negative than a simple periodic trend might predict.
Q.34. Why first ionisation enthalpy of Cr is lower than that of Zn?
Ans: Chromium has configuration [Ar]3d54s1, and removal of the 4s electron leads to a half-filled 3d5 configuration, which is relatively stable. Zinc has configuration [Ar]3d104s2, and removing an electron disturbs the filled 4s/3d arrangement; overall Zn requires more energy to remove the first electron in this context. Thus Cr has a lower first ionisation enthalpy than Zn because Cr attains a relatively stable half-filled d-subshell after ionisation.
Q.35. Transition elements show high melting points. Why?
Ans: Transition elements have several electrons (from (n-1)d and ns orbitals) available for metallic bonding. The participation of (n-1)d electrons increases the strength of the metallic bond, leading to high cohesive energies and therefore high melting points compared with metals where only s-electrons are involved.
Q.36. When Cu2+ ion is treated with KI, a white precipitate is formed. Explain the reaction with the help of chemical equation.
Ans: When Cu2+ is treated with KI, iodide reduces Cu2+ partially to Cu+, and copper(I) iodide (Cu2I2) precipitates as a white solid (which may darken on standing due to formation of iodine):
2Cu2+ + 4I- → 2CuI (s) + I2 (brown) (net behaviour shows precipitate formation and some iodine).
Q.37. Out of Cu2Cl2 and CuCl2, which is more stable and why?
Ans: CuCl2 (copper(II) chloride) is more stable than Cu2Cl2 (copper(I) chloride) in aqueous solution. This is because Cu2+ has a more favourable hydration enthalpy (more negative ΔhydHº) that compensates for the second ionisation energy of copper, stabilising the +2 oxidation state in water. Copper(I) (Cu+) tends to disproportionate into Cu and Cu2+ in aqueous solution, making CuCl2 the more stable species in that medium.
Q.38. When a brown compound of manganese (A) is treated with HCl it gives a gas (B). The gas taken in excess, reacts with NH3 to give an explosive compound (C). Identify compounds A, B and C.
Ans. The brown manganese compound A is MnO2 (manganese dioxide). When MnO2 reacts with hydrochloric acid it liberates chlorine gas (B, Cl2):
(i) MnO2 + 4HCl → MnCl2 + Cl2 + 2H2O.
Chlorine gas reacts with excess ammonia to give nitrogen trichloride (C, NCl3), an explosive compound:
(ii) 
(iii) Thus A = MnO2, B = Cl2, C = NCl3.
Q.39. Although fluorine is more electronegative than oxygen, but the ability of oxygen to stabilise higher oxidation states exceeds that of fluorine. Why?
Ans: Oxygen can form multiple bonds (for example M=O double bonds) with metals, which stabilises metals in high oxidation states through strong covalent bonding. Fluorine, while very electronegative, forms only single bonds (M-F) and cannot delocalise or share electron density in the same manner as oxygen. Therefore oxygen is better able to stabilise very high oxidation states of metals than fluorine.
Q.40. Although Cr3+ and Co2+ ions have same number of unpaired electrons but the magnetic moment of Cr3+ is 3.87 B.M. and that of Co2+ is 4.87 B.M. Why?
Ans: The observed magnetic moment has contributions from both spin and orbital angular momentum. For Cr3+ (3d3) the orbital contribution is negligible because of the symmetric distribution of electrons, so the magnetic moment is close to the spin-only value (≈ 3.87 B.M.). For Co2+ (3d7) there is a significant orbital contribution in many coordination environments, which increases the total magnetic moment above the spin-only value (often measured ≈ 4.7-5.2 B.M.). Thus the extra orbital contribution in Co2+ explains the higher observed moment.
Q.41. Ionisation enthalpies of Ce, Pr and Nd are higher than Th, Pa and U. Why?
Ans: Ce, Pr and Nd are lanthanoids with electrons entering the 4f orbitals; 4f orbitals are more strongly held (less effectively shielded) and penetrate closer to the nucleus than 5f orbitals. Actinoids (Th, Pa, U) have their 5f electrons less strongly bound and more available for bonding because 5f orbitals are more shielded and diffuse. Consequently the outer electrons of actinoids are less tightly held and ionisation enthalpies tend to be lower for actinoids than for the corresponding lanthanoids, making Ce, Pr and Nd have higher ionisation enthalpies than Th, Pa and U.
Q.42. Although Zr belongs to 4d and Hf belongs to 5d transition series but it is quite difficult to separate them. Why?
Ans: Separation of Zr and Hf is difficult because of lanthanoid contraction: the filling of the 4f orbitals in the lanthanoid series causes the ionic radii of subsequent 5d elements to be similar to their 4d congeners. Zr and Hf therefore have nearly identical atomic and ionic sizes (Zr ≈ 160 pm, Hf ≈ 159 pm), and very similar chemical behaviour, which makes their chemical separation challenging.
Q.43. Although +3 oxidation states is the characteristic oxidation state of lanthanoids but cerium shows +4 oxidation state also. Why?
Ans. Cerium can attain the +4 oxidation state because removal of the fourth electron gives the stable 4f0 configuration (the noble-gas core [Xe]). This extra stability of the f0 state makes the +4 oxidation state accessible for Ce in many compounds (for example CeO2, Ce(SO4)2).
Q.44. Explain why does colour of KMnO4 disappear when oxalic acid is added to its solution in acidic medium.
Ans: KMnO4 (permanganate, MnO4-, violet) is reduced by oxalic acid to Mn2+ (colourless). The reduction removes the permanganate chromophore and thus the violet colour disappears. The overall redox reaction in acidic medium is:

This shows MnO4- being reduced to Mn2+ while oxalic acid is oxidised to CO2, causing the decolourisation.
Q.45. When orange solution containing ion is treated with an alkali, a yellow solution is formed and when H+ ions are added to yellow solution, an orange solution is obtained. Explain why does this happen?
Ans: This is the interconversion between chromate and dichromate ions. The orange solution contains dichromate ion (Cr2O72-), which on addition of alkali shifts the equilibrium to chromate ion (CrO42-), producing a yellow solution. When acid (H+) is added to the yellow chromate solution, the equilibrium shifts back to the orange dichromate. The interconversion is reversible and pH-dependent:
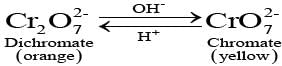
Q.46. A solution of KMnO4 on reduction yields either a colourless solution or a brown precipitate or a green solution depending on pH of the solution. What different stages of the reduction do these represent and how are they carried out?
Ans: The reduction products of permanganate depend strongly on pH:
In acidic medium (pH < 7): MnO4- is reduced to Mn2+ (colourless):
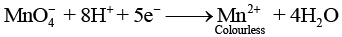
In alkaline medium (pH > 7): MnO4- is reduced to MnO42- (green):
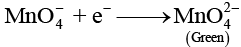
In neutral medium (pH ≈ 7): MnO4- may be reduced to MnO2 (brown precipitate):
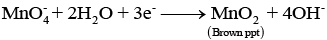
Thus the observed colour or precipitate indicates the oxidation state reached by manganese under the prevailing pH conditions.
Q.47. The second and third rows of transition elements resemble each other much more than they resemble the first row. Explain why?
Ans: Lanthanoid contraction leads to a decrease in expected atomic radii across the lanthanoid series, which makes the 4d and 5d transition elements have very similar sizes. Consequently, the second (4d) and third (5d) row transition elements resemble each other more closely in chemical behaviour and properties than either does the first (3d) row.
Q.48. Eº of Cu is + 0.34V while that of Zn is - 0.76V. Explain.
Ans: The standard electrode potential depends on the balance between the energies required to remove electrons (sublimation and ionisation energies) and the stabilisation gained (hydration enthalpy). For copper the sum of sublimation and ionisation energies is such that the resulting Cu2+ is not as strongly reducing, giving a positive Eº (+0.34 V). For zinc, removal of electrons leads to a stable 3d10 configuration (Zn2+), and the overall balance yields a negative standard potential (-0.76 V), making zinc a stronger reducing agent than copper.
Q.49. The halides of transition elements become more covalent with increasing oxidation state of the metal. Why?
Ans: As the oxidation state of the metal increases, the ionic radius of the metal ion decreases and its polarising power increases. According to Fajan's rules, increased polarising power leads to greater distortion of the electron cloud of the anion and hence increased covalent character. Therefore halides become more covalent at higher oxidation states.
Q.50. While filling up of electrons in the atomic orbitals, the 4s orbital is filled before the 3d orbital but reverse happens during the ionisation of the atom. Explain why?
Ans: According to the (n + l) rule, the 4s orbital has slightly lower energy than 3d for the neutral atom, so it fills first. After electrons occupy both orbitals, the relative energies change because of electron-electron interactions and penetration effects; the 4s orbital lies slightly higher in energy than the 3d in many ions and atoms with electrons present in both. Therefore on ionisation, the 4s electron is removed first because it is less tightly bound (higher in energy) than the 3d electrons.
Q.51. Reactivity of transition elements decreases almost regularly from Sc to Cu. Explain.
Ans: Reactivity correlates with ease of losing electrons (ionisation enthalpies). As we move from Sc to Cu across the first transition series, ionisation enthalpies generally increase because nuclear charge increases and electrons are held more tightly. As a result, the tendency of the elements to react (to lose electrons) decreases approximately regularly from Sc to Cu.
MATCHING TYPE
Note : Match the items of Column I and Column II in the following questions.
Q.52. Match the catalysts given in Column I with the processes given in Column II.
| Column I (Catalyst) | Column II (Process) |
| (i) Ni in the presence of hydrogen | (a) Zieglar Natta catalyst |
| (ii) Cu2Cl2 | (b) Contact process |
| (iii) V2O5 | (c) Vegetable oil to ghee |
| (iv) Finely divided iron | (d) Sandmeyer reaction |
| (v) TiCl4 + Al (CH3)3 | (e) Haber's Process |
| (f) Decomposition of KClO3 |
Ans: (i) → (c), (ii) → (d), (iii) → (b), (iv) → (e), (v) → (a)
Solution.
| Column I (Catalyst) | Column II (Process) |
| (i) Ni in the presence of hydrogen | (c) Vegetable oil to ghee |
| (ii) Cu2Cl2 | (d) Sandmeyer reaction |
| (iii) V2O5 | (b) Contact process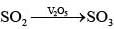 |
| (iv) Finely divided iron | (e) Haber's Process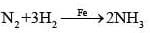 |
| (v) TiCl4 + Al (CH3)3 | (a) Zieglar Natta catalyst |
Q.53. Match the compounds/elements given in Column I with uses given in Column II.
| Column I (Compound/element) | Column II (Use) |
| (i) Lanthanoid oxide | (a) Production of iron alloy |
| (ii) Lanthanoid | (b) Television screen |
| (iii) Misch metal | (c) Petroleum cracking |
| (iv) Magnesium based alloy is constituent of | (d) Lanthanoid metal + iron |
| (v) Mixed oxides of lanthanoids are employed | (e) Bullets |
| (f) In X-ray screen |
Ans: (i) → (b), (ii) → (a), (iii) → (d), (iv) → (e), (v) → (c)
Solution.
| Column I (Compound/element) | Column II (Use) |
| (i) Lanthanoid oxide | (b) Television screen |
| (ii) Lanthanoid | (a) Production of iron alloy |
| (iii) Misch metal | (d) Lanthanoid metal + iron |
| (iv) Magnesium based alloy is constituent of | (e) Bullets |
| (v) Mixed oxides of lanthanoids are employed | (c) Petroleum cracking |
Q.54. Match the properties given in Column I with the metals given in Column II.
| Column I (Property) | Column II (Metal) |
| (i) An element which can show +8 oxidation state | (a) Mn |
| (ii) 3d block element that can show upto +7 oxidation state | (b) Cr |
| (iii) 3d block element with highest melting point | (c) Os |
| (d) Fe |
Ans: (i) → (c), (ii) → (a), (iii) → (b)
Solution.
(i) Osmium is known to show the +8 oxidation state (e.g., OsO4).
(ii) Manganese is the 3d element that can show oxidation states up to +7 (for example MnO4-).
(iii) Among the 3d elements, chromium has one of the highest melting points.
Q.55. Match the statements given in Column I with the oxidation states given in Column II.
| Column I | Column II |
| (i) Oxidation state of Mn in MnO2 is | (a) +2 |
| (ii) Most stable oxidation state of Mn is | (b) +3 |
| (iii) Most stable oxidation state of Mn in oxides is | (c) +4 |
| (iv) Characteristic oxidation state of lanthanoids is | (d) + 5 |
| (e) + 7 |
Ans. (i)→ (c), (ii) → (a), (iii) → (e), (iv) → (b)
(i) Oxidation state of Mn in MnO2 is +4.
(ii) Most stable oxidation state of Mn (elemental behaviour in many salts) is +2.
(iii) In certain oxides Mn attains high oxidation states; +7 occurs in permanganate species.
(iv) Characteristic oxidation state of lanthanoids is +3.
Q.56. Match the solutions given in Column I and the colours given in Column II.
| Column I | Column II |
| (Aqueous solution of salt) | (Colour) |
| (i) FeSO4.7H2O | (a) Green |
| (ii) NiCl2.4H2O | (b) Light pink |
| (iii) MnCl2.4H2O | (c) Blue |
| (iv) CoCl2.6H2O | (d) Pale green |
| (v) Cu2Cl2 | (e) Pink |
| (f) Colourless |
Ans: (i) → (d), (ii) → (a), (iii) → (b), (iv) → (e), (v) → (f)
| Column I | Column II |
| (Aqueous solution of salt) | (Colour) |
| (i) FeSO4.7H2O | (d) Pale green |
| (ii) NiCl2.4H2O | (a) Green |
| (iii) MnCl2.4H2O | (b) Light pink |
| (iv) CoCl2.6H2O | (e) Pink |
| (v) Cu2Cl2 | (f) Colourless |
Q.57. Match the property given in Column I with the element given in Column II.
| Column I (Property) | Column II (Element) |
| (i) Lanthanoid which shows +4 oxidation state | (a) Pm |
| (ii) Lanthanoid which can show +2 oxidation state | (b) Ce |
| (iii) Radioactive lanthanoid | (c) Lu |
| (iv) Lanthanoid which has 4f7 electronic configuration in +3 oxidation state | (d) Eu |
| (v) Lanthanoid which has 4f14 electronic configuration in +3 oxidation state | (e) Gd |
| (f) Dy |
Ans: (i) → (b), (ii) → (d), (iii) → (a), (iv) → (e), (v) → (c)
Solution.
(i) Lanthanoid which shows +4 oxidation state is cerium.
58Ce = [Xe] 4f2 5dº 6s2;Oxidation state = +3, +4
(ii) Lanthanoid which can show +2 oxidation states is europium.
63Eu =[Xe] 4f7 5dº 6s2;Oxidation state = + 2, +3
(iii) Radioactive lanthanoid is promethium. It is the only synthetic (man-made) radioactive lanthanoid.
(iv) Lanthnoid which has 4f7 electronic configuration in +3 oxidation state is gadolinium.
64Gd =[Xe] 4f7 5d1 6s2; Oxidation state = +3
(v) Lanthanoid which has 4f14 electronic configuration in +3 oxidation state is lutetium
71Lu = [Xe] 4f14 5d1 6s2;Oxidation state = +3
Q.58. Match the properties given in Column I with the metals given in Column II.
| Column I (Property) | Column II (Metal) |
| (i) Element with highest second ionisation enthalpy | (a) Co |
| (ii) Element with highest third ionisation enthalpy | (b) Cr |
| (iii) M in M (CO)6 is | (c) Cu |
| (iv) Element with highest heat of atomisation | (d) Zn |
| (e) Ni |
Ans. (i) → (c), (ii) → (d), (iii) → (b), (iv) → (a)
Solution.
(i) The highest second ionisation enthalpy in the first transition series is for Cu because Cu+ = 3d10 is very stable; removing a second electron is difficult.
(ii) Zinc as Zn2+ = 3d10 is very stable, so removal of a third electron requires very high energy.
(iii) The metal carbonyl with formula M(CO)6 is Cr(CO)6.
(iv) Nickel has the highest heat of atomisation among the first-row transition metals listed.
ASSERTION AND REASON TYPE
Note : In the following questions a statement of Assertion followed by a statement of Reason is given. Choose the correct answer out of the following choices.
(i) Both assertion and reason are true, and reason is the correct explanation of the assertion.
(ii) Both assertion and reason are true but reason is not the correct explanation of assertion.
(iii) Assertion is not true but reason is true.
(iv) Both assertion and reason are false.
Q.59. Assertion : Cu2+ iodide is not known.
Reason : Cu2+ oxidises I - to iodine.
(i) Both assertion and reason are true, and reason is the correct explanation of the assertion.
(ii) Both assertion and reason are true but reason is not the correct explanation of assertion.
(iii) Assertion is not true but reason is true.
(iv) Both assertion and reason are false.
Ans: (i)
Explanation:
(i) Assertion: Cu2+ iodide is not known. The statement is true because CuI2 is unstable and decomposes.
(ii) Reason: Cu2+ oxidises I- to iodine. The statement is true; Cu2+ oxidises iodide to iodine, preventing stable Cu(II) iodide from existing.
(iii) Justification: The reason correctly explains the assertion: Cu2+ oxidises I- to I2, so CuI2 is not isolable under normal conditions. Therefore choice (i) is correct.
Q.60. Assertion : Separation of Zr and Hf is difficult.
Reason : Because Zr and Hf lie in the same group of the periodic table.
(i) Both assertion and reason are true, and reason is the correct explanation of the assertion.
(ii) Both assertion and reason are true but reason is not the correct explanation of assertion.
(iii) Assertion is not true but reason is true.
(iv) Both assertion and reason are false.
Ans: (ii)
Explanation:
(i) Assertion: Separation of Zr and Hf is difficult. This is true.
(ii) Reason: They lie in the same group. While that is true, it is not the correct or complete explanation for the difficulty of separation.
(iii) Justification: The principal reason for the difficulty is lanthanoid contraction, which makes Zr and Hf have almost identical radii and extremely similar chemical properties; simply being in the same group is not the full explanation. Thus both statements are true but the reason is not the correct explanation, so (ii) is correct.
Q.61. Assertion : Actinoids form relatively less stable complexes as compared to lanthanoids.
Reason : Actinoids can utilise their 5f orbitals along with 6d orbitals in bonding but lanthanoids do not use their 4f orbital for bonding.
(i) Both assertion and reason are true, and reason is the correct explanation of the assertion.
(ii) Both assertion and reason are true but reason is not the correct explanation of assertion.
(iii) Assertion is not true but reason is true.
(iv) Both assertion and reason are false.
Ans: (iii)
Explanation:
(i) Assertion: Actinoids form relatively less stable complexes compared to lanthanoids. This assertion is not true in general-actinoids often form more stable and more covalent complexes because 5f orbitals can participate in bonding.
(ii) Reason: Actinoids can utilise 5f and 6d orbitals in bonding while lanthanoids do not effectively use 4f orbitals. This reason is true and explains why actinoid complexes tend to be more covalent and can be relatively more stable in some contexts. Therefore the assertion is incorrect but the reason is true, so choice (iii) applies.
Q.62. Assertion : Cu cannot liberate hydrogen from acids.
Reason : Because it has positive electrode potential.
(i) Both assertion and reason are true, and reason is the correct explanation of the assertion.
(ii) Both assertion and reason are true but reason is not the correct explanation of assertion.
(iii) Assertion is not true but reason is true.
(iv) Both assertion and reason are false.
Ans: (i)
Explanation:
(i) Assertion: Copper cannot liberate hydrogen from acids. This is true under normal conditions.
(ii) Reason: Copper has a positive standard electrode potential (Eº = +0.34 V). Metals with negative standard potentials displace H+ from acids to produce H2; copper's positive potential means it is less reducing than hydrogen and will not displace H2. Thus both statements are true and the reason correctly explains the assertion - choice (i).
Q.63. Assertion : The highest oxidation state of osmium is +8.
Reason : Osmium is a 5d-block element.
(i) Both assertion and reason are true, and reason is the correct explanation of the assertion.
(ii) Both assertion and reason are true but reason is not the correct explanation of assertion.
(iii) Assertion is not true but reason is true.
(iv) Both assertion and reason are false.
Ans: (ii)
Explanation:
(i) Assertion: Osmium has +8 as its highest oxidation state. This is true (for example, OsO4 contains Os in +8 state).
(ii) Reason: Osmium is a 5d-block element. While this is true, being a 5d element alone does not fully explain the existence of the +8 state. The ability of osmium to attain +8 depends on its electronic configuration and capacity to form multiple M=O bonds, utilising available d (and s) electrons for bonding. Hence both statements are true but the reason is not the complete/correct explanation of the assertion, so (ii) is correct.
LONG ANSWER TYPE QUESTIONS
Q.64. Identify A to E and also explain the reactions involved.
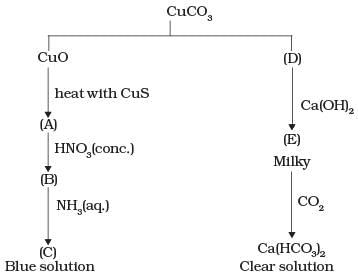
Ans: The substances from A to E are
A = Cu; B = Cu(NO3)2; C = [Cu(NH3)4]2+; D = CO2; E = CaCO3
Reactions:
(i)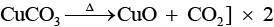
(ii)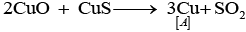
(iii)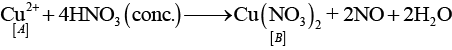
(iv)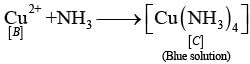
(v) 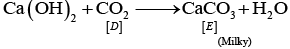
(vi) CaCO3 + H2O + CO2 → Ca HCO3)2
Q.65. When a chromite ore (A) is fused with sodium carbonate in free excess of air and the product is dissolved in water, a yellow solution of compound (B) is obtained. After treatment of this yellow solution with sulphuric acid, compound (C) can be crystallised from the solution. When compound (C) is treated with KCl, orange crystals of compound (D) crystallise out. Identify A to D and also explain the reactions.
Ans: This describes the commercial preparation of potassium dichromate from chromite ore.
A = FeCr2O4 (chromite ore); B = Na2CrO4 (sodium chromate, yellow); C = Na2Cr2O7 (sodium dichromate); D = K2Cr2O7 (potassium dichromate, orange).
(i)
(ii) 2Na2CrO4 + 2H+ → Na2Cr2O7 + 2Na+ + H2O.
(iii)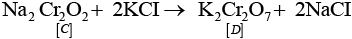
Q.66. When an oxide of manganese (A) is fused with KOH in the presence of an oxidising agent and dissolved in water, it gives a dark green solution of compound (B). Compound (B) disproportionates in neutral or acidic solution to give purple compound (C). An alkaline solution of compound (C) oxidises potassium iodide solution to a compound (D) and compound (A) is also formed. Identify compounds A to D and also explain the reactions involved.
Ans: This is the preparative sequence for potassium permanganate.
(A) = MnO2 (manganese dioxide);
(B) = K2MnO4 (potassium manganate, dark green);
(C) = KMnO4 (potassium permanganate, purple);
(D) = KIO3 (potassium iodate), formed when alkaline permanganate oxidises iodide.
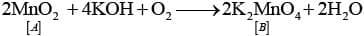
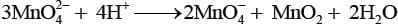
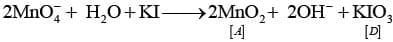
Q.67. On the basis of Lanthanoid contraction, explain the following :
(i) Nature of bonding in La2O3 and Lu2O3.
(ii) Trends in the stability of oxo salts of lanthanoids from La to Lu.
(iii) Stability of the complexes of lanthanoids.
(iv) Radii of 4d and 5d block elements.
(v) Trends in acidic character of lanthanoid oxides.
Ans:
(i) Lanthanoid contraction causes a steady decrease in ionic size across the series. La2O3 (with larger La3+) is more ionic in character, while Lu2O3 (with smaller Lu3+) has increased covalent character due to higher polarising power of the smaller cation.
(ii) As ionic size decreases from La to Lu, lattice energies and polarising powers change; generally the stability of some oxo salts decreases because smaller cations lead to stronger bonding that may favour different types of compounds. (Net effect: properties of oxo salts vary systematically across the series.)
(iii) Stability of complexes generally increases as ionic radius decreases because smaller lanthanoid ions form more stable coordinate bonds (higher polarising power) with ligands, improving complex stability.
(iv) Lanthanoid contraction makes the radii of 4d and 5d block elements very similar; hence many corresponding 4d and 5d elements show closely resembling chemistry.
(v) Acidic character of lanthanoid oxides increases from La to Lu as the cationic charge density (charge/size) increases with decreasing ionic radius, making oxides of heavier lanthanoids more acidic.
Q.68. (a) Answer the following questions :
(i) Which element of the first transition series has highest second ionisation enthalpy?
(ii) Which element of the first transition series has highest third ionisation enthalpy?
(iii) Which element of the first transition series has lowest enthalpy of atomisation?
(b) Identify the metal and justify your answer.
(i) Carbonyl M (CO)5
(ii) MO3F
Ans: (a) (i) Copper. Cu has configuration 3d104s1; removal of the second electron (from the filled d-shell) requires very high energy, giving a high second ionisation enthalpy.
(ii) Zinc. Zn2+ has a stable 3d10 configuration; removal of a third electron would break this stable filled d-shell and requires very high energy.
(iii) Zinc. Zinc has a completely filled 3d subshell and no unpaired electrons available for metallic bonding; this leads to relatively low enthalpy of atomisation compared with many transition metals.
(b) (i) Carbonyl M(CO)5 is Fe(CO)5.
Justification by the EAN (Effective Atomic Number) concept: EAN = atomic number of metal + 2×(number of CO ligands) = nearest noble gas. For M(CO)5, x + 2×5 = 36 (Kr), so x = 26 → Fe.
(ii) MO3F corresponds to MnO3F. If oxidation state of M is x: x + 3(-2) + (-1) = 0 ⇒ x -6 -1 = 0 ⇒ x = +7. M in +7 oxidation state among common transition metals is manganese, so the compound is MnO3F (Mn in +7).
Q.69. Mention the type of compounds formed when small atoms like H, C and N get trapped inside the crystal lattice of transition metals. Also give physical and chemical characteristics of these compounds.
Ans. Such compounds are called interstitial compounds. Their characteristic properties are:
(i) They have high melting points, higher than those of the pure metals.
(ii) They are very hard.
(iii) They retain metallic conductivity (they remain electrically conductive).
(iv) They are generally chemically inert compared with pure metals.
Q.70. (a) Transition metals can act as catalysts because these can change their oxidation state. How does Fe(III) catalyse the reaction between iodide and persulphate ions?
(b) Mention any three processes where transition metals act as catalysts.
Ans. (a) The reaction between iodide and persulphate ions is:
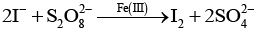
Fe(III) catalyses by a redox cycle:
2Fe3+ +2 I- →2 Fe2+ + I2
Then Fe2+ is reoxidised by persulphate (S2O82-):
2Fe2+ + S2O82- → 2Fe3+ + 2SO42-.
Overall the iron cycles between Fe(III) and Fe(II), increasing the reaction rate by providing an alternative pathway with lower activation energy.
(b) Examples of catalytic processes using transition metals:
(i) V2O5 is the catalyst in the contact process for oxidation of SO2 to SO3.
(ii) Finely divided iron is the catalyst in the Haber process for synthesis of ammonia (N2 + 3H2 → 2NH3).
(iii) MnO2 acts as a catalyst in the thermal decomposition of KClO3 to produce oxygen.
Q.71. A violet compound of manganese (A) decomposes on heating to liberate oxygen and compounds (B) and (C) of manganese are formed. Compound (C) reacts with KOH in the presence of potassium nitrate to give compound (B). On heating compound (C) with conc. H2SO4 and NaCl, chlorine gas is liberated and a compound (D) of manganese along with other products is formed.
Identify compounds A to D and also explain the reactions involved.
Ans: Since, compound (C) on treating with conc. H2SO4 and NaCl gives Cl2 gas, so it is manganese dioxide (MnO2). It is obtained along with MnO42- when KMnO4(violet) is heated
Thus, (A) =KMnO4
(B) = K2MnO4
(C) = MnO2
(D) =MnCl2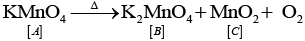
2MnO2 + 4KOH2 + O → 2K2 MnO4 + 2H2O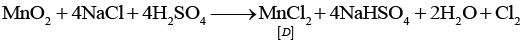
FAQs on NCERT Exemplar: The d & f-Block Elements
| 1. What are d-block and f-block elements? |  |
| 2. What are the properties of d-block elements? |  |
| 3. How do d-block elements form coordination compounds? |  |
| 4. What is the significance of f-block elements? |  |
| 5. How do d-block and f-block elements contribute to the color of transition metal compounds? |  |





