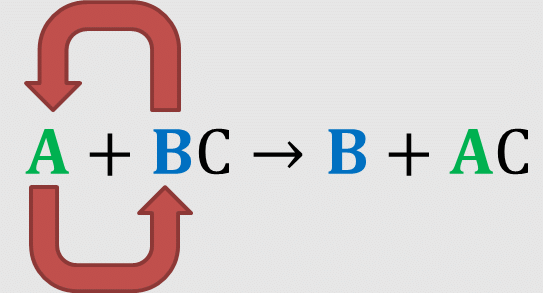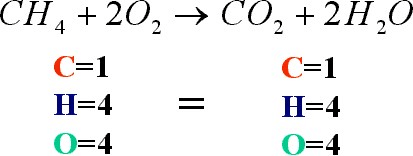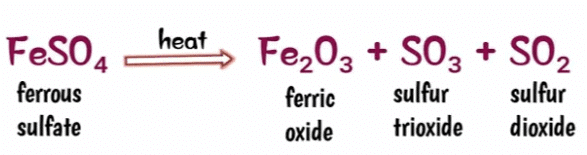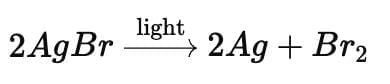Practice Questions: Chemical Reactions & Equations
Fill in the Blanks
Q 1. Addition of hydrogen in a substance in a reaction is known as ______ reaction.
Reduction
Q 2. In a ______ reaction, two or more substances combine to form a new single substance.
Combination
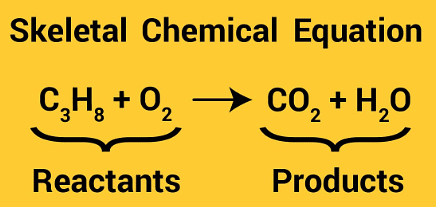
Q 3. Unbalanced reactions are also known as ______.
Skeletal
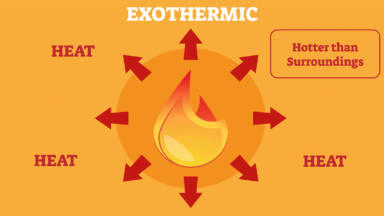
Q 4. Reactions in which heat is given out along with the products are called ______ reactions.
Exothermic
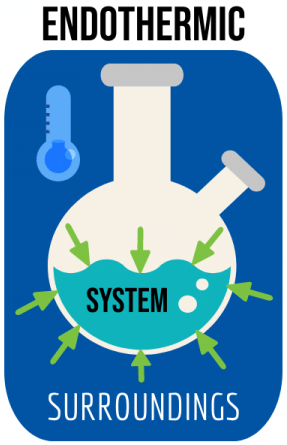
Q 5. Reactions in which energy is absorbed are known as ______.
Endothermic
Q 6. When an element displaces another element from its compound, a ______ reaction occurs.
Displacement
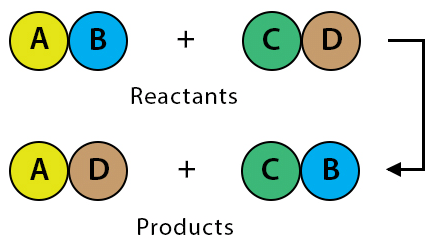
Q 7. Those reactions, in which two compounds react by an exchange of ions to form two new compounds, are called ______ reactions.
Double displacement
Q 8. Precipitation reactions produce ______ salts.
Insoluble
Q 9. The reduction is the ______ of oxygen or gain of hydrogen.
Loss
Q 10. The digestion of food in the body is an example of ______ reaction.
Exothermic reaction
Q 11. The addition of oxygen to a substance is called ______.
Oxidation
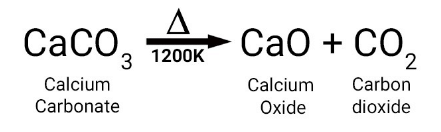
Q 12. When calcium carbonate is heated, it decomposes to give ______ and ______.
CaO(s) and CO2 (g)
Q 13. Complete the missing components/variables given as x and y in the following reactions:
(a) Pb (NO3)2 (aq) + 2KI (aq) → Pbl2 (x) + 2KNO3 (y)
(b) Cu (s) + 2AgNO3 (aq) → Cu (NO3)2 (aq) + x (s)
(a) x → (s); y → (aq)
(b) x → 2Ag
True and False
Q 1. The number of atoms of each element is conserved in any chemical reaction.
True
Explanation: The law of conservation of mass applies in all chemical equations. This means that the number of atoms of products present is conserved in the number of atoms of reactants.
Q 2. Oxidation is the loss of electrons from a substance.
True
Explanation: Oxidation is a reaction that removes an electron from a substance.
Q 3. Reduction is the gain of electrons by a substance.
True
Explanation: The gain of electrons is called reduction. Because any loss of electrons by one substance must be accompanied by a gain in electrons by something else, oxidation and reduction always occur together.
Q 4. A complete chemical equation represents the reactants, products and their physical states symbolically.
True
Explanation: A chemical equation is the symbolic representation of a chemical reaction in the form of symbols and formulae, wherein the reactant entities are given on the left-hand side and the product entities on the right-hand side.
Q 5. A magnesium ribbon burns with a dazzling flame in the air (oxygen) and changes into a white substance, magnesium oxide.
True
Explanation: When magnesium ribbon is burnt in the air that is combustion reaction of the magnesium metal. On burning in the presence of oxygen magnesium gives white colour magnesium oxide. Here, magnesium metal is getting oxidized into magnesium ion.
Q 6. Rusting is a double decomposition reaction.
False
Explanation: Rusting is not a double decomposition reaction. It is an oxidation (redox) reaction where iron (Fe) reacts with oxygen (O₂) and water (H₂O) to form hydrated iron(III) oxide (rust, Fe₂O₃·nH₂O). The process involves iron losing electrons (oxidation) to form Fe³⁺ ions, which combine with oxygen and water. A double decomposition reaction involves two compounds exchanging ions to form two new compounds, which does not occur in rusting.
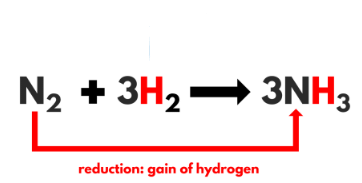
Q 7. The reaction between nitrogen and hydrogen to give ammonia is an example of a combination reaction.
True
Explanation: When hydrogen gas combines with nitrogen to form Ammonia.
The following chemical reaction will take place:
Our equilibrium reaction will be N2(g) + 3H2(g) ⇔ 2NH3(g) + Heat.In this case, Hydrogen and nitrogen react together to form ammonia.
Q 8. The action of heat on ferrous sulphate is an example of a decomposition reaction.
True
Explanation: On heating, ferrous sulphate crystals lose water, and anhydrous ferrous sulphate (FeSO4) is formed. So, their colour changes from light green to white. On further heating, anhydrous ferrous sulphate decomposes to form ferric oxide (Fe2O3), sulphur dioxide (SO2) and sulphur trioxide (SO3)The reaction for the decomposition of ferrous sulphate is as follows.
Q 9. The formation of Na+ and CI- ions from sodium and chlorine is an example of a redox reaction.
True
Explanation: When sodium metal reacts with chlorine gas to form sodium chloride (NaCl), the sodium metal loses an electron, which is then gained by chlorine.
Very Short Answer Type Questions
Q 1. Which substance is flushed in bags of potato chips to prevent rancidity?
Nitrogen
Q.2. What is the name of reaction in which an atom, molecule or ion gains oxygen?
Oxidation reaction
Q 3. What is the name of reaction in which an atom, molecule or ion loses oxygen?
Reduction reaction
Q 4. What is the chemical name of rust?
Iron oxide
Chemical Name of Rust is Iron (III) Oxide
Q 5. Which substance gets oxidised in the rancidity of food?
Fats present in food.
Q 6. In which reaction is heat generally evolved?
Exothermic reaction
Q 7. In which reaction is heat generally absorbed?
Endothermic reaction
Q 8. Which reaction is used in black and white photography?
Photolytic decomposition
Q 9. Which chemical substance is used to print the impression of black and white photography paper?
Silver chloride or Silver bromide.
Q 10. What is the chemical name of quick lime?
Calcium oxide
Q 11. What is the chemical name of limestone?
Calcium carbonate
Q 12. What is the chemical name of slaked lime?
Calcium hydroxide
Q 13. Which chemical substance is used in whitewashing?
Calcium hydroxide
Q 14. What is the chemical equation?
The symbolic representation of a chemical reaction is called a chemical equation.
Q 15. Can a combination reaction be a redox reaction?
Yes, a combination reaction can be a redox reaction.
In a combination reaction, two elements are combined to make a single product.
2H2 + O2 = 2H2O (water's formula)
FAQs on Practice Questions: Chemical Reactions & Equations
| 1. How do I identify the reactants and products in a chemical equation? |  |
| 2. What's the difference between a balanced and unbalanced chemical equation? |  |
| 3. Why do some reactions produce a gas or precipitate and how do I represent that? |  |
| 4. What does the arrow symbol mean in a chemical equation and are there different types? |  |
| 5. How can I remember the steps to balance a chemical equation for CBSE exams? |  |