Classification of Soils | Soil Mechanics - Civil Engineering (CE) PDF Download
Classification of Soils
It is adopted by IS code. It was given by A-Casagrande. It uses particle size distribution for coarse soils and plasticity for fine soils.
Objective
Sorting soils into groups showing similar behaviour based on index property, Generally used property are
- Grand Size Distribution
- Plasticity
Depending upon intended use different classification systems have evolved:
- Unified Soil Classification System (USCS)
Given by Casagrande
Intended for use in Airfield, Construction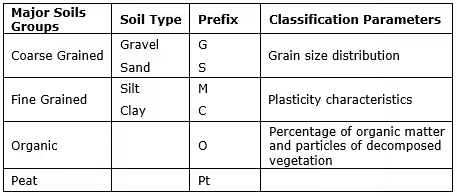
Note: ISCS is a modified USCS system. - AASHTO Classification System
(i) For Highway Construction
Soil Classified into 8 groups divided into subgroups based on group index. GI.
(ii) GI value ranges between
O(Good Subgrade Material) to 20 (Poor Subgrade Material) - Indian Standard Soil Classification System (ISSCS) % Fineness:
In the Indian Standard Soil Classification System (ISSCS), soils are classified into groups according to size, and the groups are further divided into coarse, medium and fine sub-groups.
The grain-size range is used as the basis for grouping soil particles into boulder, cobble, gravel, sand, silt or clay.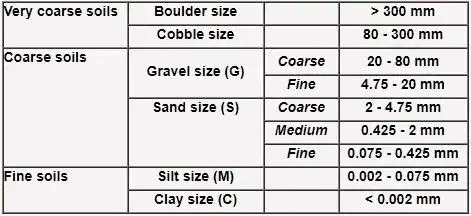
Gravel, sand, silt, and clay are represented by group symbols G, S, M, and C respectively.
Physical weathering produces very coarse and coarse soils. Chemical weathering produce generally fine soils.
(i) % of soil passing through the 75μ sieve.
(a) % Fineness < 50 % = Soil contain mainly
Coarse Grained fraction otherwise Fine grained fraction
(b) Fraction retained over the 75μ is undergone with plasticity studies, i.e. WL + IP identifies.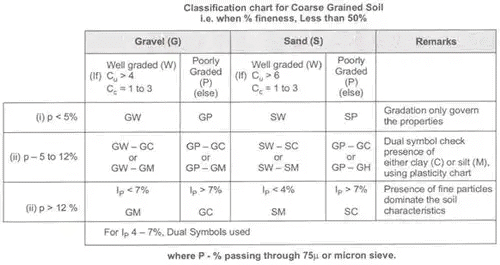


IS Code 1498 (1970)
IS 1498 is an Indian Standard code titled "Classification and Identification of Soils for General Engineering Purposes." Issued by the Bureau of Indian Standards (BIS), this code provides a systematic method for classifying soils based on their particle size distribution and plasticity characteristics. It adopts the Unified Soil Classification System (USCS) as a basis, categorizing soils into groups such as gravel, sand, silt, clay, and organic soils. The classification helps engineers and geotechnical professionals determine the suitability of soils for foundations, embankments, and other civil engineering works. IS 1498 plays a crucial role in ensuring consistency and reliability in soil investigation and design practices across India.
On the basis of fineness, coarse grain soils are further classified:
- Case-I: Well fineness is < 5%
(i) GW – Well graded gravel
Cu . 4
1 < Cc < 3
Fineness < 5%
(ii) GP – Poorly graded gravel
Above values of Cu and Cc are not satisfied.
(iii) SW – Well graded sand
Cu > 6
1 < Cc <3
(iv) SP – Poorly graded sand/uniformly graded sand Cu and Cc are not in range. - Case-II: If fineness is 5% to 12% the dual symbol are used.
(i) GW – GC well graded gravel containing clay.
Fineness – 5 to 12%
Clay > Silt
Gravel > Sand
Cu > 4; 1 ≤ Cc ≤ 3
(ii) GW – GM Well graded gravel containing silt
Cu > 4
1 ≤ Cu ≤ 3
Silt > Clay
Gravel > Sand
(iii) SW – SC Well graded sand containing clay
Sand > Gravel
Clay > Silt
Cu > 6
1 ≤ Cc ≤ 12%
(iv) SW – SM Well graded sand containing silt
Sand > Gravel
Silt > Silt
Cu > Clay
1 ≤ Cc ≤ 3
Fineness 5 to 12%
For poorly graded soils like GP-GC, GP, GM, SP-SC SP-SM the values of Cu and Cc are not satisfied. - Case-III: When fineness is more than 12%
GC: Clayey gravel
Gravel > Sand
Clay > Silt Ip > 7%
GM: Silty gravel
Sand < Gravel
Clay < Silt Ip < 4%
SC: Clayey silt
Sand > Gravel
Silti < Clay Ip > 7%
SM: Silty sand
Sand > Gravel
Silt > Clay Ip < 4%
Note: For IP between 4 and 7, Dual Symbols are used.
Classification of Fine Soils
- Silts (0.002 mm to 0.075 mm)
(i) Coarse 0.02 to 0.075 mm
(ii) Medium 0.01 to 0.02 mm
(iii) Fine 0.002 to 0.01 mm - Clay → <0.002 mm
(i) Low plastic soils (LL < 35%)
CL → Low plastic inorganic clay
ML → Low plastic silt
OL → Low plastic organic clay
(ii) Medium plastic soils (35% < 50%)
CI → Medium plastic inorganic clay
MI → Medium plastic silt
OI → Medium plastic organic clay
(iii) High plastic soils (LL > 50%)
CH → High plastic inorganic clay
MH → High plastic silt
OH → High plastic organic clay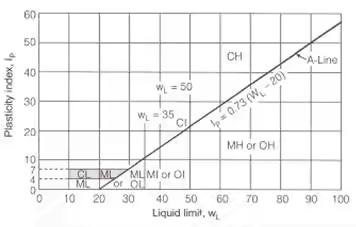 Equation of A-line IP = 0.73 (WL – 20)
Equation of A-line IP = 0.73 (WL – 20)
Equation of U-line IP = 0.9 (WL – 8)
Grain-Size Distribution Curve
The size distribution curves, as obtained from coarse and fine grained portions, can be combined to form one complete grain-size distribution curve (also known as grading curve). A typical grading curve is shown.
From the complete grain-size distribution curve, useful information can be obtained such as:
(i) Grading characteristics, which indicate the uniformity and range in grain-size distribution.
(ii) Percentages (or fractions) of gravel, sand, silt and clay-size.
A grading curve is a useful aid to soil description. The geometric properties of a grading curve are called grading characteristics.
To obtain the grading characteristics, three points are located first on the grading curve.
D60 = size at 60% finer by weight
D30 = size at 30% finer by weight
D10 = size at 10% finer by weight
The grading characteristics are then determined as follows:
(i) Effective size = D10
(ii) Uniformity coefficient, Cu = D60/D10
(iii) Curvature coefficient, Cc = (D30)2/D60D10
Both Cu and Cc will be 1 for a single-sized soil.
Cu > 5 indicates a well-graded soil, i.e. a soil which has a distribution of particles over a wide size range.
Cc between 1 and 3 also indicates a well-graded soil.
Cu < 3 indicates a uniform soil, i.e. a soil which has a very narrow particle size range.
Structure of Soil
- A soil particle may be a mineral or a rock fragment. A mineral is a chemical compound formed in nature during a geological process, whereas a rock fragment has a combination of one or more minerals. Based on the nature of atoms, minerals are classified as silicates, aluminates, oxides, carbonates and phosphates.
- Out of these, silicate minerals are the most important as they influence the properties of clay soils. Different arrangements of atoms in the silicate minerals give rise to different silicate structures.
Basic Structural Units
Soil minerals are formed from two basic structural units: tetrahedral and octahedral. Considering the valencies of the atoms forming the units, it is clear that the units are not electrically neutral and as such do not exist as single units.
The basic units combine to form sheets in which the oxygen or hydroxyl ions are shared among adjacent units. Three types of sheets are thus formed, namely silica sheet, gibbsite sheet and brucite sheet.
Isomorphous substitution is the replacement of the central atom of the tetrahedral or octahedral unit by another atom during the formation of the sheets.
The sheets then combine to form various two-layer or three-layer sheet minerals. As the basic units of clay minerals are sheet-like structures, the particle formed from stacking of the basic units is also plate-like. As a result, the surface area per unit mass becomes very large.
- A tetrahedral unit consists of a central silicon atom that is surrounded by four oxygen atoms located at the corners of a tetrahedron. A combination of tetrahedrons forms a silica sheet.
- An octahedral unit consists of a central ion, either aluminium or magnesium, that is surrounded by six hydroxyl ions located at the corners of an octahedron. A combination of aluminium-hydroxyl octahedrons forms a gibbsite sheet, whereas a combination of magnesium-hydroxyl octahedrons forms a brucite sheet.
- Montmorillonite Mineral
The bonding between the three-layer units is by van der Waals forces. This bonding is very weak and water can enter easily. Thus, this mineral can imbibe a large quantity of water causing swelling. During dry weather, there will be shrinkage. - Illite Mineral
Illite consists of the basic montmorillonite units but are bonded by secondary valence forces and potassium ions, as shown. There is about 20% replacement of aluminium with silicon in the gibbsite sheet due to isomorphous substitution. This mineral is very stable and does not swell or shrink. - Kaolinite Mineral
A basic kaolinite unit is a two-layer unit that is formed by stacking a gibbsite sheet on a silica sheet. These basic units are then stacked one on top of the other to form a lattice of the mineral. The units are held together by hydrogen bonds. The strong bonding does not permit water to enter the lattice. Thus, kaolinite minerals are stable and do not expand under saturation.
Kaolinite is the most abundant constituent of residual clay deposits.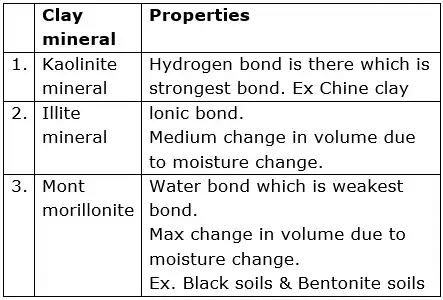
|
30 videos|126 docs|74 tests
|
FAQs on Classification of Soils - Soil Mechanics - Civil Engineering (CE)
| 1. What is soil classification in civil engineering? |  |
| 2. How is soil classified in civil engineering? |  |
| 3. Why is soil classification important in civil engineering? |  |
| 4. What are the different soil groups in the Unified Soil Classification System (USCS)? |  |
| 5. How does soil classification affect civil engineering projects? |  |

















