Isomerism in Alkanes, Alkenes & Alkynes
Isomerism
Isomerism is the phenomenon in which two or more distinct compounds have the same chemical formula but different structures and often different physical and chemical properties. Compounds that share an identical molecular formula but differ in the arrangement of atoms or in the spatial orientation of atoms are called isomers.
The word "isomer" is derived from the Greek words isos (equal) and meros (parts). The term was introduced by the Swedish chemist Jacob Berzelius in the year 1830.
Classification of Isomerism
Isomerism is broadly classified into two main types:
- Structural (constitutional) isomerism - isomers differ in the order in which atoms are bonded.
- Stereoisomerism - isomers have the same bond connectivity but differ in the three-dimensional arrangement of atoms.
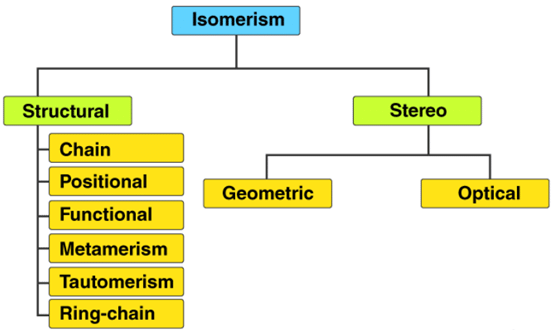
Structural (Constitutional) Isomerism
Structural isomers (constitutional isomers) are compounds having the same molecular formula but different connectivity of atoms. They are assigned different IUPAC names when the connectivity or functional groups differ.
Chain (Skeletal) Isomerism
Chain isomerism arises from different arrangements of the carbon skeleton (different branching).
- Also termed skeletal isomerism.
- Common in alkanes where branching can change the structure without changing molecular formula.
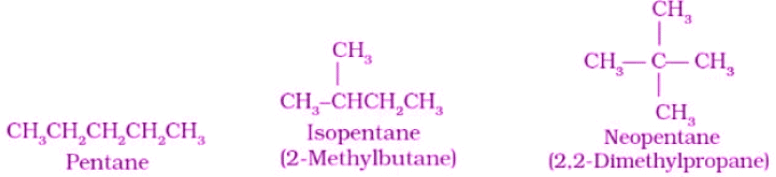
- Example: For C5H12, three chain isomers are possible: n-pentane, isopentane (2-methylbutane) and neopentane (2,2-dimethylpropane).
Position Isomerism
Position isomerism occurs when a functional group or substituent is attached to different carbon atoms in the same carbon chain.
- The molecular formula remains identical; the point of attachment differs.
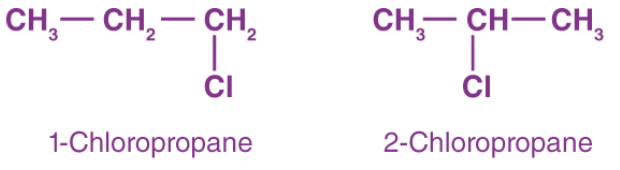
- Example: C3H7Cl gives 1-chloropropane and 2-chloropropane as position isomers.
- In alkenes and alkynes, position isomerism arises from different locations of the double or triple bond: e.g. 1-butene and 2-butene (C4H8).
Functional Group Isomerism
Functional isomerism (functional group isomerism) exists when isomers contain different functional groups while having the same molecular formula.
- The connectivity changes such that a distinct functional group results.
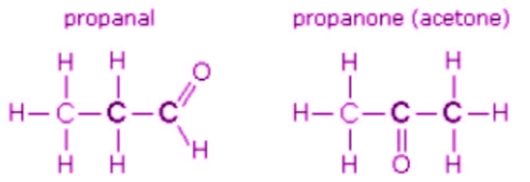
- Example: C3H6O can be propanal (an aldehyde) and propanone (acetone, a ketone).
- Functional isomerism is common among compounds having the same degree of unsaturation but different functional groups.
Metamerism
Metamerism is a type of structural isomerism observed in compounds that contain a polyvalent functional group (commonly divalent atoms such as oxygen, nitrogen or sulfur) with different alkyl groups on either side.
- Occurs mostly in ethers, thioethers, secondary amines, etc., where R-X-R' can have R ≠ R'.
- Example: The molecular formula C4H10O corresponds to different ethers such as ethoxyethane (C2H5-O-C2H5) and methoxypropane (CH3-O-C3H7).
- Metamers are structural isomers that differ in the nature or length of alkyl groups attached to the heteroatom.
Tautomerism (Special Case)
Tautomerism is a special kind of isomerism where isomers (called tautomers) readily interconvert by transfer of a proton and a shift of a double bond; they usually exist in dynamic equilibrium.
- Typical example: keto-enol tautomerism. A keto form (C=O) and its enol form (C=C-OH) interconvert by intramolecular proton transfer.
- Example: Acetaldehyde (ethanal) and its enol form (ethenol) - the keto form is generally more stable under normal conditions.
- Tautomerism has important implications in reactivity and biological systems because the two forms often have very different chemical behaviour.
Ring-Chain Isomerism
Ring-chain isomerism occurs when one isomer is an open-chain compound and another is a cyclic compound, both having the same molecular formula.
- The open-chain and cyclic forms differ in number and type of bonds (for example, the open-chain may contain a double bond that is absent in the ring form).
- Example: C3H6 corresponds to propene (an open-chain alkene) and cyclopropane (a cyclic alkane).
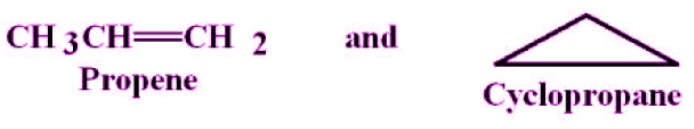
Stereoisomerism
Stereoisomerism arises when isomers have the same sequence of bonded atoms (same connectivity) but differ in the three-dimensional arrangement of those atoms. Stereoisomers often have markedly different chemical and physical properties.
Geometric Isomerism (Cis-Trans)
Geometric isomerism, also known as cis-trans isomerism, is due to restricted rotation about a bond (commonly a double bond or in cyclic systems) that results in distinct spatial arrangements of substituents.
- Occurs most commonly in alkenes where the C=C double bond prevents free rotation.
- Requirement for cis-trans isomerism in an alkene: each carbon of the double bond must have two different substituents.
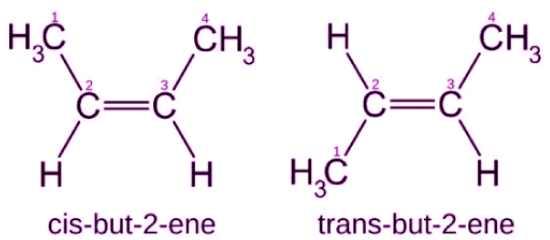
- Example: But-2-ene (CH3-CH=CH-CH3) exists as cis-but-2-ene (both CH3 groups on the same side) and trans-but-2-ene (CH3 groups on opposite sides).
- In cyclic compounds, restricted rotation about the ring bonds can also give cis and trans forms (substituents on the same or opposite faces of the ring).
- Alkanes do not show geometric isomerism because single C-C bonds allow free rotation; alkynes (C≡C) are linear at the triple-bonded carbons and therefore generally do not show cis-trans isomerism across the triple bond.
Optical Isomerism (Enantiomerism)
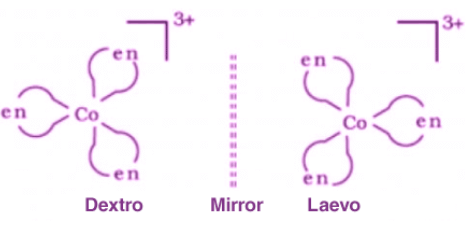
Optical isomerism occurs when molecules are non-superimposable mirror images of each other. Such pairs are called enantiomers.
- Optical isomers have the same connectivity but differ in spatial arrangement in a way that they are mirror images and cannot be superimposed.
- Presence of a chiral centre (typically a carbon atom bonded to four different groups) is a common cause of optical isomerism.
- Enantiomers rotate the plane of plane-polarised light in equal magnitude but opposite directions. A compound that rotates light to the right is called dextro (+); to the left is laevo (-).
- A racemic mixture contains equal amounts of both enantiomers and is optically inactive because the rotations cancel out.
- Example: 2-butanol (CH3-CH(OH)-CH2-CH3) contains a chiral carbon and exists as a pair of enantiomers.
- Optical isomerism is important in biology and pharmaceuticals because enantiomers can have different biological activities.
Isomerism in Alkanes, Alkenes and Alkynes - Specific Cases
This section summarises how the principal types of isomerism appear in alkanes, alkenes and alkynes and provides typical examples useful for class-level study.
- Alkanes (general formula CnH2n+2): Chain isomerism is most common; position isomerism applies to substituents (e.g. halogenated derivatives); alkanes do not exhibit geometric isomerism because single bonds rotate freely; optical isomerism may occur if a carbon atom has four different substituents (rare in simple straight-chain alkanes, more common in substituted derivatives).
- Alkenes (general formula CnH2n with one C=C): Position isomerism of the double bond is common (e.g. 1-butene vs 2-butene). Geometric (cis-trans) isomerism is possible when each carbon of the C=C has two different substituents, e.g. cis-2-butene and trans-2-butene. Optical isomerism can occur in substituted alkenes when chirality is present elsewhere in molecule, but the C=C itself does not create a chiral centre (except in special systems such as substituted allenes or cumulenes).
- Alkynes (general formula CnH2n-2 with one C≡C): Position isomerism occurs for internal vs terminal alkynes (e.g. 1-butyne vs 2-butyne). Alkynes are linear at the triple bond and generally do not show cis-trans isomerism across the triple bond. Optical isomerism can appear if the molecule contains a chiral centre elsewhere.
Practical Rules to Identify Common Isomer Types
- Compare molecular formulas; if identical, examine connectivity to decide structural isomerism.
- Look for differences in carbon skeleton (branching) to identify chain isomers.
- Check the position of functional groups, double bonds or triple bonds for position isomers.
- If functional groups differ (e.g. aldehyde vs ketone), identify functional group isomerism.
- For geometric isomerism in alkenes, confirm that each carbon of C=C has two different substituents; if yes, cis-trans (or E/Z) is possible.
- For optical isomerism, search for a carbon atom bonded to four different groups (a chiral centre); if present, enantiomers may exist.
Examples and Representative Structures
- C5H12: n-pentane, 2-methylbutane, 2,2-dimethylpropane - illustrates chain isomerism.
- C3H7Cl: 1-chloropropane and 2-chloropropane - position isomerism example.
- C3H6O: propanal and propanone - functional group isomerism.
- C3H6: propene and cyclopropane - ring-chain isomerism.
- But-2-ene: cis and trans forms - geometric isomerism.
- 2-butanol: two enantiomers due to a chiral centre - optical isomerism.
- 1-butyne and 2-butyne: position isomerism in alkynes; no cis-trans about the C≡C.
Importance and Applications
- Isomerism explains why compounds with the same formula can have different boiling points, melting points, densities, and chemical reactivities.
- In organic synthesis and drug design, recognising and controlling isomerism (especially stereochemistry and enantiomeric purity) is crucial because different isomers can have different biological activities.
- Industrial separation and characterisation techniques (e.g. chromatography, spectroscopy) often exploit differences among isomers.
FAQs on Isomerism in Alkanes, Alkenes & Alkynes
| 1. What is isomerism in alkanes, alkenes, and alkynes? |  |
| 2. How many types of isomerism exist in alkanes, alkenes, and alkynes? |  |
| 3. Can you provide an example of structural isomerism in alkanes? |  |
| 4. What is cis-trans isomerism in alkenes? |  |
| 5. How does isomerism affect the properties of alkanes, alkenes, and alkynes? |  |






