Matter in Our Surroundings Class 9 Worksheet Science Chapter 1
| Table of contents |

|
| Multiple Choice Questions |

|
| Fill in the Blanks |

|
| Very Short Answer Questions |

|
| Crossword Puzzle |

|
Multiple Choice Questions
Q.1. According to ancient philosophers matter consists of:
(a) Three constituents
(b) Four constituents
(c) Five constituents
(d) Six constituents.
Correct Answer is Option (c)
Matter is made up of five constituents also called tatvas (air, water, earth, fire and sky).
Q.2. Dry ice is:
(a) Solid ammonia
(b) Solid carbon dioxide
(c) Solid sulphur dioxide
(d) Normal ice
Correct Answer is Option (b)
Dry ice is solid carbon dioxide (CO2).
Q.3. Which of the following statements is not correct for liquid state?
(a) Particles are loosly packed in the liquid state
(b) Fluidity is the maximum in the liquid state
(c) Liquids can be compressed
(d) Liquids take up the shape of any container in which these are placed
Correct Answer is Option (b)
Fluidity is maximum in the gaseous state and not in the liquid state.
Q.4. Which of the following will sublime?
(a) Common salt
(b) Sugar
(c) Camphor
(d) Potassium nitrate
Correct Answer is Option (c)
Camphor, ammonium chloride, Phenophthelene sublimes i.e. change from solid to gaseous state directly without passing through liquid state upon heating.
Q.5. When the liquid starts boiling, the further heat energy which is supplied:
(a) Is lost to the surrounding as such
(b) Increases the temperature of the liquid
(c) Increases the kinetic energy of the particles in the liquid
(d) Is absorbed as latent heat of vaporisation by the liquid
Correct Answer is Option (d)
Heat is absorbed as latent heat of vapourisation. As long as liquid is not boiled, the heat energy which is supplied increases the kinetic energy of particles present in water. Once the liquid starts boiling the heat energy is used to brinchange in the state (liquid-gas). It is known as latent heat of vapourisation.
Fill in the Blanks
1. Matter is made up of small_________.
Correct Answer is Particles
2. The forces of attraction between the particles are _______ in solids, ______ in liquids and _________ in gases.
Correct Answer is Maximum, intermediate, minimum
3. __________ is the change of gaseous state directly to solid state without going through liquid state, and vice-versa.
Correct Answer is Deposition or Desublimation.
4. Evaporation causes __________.
Correct Answer is Cooling
5. Latent heat of fusion is the amount of heat energy required to change 1 kg of solid into liquid at its ________.
Correct Answer is Melting point
6. Solid, liquid and gas are called the three _______ of matter.
Correct Answer is States
7. The smell of perfume gradually spreads across a room due to ______.
Correct Answer is Diffusion
8. Rapid evaporation depends on the ______ area exposed to atmosphere.
Correct Answer is Surface
9. As the temperature of a system increases, the pressure of the gases ______.
Correct Answer is Increases
10. As the volume of a specific amount of gas decreases, it’s pressure _______.
Correct Answer is Increases
11. As the temperature of a gas decreases, It’s volume ______.
Correct Answer is Decreases
12. Gas molecules at higher temperatures have more _______ than at cooler temperatures.
Correct Answer is Kinetic energy
13. A sponge has minute ________, in which ________ is trapped.
Correct Answer is holes, air.
14. The pressure inside of a sealed tube if you raise the temperature go ______
Correct Answer is Up
15. Forces of attraction in liquids are _______ than in solid.
Correct Answer is Weaker
16. Latent heat of ________ is the amount of heat energy required to change 1 kg of solid into liquid at its melting point.
Correct Answer is fusion
Very Short Answer Questions
Q.1. Name one property which is shown by naphthalene and not by sodium chloride.
Naphthalene undergoes sublimation upon heating i.e. it directly changes into vapours. Whereas Sodium chloride (common salt) does not undergo sublimation. It melts on strong heating.
Q.2. A rubber band changes its shape when stretched. Can it be regarded as solid?
Rubber is a solid. It has elastic property due to which it undergoes change in shape when pressure is applied and regains its original shape when pressure is released.
Q.3. Gases can be compressed but solids cannot. Explain.
In gases, interparticle spaces are quite large. On applying pressure, these spaces decrease and the molecules of gas come closer. As a result, the gases can be compressed. Whereas in solids, particles are compactly packed leaving negligible interparticle space thus solids cannot be compressed.
Q.4. Define latent heat of vaporization.
Latent heat of vaporization is the heat energy required to change 1 kg of a liquid to gas at atmospheric pressure at its boiling point.
Q.5. What happens to the heat energy which is supplied to the solid once it has started melting?
Once the solid has started melting heat energy absorbed is consumed in bringing about the change in state from solid to liquid (overcoming the force of attraction between the particles of solids). The heat absorbed is known as latent heat of fusion.
Q.6.The freezing point of water is 0°C. What is the corresponding temperature on the Kelvin scale?
Temperature on Kelvin scale = 0°C+273 = 273K.
Q.7. Are the melting point temperature of the solid state and the freezing point temperature of the liquid state of a substance different?
No, these are the same. For example, melting point of ice and freezing point of water are both 0°C or 273 K.
Q.8. A substance is in liquid state at room temperature and changes into gas upon heating. What will you call its gaseous state?
The gaseous state of the substance is called vapour. Gaseous state of a substance which exists as liquid also is known as vapour.
Q.9. When a crystal of copper sulphate is placed at the bottom of a beaker containing water, the colour of water slowly becomes blue, why?
Copper sulphate on dissolution in water releases (Cu2+ ions) and SO42– ions. Due to diffusion of Cu2+ ions the colour of water slowly becomes blue.
Q.10. The boiling point of ethyl alcohol is 78°C. What is the corresponding temperature on kelvin scale?
Temperature on kelvin scale = 78 + 273 = 351 K
Crossword Puzzle
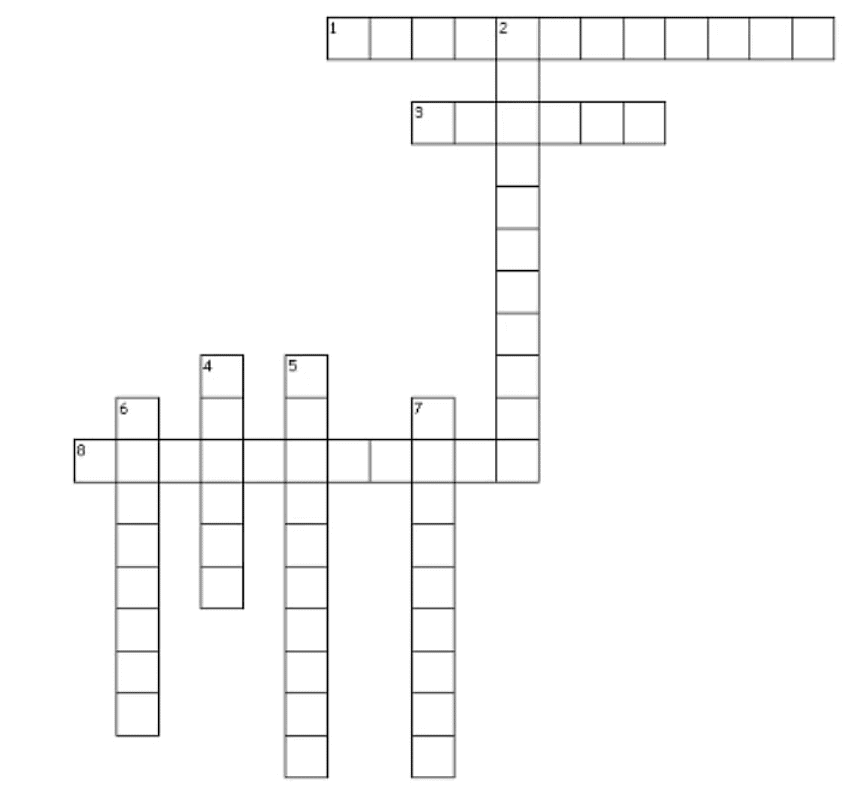
Across
1. BEC stands for Bose-Einstein-______
3. The state consists of super energetic and super excited particles
8. Conversion of solid to vapour is called ______
Down
2. This is the phenomenon of change of a liquid into vapours at any temperature below its boiling point
4. SI unit of Temperature
5. CNG stands ____ natural gas
6. It is the amount of water vapour present in air.
7. LPG stands for ______petroleum gas.
Answer:
1. Condensation
2. evaporation
3. plasma
4. kelvin
5. Compressed
6. Humidity
7. liquefied
8. sublimation
|
84 videos|541 docs|60 tests
|
FAQs on Matter in Our Surroundings Class 9 Worksheet Science Chapter 1
| 1. What is matter and how is it classified? |  |
| 2. What are the physical states of matter? |  |
| 3. How does temperature affect the state of matter? |  |
| 4. What is the difference between a physical change and a chemical change? |  |
| 5. What are some common examples of mixtures? |  |
















