Class 7 Exam > Class 7 Notes > Science Class 7 (Old NCERT) > NCERT Summary: Heat
Heat Summary Class 7 NCERT Summary Chapter 3
Hot and Cold
- The measure of degree of hotness or coldness of a substance is called its temperature.
- The device that is used to measure the temperature of a substance is called thermometer.
Measuring Temperature
Thermometers are of two types:
- Clinical thermometer: Used for measuring temperature of human body.
- Laboratory thermometer: Used for measuring temperature of common objects.

Reading a thermometer
- The temperature range of clinical thermometer is 35ºC to 42°C and that of laboratory thermometer is -10ºC to 110ºC. The unit for temperature is °C.
Step 1: Note the temperature difference indicated between the two bigger marks.
Step 2: Note down the number of divisions between these marks. If there are five divisions between them the, one small division can read 1/5 = 0.2°C
Step 3: Wash the thermometer with an antiseptic solution and give it a few jerks while holding firmly. Ensure that the level of mercury falls below 35°C.
Step 4: Now place the bulb of the thermometer under your tongue. Take the thermometer out after one minute and note the reading. This is your body temperature. The temperature should always be stated with its unit, °C.
- The normal temperature of human body is 37°C.
- The two most commonly used scales in which temperature is read are the Celsius scale and Fahrenheit scale.
Precaution for clinical thermometer
- Do not use a clinical thermometer for measuring the temperature of any object other than the human body.
- Also avoid keeping the thermometer in the sun or near a flame. It may break.
Precaution for laboratory thermometer
- In addition to the precautions needed while reading a clinical thermometer, the laboratory thermometer should be kept upright not tilted.
- Bulb should be surrounded from all sides by the substance of which the temperature is to be measured. The bulb should not touch the surface of the container.

Transfer of Heat
- Conduction:The transfer of heat in solids usually takes place by this method. Substances that allow heat to pass through them quite easily are called conductors and those that do not, are called insulators.
- The substances that allow heat to pass through them are called conductors of heat. Iron, copper, and aluminium are examples of conductors.
- The substances that do not allow heat to pass through them are called poor conductors of heat or insulators. Plastic and wood are examples of insulators.
- Convection: The transfer of heat in liquids and gases takes place by convection. Land breeze and sea breeze are a result of unequal heating of air present over land and sea by the sun.
- Radiation: Transfer of heat by radiation requires no medium. Heating of earth by sun is an example of heat transfer by radiation.
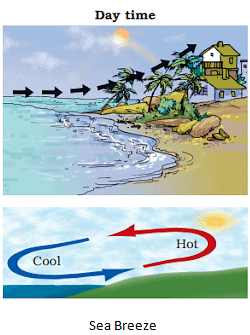
Sea Breeze
- The people living in the coastal areas experience an interesting phenomenon. During the day, the land gets heated faster than the water. The air over the land becomes hotter and rises up.
- The cooler air from the sea rushes in towards the land to take its place. The warm air from the land moves towards the sea to complete the cycle. The air from the sea is called the sea breeze.
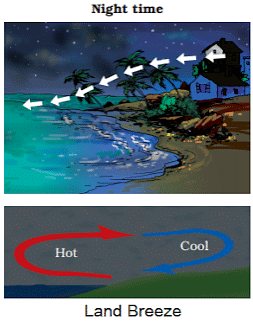
Land Breeze
- At night it is exactly the reverse. The water cools down more slowly than the land. So, the cool air from the land moves towards the sea. This is called the land breeze.
Kinds of clothes we wear in winter and Summer
- Dark surfaces absorb more heat and, therefore, we feel comfortable with dark coloured clothes in the winter.
- Light coloured clothes reflect most of the heat that falls on them and, therefore, we feel more comfortable wearing them in the summer.
- In the winter, we use woollen clothes. Wool is a poor conductor of heat. The air get trapped in between the wool fibres. This air prevents the flow of heat from our body to the cold surroundings which make us feel warm.
The document Heat Summary Class 7 NCERT Summary Chapter 3 is a part of the Class 7 Course Science Class 7 (Old NCERT).
All you need of Class 7 at this link: Class 7
|
111 videos|286 docs|28 tests
|
FAQs on Heat Summary Class 7 NCERT Summary Chapter 3
| 1. गर्मी क्या है और यह कैसे फैलती है ? |  |
Ans. गर्मी एक ऊर्जा का रूप है जो किसी वस्तु या प्रणाली के तापमान को बढ़ाने का काम करती है। यह तापीय ऊर्जा के रूप में होती है और यह तीन मुख्य तरीकों से फैलती है: संवहन (Conduction), संवहन (Convection), और विकिरण (Radiation)। संवहन में, गर्मी ठोस वस्तुओं में अणुओं के बीच संपर्क से फैलती है। संवहन में, गर्म हवा या तरल पदार्थ के प्रवाह से गर्मी फैलती है। विकिरण में, गर्मी बिना किसी माध्यम के भी फैल सकती है, जैसे कि सूर्य की किरणें।
| 2. गर्मी और तापमान में क्या अंतर है ? |  |
Ans. गर्मी और तापमान दोनों भौतिकी के महत्वपूर्ण अवधारणाएं हैं, लेकिन वे एक-दूसरे से भिन्न हैं। गर्मी ऊर्जा का एक रूप है जो तापमान को प्रभावित करता है, जबकि तापमान किसी वस्तु या प्रणाली की गर्मी की स्थिति को मापने का एक माप है। तापमान को डिग्री सेल्सियस या केल्विन में मापा जाता है, जबकि गर्मी joules में मापी जाती है।
| 3. गर्मी के प्रवाह को प्रभावित करने वाले कारक कौन से हैं ? |  |
Ans. गर्मी के प्रवाह को प्रभावित करने वाले कई कारक होते हैं, जैसे कि सामग्री की प्रकृति (उदाहरण: धातुओं की गर्मी संचालित करने की क्षमता अधिक होती है), तापमान का अंतर (जितना अधिक तापमान का अंतर होगा, गर्मी का प्रवाह उतना ही तेजी से होगा), और सतह का क्षेत्र (बड़ी सतह क्षेत्र वाले वस्त्रों में गर्मी का प्रवाह अधिक होता है)।
| 4. गर्मी के तीन रूप कौन से हैं ? |  |
Ans. गर्मी के तीन मुख्य रूप होते हैं: संवहन, संवहन, और विकिरण। संवहन में गर्मी का प्रवाह ठोस वस्तुओं के बीच संपर्क द्वारा होता है। संवहन में, गर्म वायु या तरल पदार्थ के प्रवाह से गर्मी फैलती है। विकिरण में, गर्मी बिना किसी माध्यम के भी फैल सकती है, जैसे कि सूर्य से आने वाली गर्मी।
| 5. तापीय संतुलन क्या है ? |  |
Ans. तापीय संतुलन वह स्थिति है जब दो या दो से अधिक वस्तुओं के बीच कोई गर्मी का प्रवाह नहीं होता है, यानी उनकी तापमान समान होता है। जब दो वस्तुएं एक साथ संपर्क में होती हैं और अंततः समान तापमान पर पहुँच जाती हैं, तो उन्हें तापीय संतुलन में कहा जाता है। यह स्थिति तब तक बनी रहती है जब तक कि बाहरी परिस्थितियों में कोई बदलाव नहीं होता।
Related Searches






















