Worksheets With Solutions: Chemical & Chemical Changes
Section A: Multiple Choice Questions
Q.1. Which of the following is not a chemical substance?
(a) Oxygen
(b) Water
(c) Sugar
(d) Sand
Ans: (d)
Explanation: Sand is not a pure chemical substance because it is made up of a mixture of different minerals and particles. A chemical substance is a pure material with a fixed composition and specific properties. For example, oxygen, water (H2O) and sugar (a pure compound like sucrose) have definite compositions, while sand does not.
Q.2. Which of the following is a chemical change?
(a) Melting of ice
(b) Burning of wood
(c) Cutting of paper
(d) Mixing of salt and water
Ans: (b)
Explanation: Burning of wood is a chemical change because new substances (such as carbon dioxide, water vapour and ash) are formed and energy (heat and light) is released. In a chemical change the composition of the original substance changes. The other options are physical changes: melting of ice changes only the state of water, cutting paper changes its shape, and mixing salt and water creates a mixture without forming new substances.
Q.3. What is the chemical symbol for gold?
(a) Ag
(b) Au
(c) Fe
(d) Pb
Ans: (b)
Explanation: The chemical symbol for gold is Au. Chemical symbols are short notations for elements; many are taken from the element's Latin name. In this case, Au comes from the Latin word aurum.
Q.4. Which of the following is a physical change?
(a) Rusting of iron
(b) Baking of a cake
(c) Digestion of food
(d) Burning of fuel
Ans: None of the above
Explanation: All four options involve chemical changes. Rusting of iron produces a new substance (iron oxide). Baking of a cake causes new substances to form as ingredients react. Digestion of food breaks down substances chemically. Burning of fuel is a combustion reaction that forms new products. Since a physical change does not produce new substances, none of these choices is a physical change.
Q.5. What is the chemical formula for water?
(a) H2SO4
(b) CO2
(c) NaCl
(d) H2O
Ans: (d)
Explanation: The chemical formula for water is H2O. A chemical formula shows the types of atoms and their numbers in a molecule: each water molecule has two hydrogen atoms and one oxygen atom.
Section B: Short Questions
Q.1. Define a chemical change.
Ans:
- Chemical change is a process in which one or more substances are changed into new substances with different chemical properties.
- It involves breaking and forming of chemical bonds between atoms or molecules.
- Examples include burning of wood, rusting of iron and digestion of food.
Q.2. What is the difference between a chemical substance and a mixture?
Ans:
- Chemical substance: A pure material with a fixed composition and definite properties (for example, water, oxygen, sodium chloride).
- Mixture: A combination of two or more substances mixed physically, not chemically bonded, and each keeps its own properties (for example, air, sand, sugar water).
Q.3. Write the chemical formula for sodium chloride.
Ans: NaCl.
Q.4. Give an example of a physical change.
Ans: Melting of ice. It is a physical change because only the state of water changes from solid to liquid and no new substance is formed.
Q.5. Explain what happens during crystallization.
Ans:
- During crystallization, a dissolved substance separates from a solution and forms solid crystals with a regular, repeating arrangement of its molecules or atoms.
- This usually happens when a hot, saturated solution cools or when the solvent evaporates, so the solute particles come together and arrange into a crystal lattice.
- An everyday example is purifying sugar by allowing sugar crystals to form from a concentrated solution.
Section C: Fill in the Blanks
Q.1. Chemical substances are made up of _____________.
Ans: Atoms
Q.2. The shorthand way of representing an element is its _______________.
Ans: Chemical symbol
Q.3. The combination of symbols and numbers which indicates the number of atoms in a molecule is called the _______________.
Ans: Chemical formula
Q.4. A chemical reaction is represented by a _____________.
Ans: Chemical equation
Q.5. The process of separating a solid from a solution by cooling it is called _______________.
Ans: Crystallization
Section D: Match the Column
Match the following symbols with their respective elements:
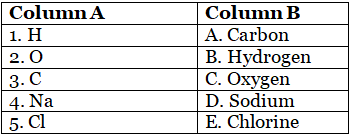
Ans:
Section E: True or False
Q.1. A chemical change is reversible.
Ans: False
Explanation: A chemical change usually produces new substances and is not easily reversed by simple physical methods. Reversing a chemical change often requires another chemical reaction or special processes, so it is generally considered not reversible.
Q.2. Physical changes do not change the nature of the substance.
Ans: True
Explanation: In a physical change the form, shape or state of a substance may change but its chemical composition remains the same. For example, melting ice changes water from solid to liquid without forming a new substance.
Q.3. Chemical formulae are used to represent the composition of a substance.
Ans: True
Explanation: A chemical formula shows which elements are present in a compound and the number of atoms of each element (for example, H2O tells us there are two hydrogen atoms and one oxygen atom in each water molecule).
Q.4. Crystallization is a physical change.
Ans: True
Explanation: Crystallization is a physical process in which a dissolved substance separates out of a solution and forms solid crystals. The substance does not change into a different chemical; only its physical form changes from dissolved to solid.
Q.5. Chemical reactions occur when substances are heated.
Ans: False
Explanation: Heating can cause or speed up many chemical reactions, but it is not the only way reactions occur. Chemical reactions can also take place at room temperature or under other conditions, so the statement is not always true.
FAQs on Worksheets With Solutions: Chemical & Chemical Changes
| 1. What are chemical changes? |  |
| 2. How can we identify a chemical change? |  |
| 3. What is the difference between a physical change and a chemical change? |  |
| 4. Can chemical changes be reversed? |  |
| 5. What role do catalysts play in chemical reactions? |  |