Metamerism and Ring Chain Isomerism
This type of isomerism is due to the unequal distribution of carbon atoms on either side of the multivalent functional group (i.e. -O-,-S-,-NH-, -CO- etc.) in the molecule of compounds belonging to the same class. For example, methyl propyl ether and diethyl ether both have same molecular formula
(a) CH3CH2COCH2CH3 is a metamer of CH3COCH2CH2CH3
Pentan-3-one Pentan-2-one
These two are also related as position isomers as the position of 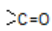 in the two isomers is different.
in the two isomers is different.
(b) CH3-O-CH2CH2CH3 is a metamer of CH3CH2O-CH2CH3
Methoxy propane Ethoxy ethane
(Methyl n-propyl ether) (Diethyl ether)
(c) CH3CH2-NH-CH2CH3 is a metamer of CH3-NH-CH2CH2CH3
Diethyl amine (Methyl n-propyl amine)
Ring-Chain Isomerism
Compounds having same molecular formula but possessing open chain and cyclic structures are called ring-chain isomers and the phenomenon is called ring-chain isomerism.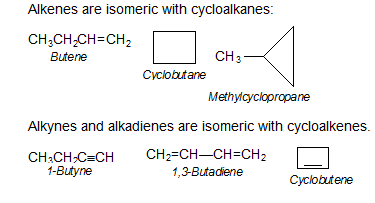
Degree of Unsaturation
Deficiency of two hydrogen atoms in a molecule is a result of either a pi-bond or a ring in the structure of that molecule. The sum of pi-bonds and rings in the structure of a compound collectively is called degree of unsaturation or double bond equivalents in that compound. The most general type of formula for any organic species is (CaHbNcOd).
If the compound contains other atoms also, the tetravalent atoms are replaced by carbon, monovalent atoms are replaced by hydrogen, divalent atoms are replaced by oxygen and trivalent atoms are replaced by nitrogen. Then all oxygen and all nitrogen atoms are removed from the formula. However, for the removal of each N atom, one H atom is removed from the molecular formula. As a result of all these operations, we will get a hydrocarbon. Now this concluded hydrocarbon is compared with saturated alkane to determine the degree of unsaturation or double bond equivalents.

