The SN1 Reaction & Characteristics of the SN1 Reaction | Chemistry Optional Notes for UPSC PDF Download
| Table of contents |

|
| Introduction |

|
| The Substrate |

|
| The Leaving Group |

|
| The Nucleophile |

|
| The Solvent |

|
| Strategy |

|
Introduction
- Most nucleophilic substitutions take place by the SN2 pathway just discussed. The reaction is favored when carried out with an unhindered substrate and a negatively charged nucleophile in a polar aprotic solvent, but is disfavored when carried out with a hindered substrate and a neutral nucleophile in a protic solvent.
- You might therefore expect the reaction of a tertiary substrate (hindered) with water (neutral, protic) to be among the slowest of substitution reactions. Remarkably, however, the opposite is true. The reaction of the tertiary halide 2-bromo-2-methylpropane (CH3)3CBr with H2O to give the alcohol 2-methyl-2-propanol is more than 1 million times faster than the corresponding reaction of CH3Br to give methanol.
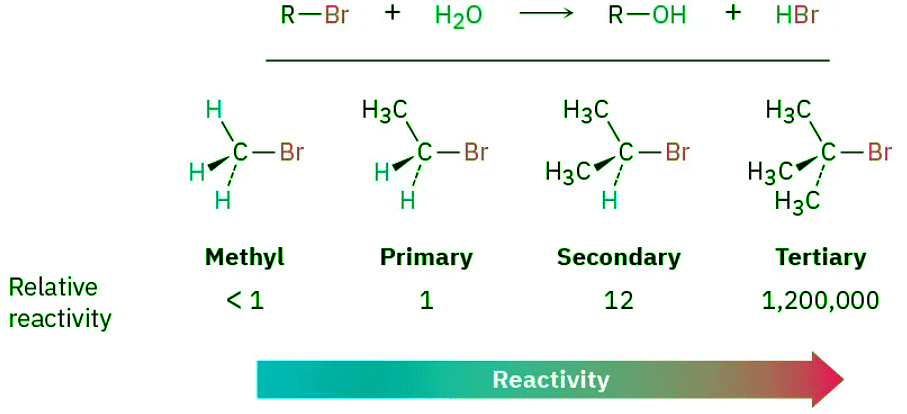
- A nucleophilic substitution reaction is occurring—a hydroxyl group is replacing a halogen—yet the reactivity order seems backward. These reactions can’t be taking place by the SN2 mechanism we’ve been discussing, so we must therefore conclude that they are occurring by an alternative substitution mechanism. This alternative mechanism is called the SN1 reaction, for substitution, nucleophilic, unimolecular.
- In contrast to the SN2 reaction of CH3Br with OH–, the SN1 reaction of (CH3)3CBr with H2O has a rate that depends only on the alkyl halide concentration and is independent of the H2O concentration. In other words, the process is a first-order reaction; the concentration of the nucleophile does not appear in the rate equation.
Reaction rate=Rate of disappearance of alkyl halide
= k × [RX] - To explain this result, we need to know more about kinetics measurements. Many organic reactions occur in several steps, one of which usually has a higher-energy transition state than the others and is therefore slower. We call this step with the highest transition-state energy the rate-limiting step, or rate-determining step. No reaction can proceed faster than its rate-limiting step, which acts as a kind of traffic jam, or bottleneck. In the SN1 reaction of (CH3)3CBr with H2O, the fact that the nucleophile concentration does not appear in the first-order rate equation means that it is not involved in the rate-limiting step and must therefore be involved in some other, non-rate-limiting step. The mechanism shown in Figure 11.9 accounts for these observations.
Figure 11.9 MECHANISM The mechanism of the SN1 reaction of 2-bromo-2-methylpropane with H2O involves three steps. Step 1 —the spontaneous, unimolecular dissociation of the alkyl bromide to yield a carbocation—is rate-limiting.
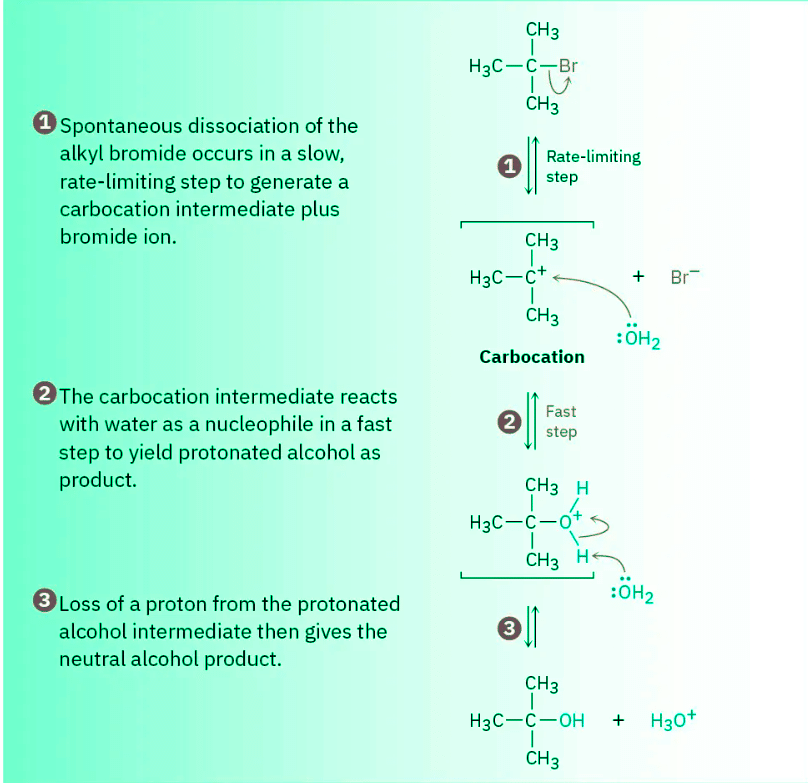
- Unlike what occurs in an SN2 reaction, where the leaving group is displaced while the incoming nucleophile approaches, an SN1 reaction takes place by loss of the leaving group before the nucleophile approaches. 2-Bromo-2-methylpropane spontaneously dissociates to the tert-butyl carbocation (CH3)3C+, plus Br– in a slow, rate-limiting step, and the intermediate carbocation is then immediately trapped by the nucleophile water in a faster second step. Thus, water is not a reactant in the step whose rate is measured. The energy diagram is shown in Figure 11.10.
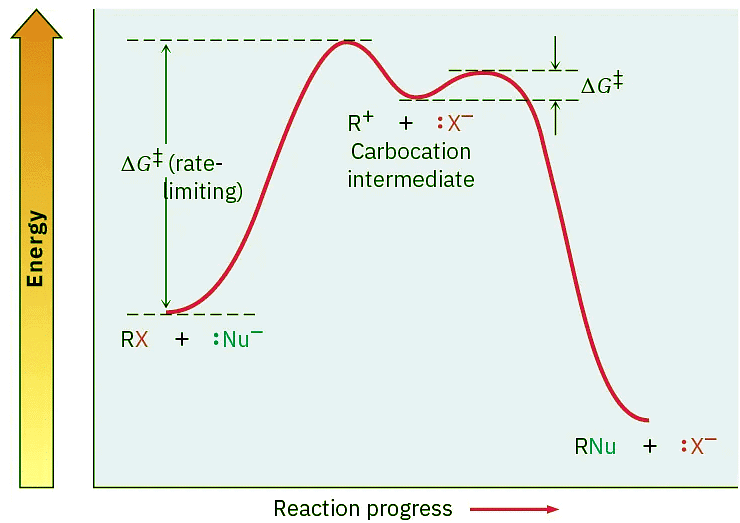
Figure 11.10: An energy diagram for an SN1 reaction. The rate-limiting step is the spontaneous dissociation of the alkyl halide to give a carbocation intermediate. Reaction of the carbocation with a nucleophile then occurs in a second, faster step.
- Because an SN1 reaction occurs through a carbocation intermediate, its stereochemical outcome is different from that of an SN2 reaction. Carbocations, as we’ve seen, are planar, sp2-hybridized, and achiral. Thus, if we carry out an SN1 reaction on one enantiomer of a chiral reactant and go through an achiral carbocation intermediate, the product loses its optical activity (Section 8.12). That is, the symmetrical intermediate carbocation can react with a nucleophile equally well from either side, leading to a racemic, 50 : 50 mixture of enantiomers (Figure 11.11).
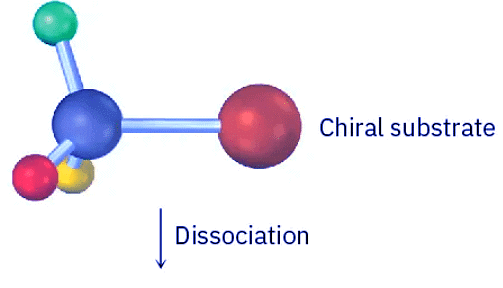
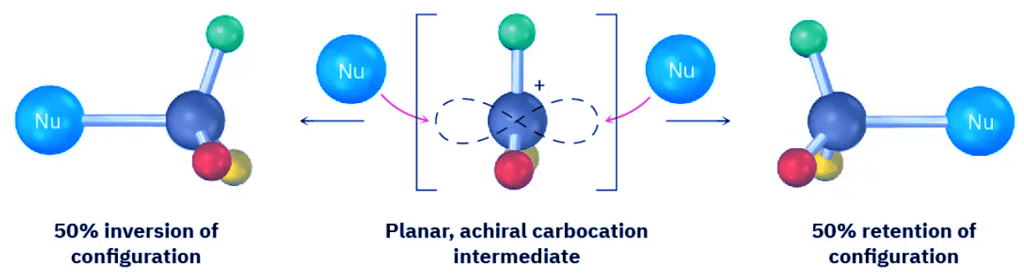
Figure 11.11: Stereochemistry of the SN1 reaction. Because the reaction goes through an achiral intermediate, an enantiomerically pure reactant gives an optically inactive racemic product.
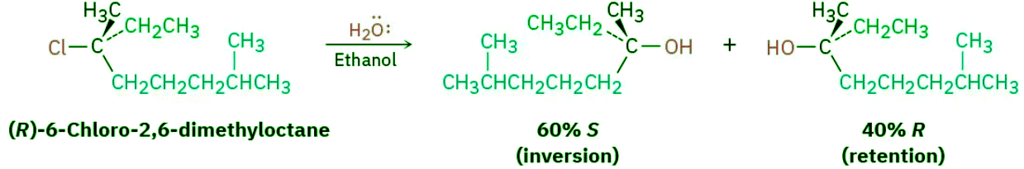
- The conclusion that SN1 reactions on enantiomerically pure substrates should give racemic products is nearly, but not exactly, what is found. In fact, few SN1 displacements occur with complete racemization. Most give a minor (0–20%) excess of inversion. The reaction of (R)-6-chloro-2,6-dimethyloctane with H2O, for example, leads to an alcohol product that is approximately 80% racemized and 20% inverted (80% R,S + 20% S is equivalent to 40% R + 60% S).
This lack of complete racemization in SN1 reactions is due to the fact that ion pairs are involved. According to this explanation, first proposed by Saul Winstein at UCLA, dissociation of the substrate occurs to give a structure in which the two ions are still loosely associated and in which the carbocation is effectively shielded from reaction on one side by the departing anion. If a certain amount of substitution occurs before the two ions fully diffuse apart, then a net inversion of configuration will be observed (Figure 11.12).
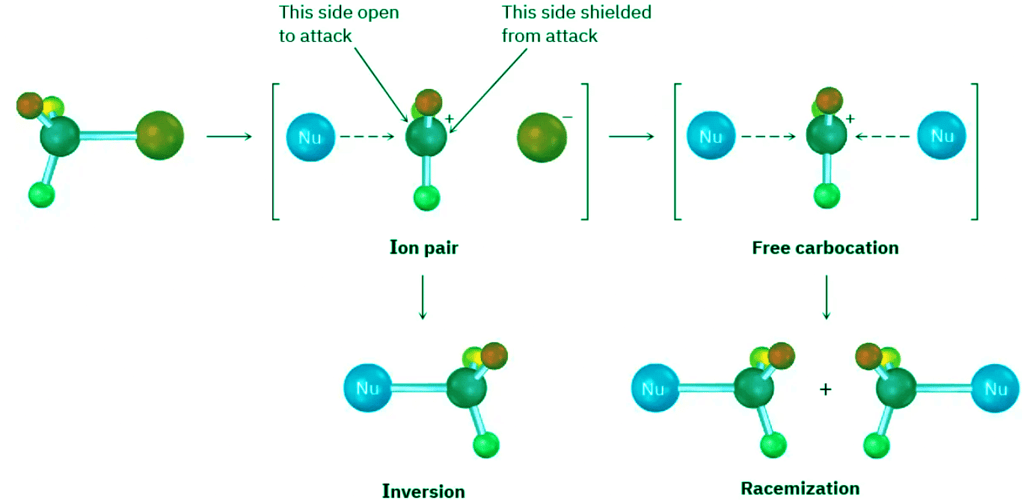
Figure 11.12: Ion pairs in an SN1 reaction. The leaving group shields one side of the carbocation intermediate from reaction with the nucleophile, thereby leading to some inversion of configuration rather than complete racemization.
The Substrate
- According to the Hammond postulate (Section 7.10), any factor that stabilizes a high-energy intermediate also stabilizes the transition state leading to that intermediate. Because the rate-limiting step in an SN1 reaction is the spontaneous, unimolecular dissociation of the substrate to yield a carbocation, the reaction is favored whenever a stabilized carbocation intermediate is formed. The more stable the carbocation intermediate, the faster the SN1 reaction.
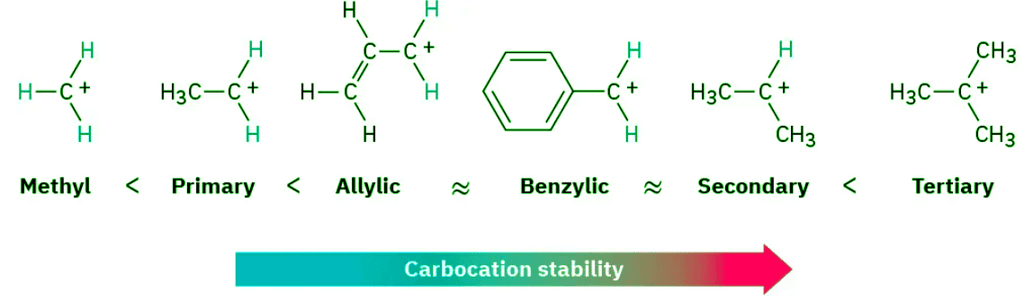
- We saw in Section 7.9 that the stability order of alkyl carbocations is 3° > 2° > 1° > methyl. To this list we should also add the resonance-stabilized allyl and benzyl cations. Just as allylic radicals are unusually stable because the unpaired electron can be delocalized over an extended π orbital system (Section 10.4), so allylic and benzylic carbocations are unusually stable. (The word benzylic means “next to an aromatic ring.”) As Figure 11.13 indicates, an allylic cation has two resonance forms. In one form, the double bond is on the “left”; in the other form it’s on the “right.” A benzylic cation has five resonance forms, all of which contribute to the overall resonance hybrid.
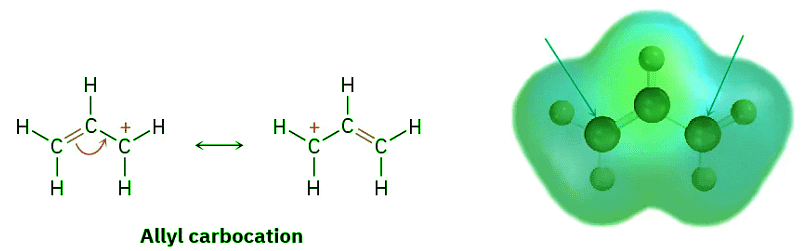
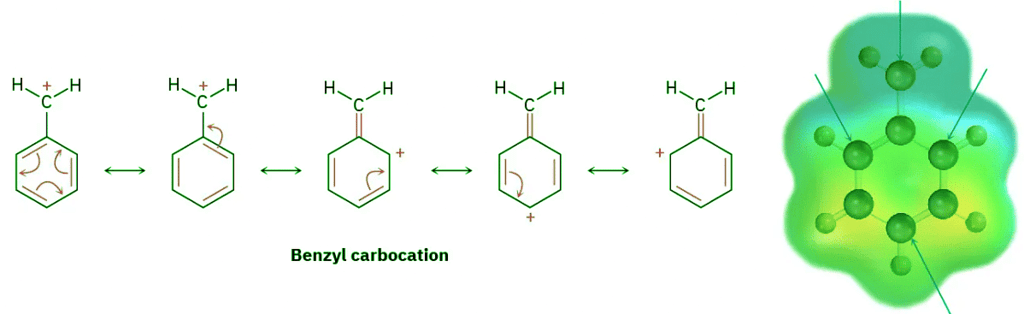
Figure 11.13: Resonance forms of allylic and benzylic carbocations. The positive charge is delocalized over the π system in both. Electron-poor atoms are indicated by blue arrows.
- Because of resonance stabilization, a primary allylic or benzylic carbocation is about as stable as a secondary alkyl carbocation, and a secondary allylic or benzylic carbocation is about as stable as a tertiary alkyl carbocation. This stability order of carbocations is the same as the order of SN1 reactivity for alkyl halides and tosylates.
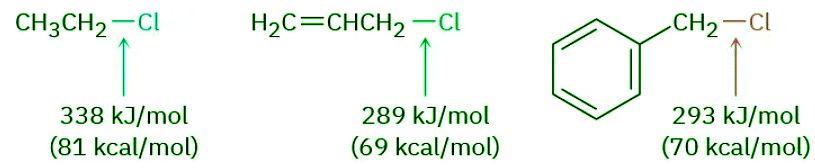
- We should also note parenthetically that primary allylic and benzylic substrates are particularly reactive in SN2 reactions as well as in SN1 reactions. Allylic and benzylic C–X bonds are about 50 kJ/mol (12 kcal/mol) weaker than the corresponding saturated bonds and are therefore more easily broken.
The Leaving Group
We said during the discussion of SN2 reactivity that the best leaving groups are those that are most stable; that is, those that are the conjugate bases of strong acids. An identical reactivity order is found for the SN1 reaction because the leaving group is directly involved in the rate-limiting step. Thus, the SN1 reactivity order is
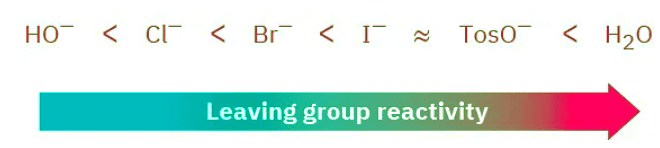
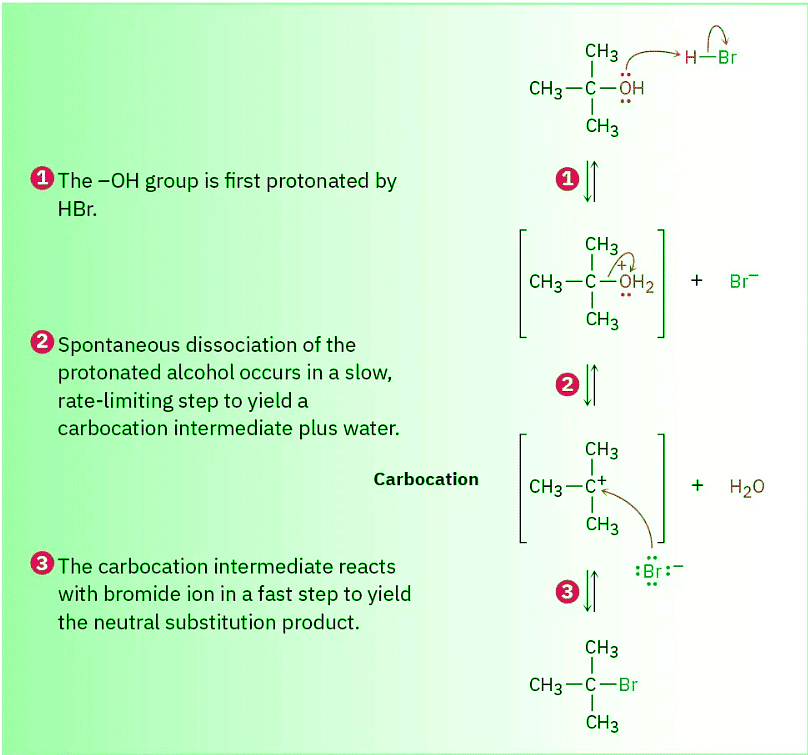
Note that in the SN1 reaction, which is often carried out under acidic conditions, neutral water is sometimes the leaving group. This occurs, for example, when an alkyl halide is prepared from a tertiary alcohol by reaction with HBr or HCl (Section 10.5). As shown in Figure 11.14, the alcohol is first protonated and then spontaneously loses H2O to generate a carbocation, which reacts with halide ion to give the alkyl halide. Knowing that an SN1 reaction is involved in the conversion of alcohols to alkyl halides explains why the reaction works well only for tertiary alcohols. Tertiary alcohols react fastest because they give the most stable carbocation intermediates.
Figure 11.14 MECHANISM The mechanism of the SN1 reaction of a tertiary alcohol with HBr to yield an alkyl halide. Neutral water is the leaving group (step 2).
The Nucleophile
- The nature of the nucleophile plays a major role in the SN2 reaction but does not affect an SN1 reaction. Because the SN1 reaction occurs through a rate-limiting step in which the added nucleophile has no part, the nucleophile can’t affect the reaction rate. The reaction of 2-methyl-2-propanol with HX, for instance, occurs at the same rate regardless of whether X is Cl, Br, or I. Furthermore, neutral nucleophiles are just as effective as negatively charged ones, so SN1 reactions frequently occur under neutral or acidic conditions.
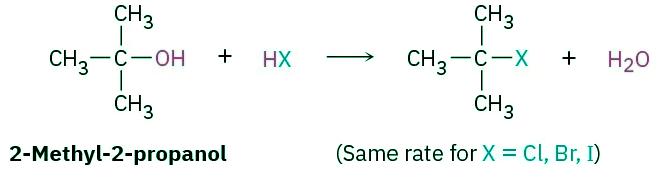
The Solvent
- What about the solvent? Do solvents have the same effect in SN1 reactions that they have in SN2 reactions? The answer is both yes and no. Yes, solvents have a large effect on SN1 reactions, but no, the reasons for the effects on SN1 and SN2 reactions are not the same. Solvent effects in the SN2 reaction are due largely to stabilization or destabilization of the nucleophile reactant, while solvent effects in the SN1 reaction are due largely to stabilization or destabilization of the transition state.
- The Hammond postulate says that any factor stabilizing the intermediate carbocation should increase the rate of an SN1 reaction. Solvation of the carbocation—the interaction of the ion with solvent molecules—has such an effect. Solvent molecules orient around the carbocation so that the electron-rich ends of the solvent dipoles face the positive charge (Figure 11.15), thereby lowering the energy of the ion and favoring its formation.
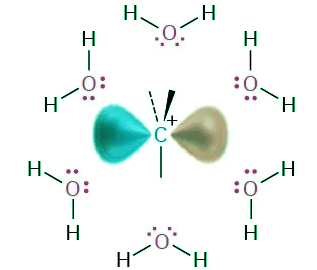
Figure 11.15: Solvation of a carbocation by water. The electron-rich oxygen atoms of solvent molecules orient around the positively charged carbocation and thereby stabilize it.
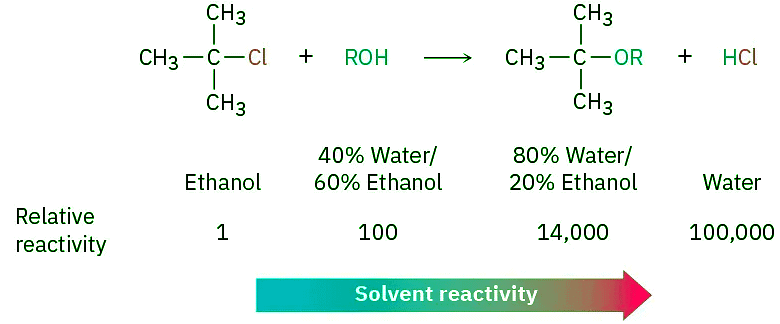
- The properties of a solvent that contribute to its ability to stabilize ions by solvation are related to the solvent’s polarity. SN1 reactions take place much more rapidly in strongly polar solvents, such as water and methanol, than in less polar solvents, such as ether and chloroform. In the reaction of 2-chloro-2-methylpropane, for example, a rate increase of 100,000 is observed upon going from ethanol (less polar) to water (more polar). The rate increases when going from a hydrocarbon solvent to water are so large they can’t be measured accurately.
It should be emphasized again that both the SN1 and the SN2 reaction show solvent effects, but that they do so for different reasons. SN2 reactions are disfavored in protic solvents because the ground-state energy of the nucleophile is lowered by solvation. SN1 reactions are favored in protic solvents because the transition-state energy leading to carbocation intermediate is lowered by solvation.
A Summary of SN1 Reaction Characteristics
The effects on SN1 reactions of the four variables—substrate, leaving group, nucleophile, and solvent—are summarized in the following statements:

Strategy
Look at the substrate, leaving group, nucleophile, and solvent. Then decide from the summaries at the ends of Section 11.3 and Section 11.5 whether an SN1 or an SN2 reaction is favored. SN1 reactions are favored by tertiary, allylic, or benzylic substrates, by good leaving groups, by nonbasic nucleophiles, and by protic solvents. SN2 reactions are favored by primary substrates, by good leaving groups, by good nucleophiles, and by polar aprotic solvents.
Solution
- (a) This is likely to be an SN1 reaction because the substrate is secondary and benzylic, the nucleophile is weakly basic, and the solvent is protic.
- (b) This is likely to be an SN2 reaction because the substrate is primary, the nucleophile is a good one, and the solvent is polar aprotic.
Problem 11-13 Predict whether each of the following substitution reactions is likely to be SN1 or SN2: (a)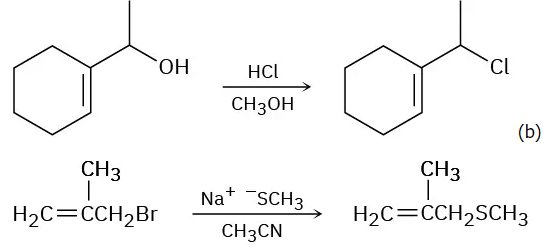
FAQs on The SN1 Reaction & Characteristics of the SN1 Reaction - Chemistry Optional Notes for UPSC
| 1. What is the SN1 reaction? |  |
| 2. What are the characteristics of the SN1 reaction? |  |
| 3. What is a leaving group in the SN1 reaction? |  |
| 4. What is a nucleophile in the SN1 reaction? |  |
| 5. How does the solvent affect the SN1 reaction? |  |














