Carbonyl Groups: Physical and Chemical Properties | Chemistry for JEE Main & Advanced PDF Download
Physical Properties of Carbonyl Compounds
Boiling Point: The boiling points of methanol and ethanal are -19°C and +21°C, respectively. Ethanal's boiling point is close to room temperature. Generally, aldehydes and ketones' boiling points increase with higher molecular weight due to stronger intermolecular forces.
- Van der Waals Dispersion Forces: As aldehyde and ketone molecules become longer with more electrons, van der Waals forces strengthen, leading to higher boiling points with an increase in carbon atoms.
- Van der Waals Dipole-Dipole Attraction: Aldehydes and ketones are polar due to the carbon-oxygen double bond. This polarity results in dipole-dipole attractions, contributing to higher boiling points compared to similar-sized hydrocarbons.
Solubility: Aldehydes and ketones are soluble in water, but solubility decreases with longer carbon chains. Small-sized ones like methanal, ethanal, and propanone are highly miscible with water in various proportions.
Aldehydes and ketones don't make hydrogen bonds among themselves, but they can bond with water molecules. This is why aldehydes and ketones dissolve well in water.
Since aldehydes and ketones both possess the carbonyl functional group, they undergo similar chemical reactions.
Nucleophilic Addition Reaction
Carbonyl groups exhibit a carbon-oxygen double bond (C=O) and are highly reactive toward nucleophiles due to the electrophilic nature of the carbon atom in the carbonyl group.
Mechanism:
- In simple terms, when a nucleophile attacks a carbonyl group, it goes for the carbon atom in the carbonyl group, which becomes more positive (electrophilic) due to the presence of oxygen.
- This attack happens in a way that is kind of sideways to the carbon-oxygen double bond.
- As a result, the carbon atom changes its structure a bit, and an intermediate structure is formed, which has a carbon atom bonded to four things (we call this sp3 hybridization). This intermediate then grabs a proton from the surrounding environment, and the end product is a neutral substance.
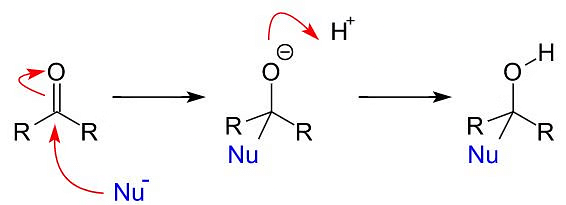 Nucleophilic Attack on Carbonyl Carbon
Nucleophilic Attack on Carbonyl Carbon
Reactivity:
- In simpler terms, aldehydes are usually more reactive than ketones in nucleophilic addition reactions. This is because of both steric and electronic reasons.
- Sterically, ketones have two large groups attached, making it harder for a nucleophile to get close to the carbon in the carbonyl group compared to aldehydes, which have only one such hindering group.
- Electronically, aldehydes are more reactive because two alkyl groups in ketones decrease the reactivity of the carbon in the carbonyl group more than in aldehydes, where there's only one such alkyl group.
Examples of Nucleophilic Addition Reactions:
- Grignard Reaction: Formation of a magnesium alkoxide, followed by attack of the nucleophilic carbon on the carbonyl group.
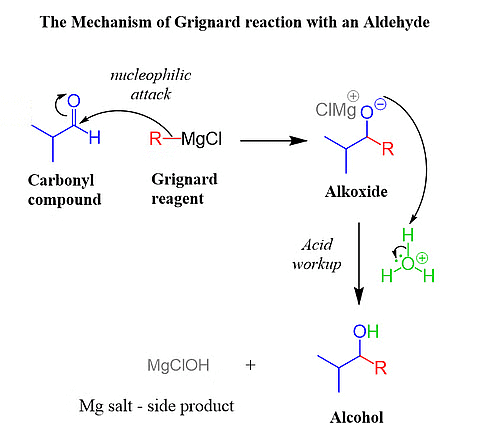 Grignard's Reagent Reaction with Carbonyl Groups
Grignard's Reagent Reaction with Carbonyl Groups
- Hydrogen Cyanide Addition: Nucleophilic attack of cyanide ion on the carbonyl carbon, followed by protonation.
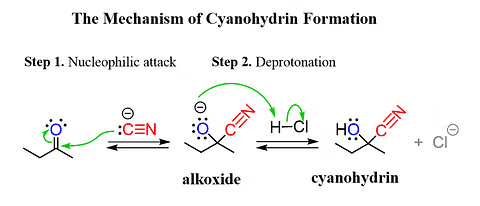 Addition of Hydrogen Cyanide to Carbonyl Group
Addition of Hydrogen Cyanide to Carbonyl Group
- Addition of sodium hydrogensulphite:
- The addition of sodium hydrogensulfite (NaHSO3) to carbonyl groups is a chemical reaction that involves the formation of crystalline derivatives known as bisulfite addition compounds.
- The carbonyl compound reacts with NaHSO3 to form a bisulfite addition compound, which is a reversible and generally a stereospecific process.
- This reaction is often employed in the purification and isolation of carbonyl-containing compounds, as the resulting bisulfite addition compounds are usually highly soluble in water, facilitating their separation from other reaction products.
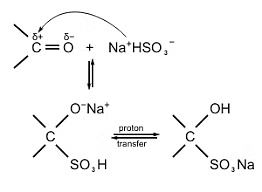 Addition of NaHSO3 to Carbonyl Group
Addition of NaHSO3 to Carbonyl Group
- Addition of alcohols:
- The addition of alcohol to carbonyl compounds is a common chemical reaction known as alcohol addition or acetal formation.
- In this process, alcohol reacts with a carbonyl compound, such as an aldehyde or ketone, to form a hemiacetal or acetal, respectively.
- The reaction involves the nucleophilic attack of the alcohol oxygen on the electrophilic carbon of the carbonyl group. This results in the formation of a new carbon-oxygen bond, leading to the conversion of the carbonyl compound into a hemiacetal or acetal. The reaction is often catalyzed by an acid, which helps facilitate the nucleophilic attack and subsequent proton transfer steps.
- The resulting hemiacetals and acetals are stable compounds and can be easily reversed under acidic conditions to regenerate the original carbonyl compound and alcohol.
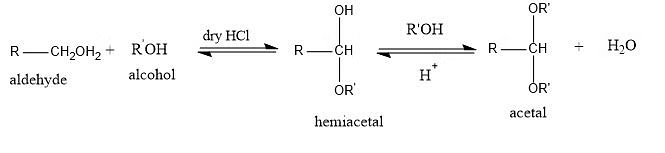 Addition of Alcohols to Carbonyl Groups
Addition of Alcohols to Carbonyl Groups
- Addition of ammonia and its derivatives:
- Compounds like ammonia and its derivatives, represented as H2N-Z, can attach to the carbonyl group of aldehydes and ketones.
- This reaction is reversible and gets a boost from an acidic catalyst. The formation of the product is favored in the equilibrium because the intermediate quickly loses water to create >C=N-Z.
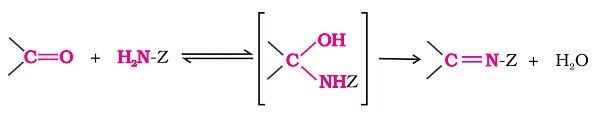 Addition of Ammonia and its Derivatives
Addition of Ammonia and its Derivatives
Z = Alkyl, aryl, OH, NH2, C6H5NH, NHCONH2, etc.
Note:
NH3 gives imine as product with carbonyl compound.
1o amines give N-alkyl imine and 2o amines give enamine.
Imine Formation
Reduction Reactions of Aldehydes and Ketones
Reduction to alcohols
Sodium borohydride (NaBH4) or lithium aluminum hydride (LiAlH4), along with catalytic hydrogenation, can be used to transform aldehydes into primary alcohols and ketones into secondary alcohols through a reduction process.
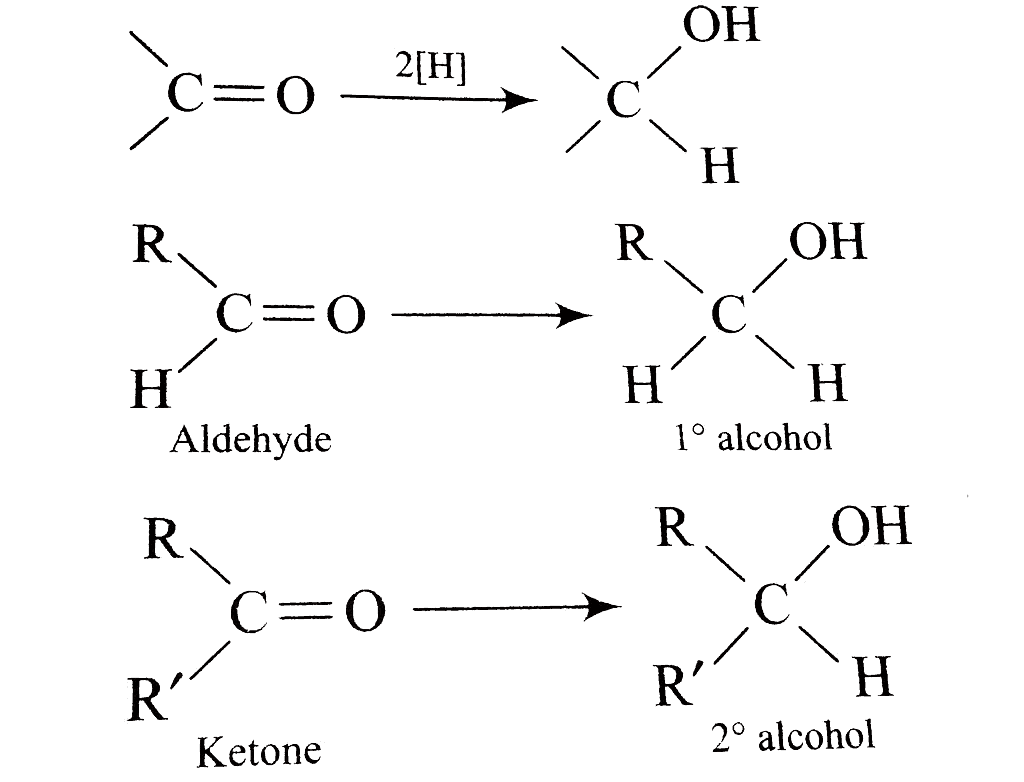 Reduction to Alcohols
Reduction to Alcohols
Reduction to Hydrocarbons
1. Clemmensen Reduction: The treatment of aldehydes and ketones with zinc amalgam and concentrated hydrochloric acid, known as the Clemmensen reduction, results in the reduction of the carbonyl group to a CH2 group.
 Clemmensen Reduction
Clemmensen Reduction
2. Wolf-Kishner Reduction: Alternatively, the reduction can be achieved by using hydrazine followed by heating with sodium or potassium hydroxide in a high-boiling solvent like ethylene glycol, a process known as Wolff-Kishner reduction. In this method, the carbonyl group is converted into a CH2 group.
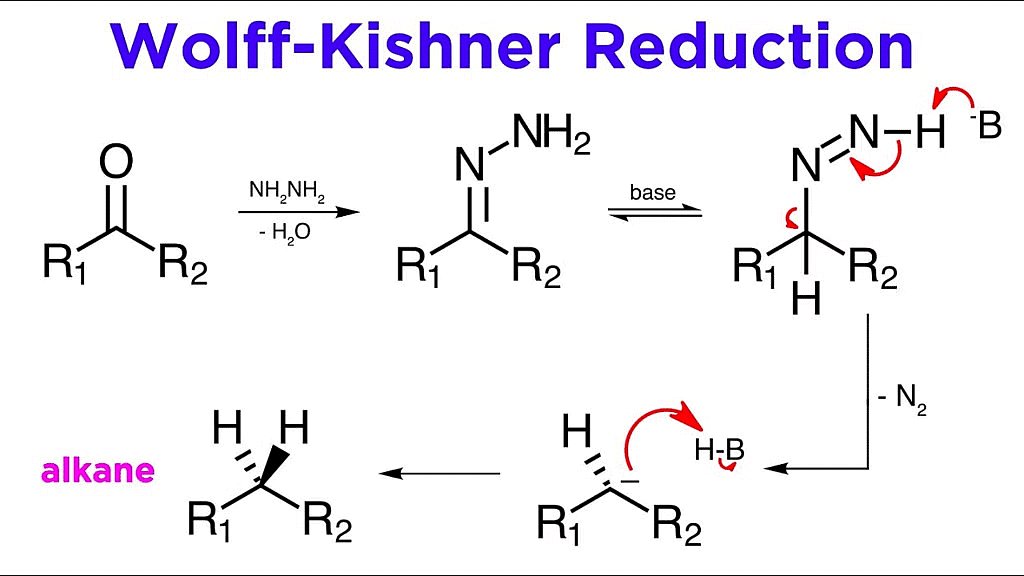 Wolff-Kishner Reduction
Wolff-Kishner Reduction
Oxidation Reactions of Aldehydes and Ketones
Aldehydes and ketones show differences in how they react to oxidation. Aldehydes are easily turned into carboxylic acids when treated with common oxidizing agents like nitric acid, potassium permanganate, or potassium dichromate. Even milder agents like Tollens' reagent and Fehlings' reagent can oxidize aldehydes.
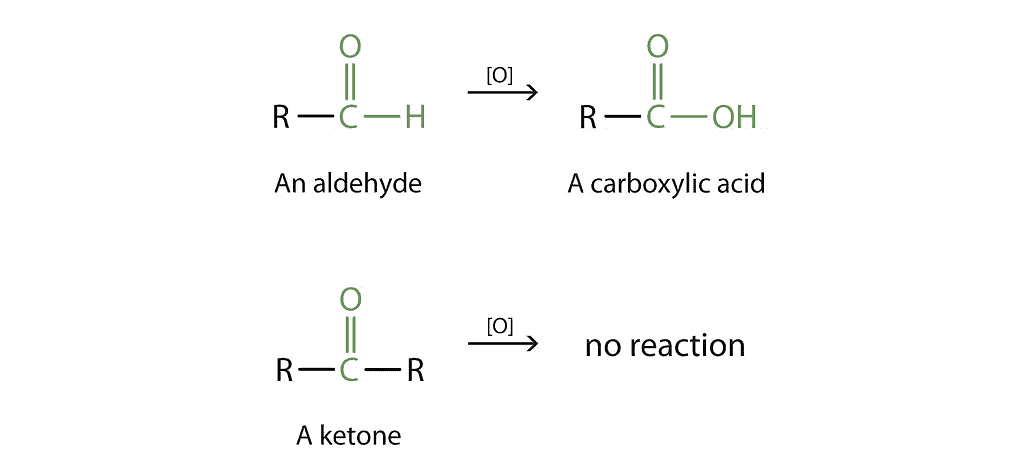 Oxidation of Carbonyl Groups
Oxidation of Carbonyl Groups
On the other hand, ketones need more intense conditions for oxidation. This involves strong oxidizing agents and higher temperatures. When ketones undergo oxidation, the carbon-carbon bonds are broken, leading to a mix of carboxylic acids with fewer carbon atoms than the original ketone.
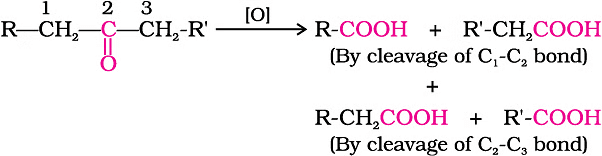 Oxidation of Ketones
Oxidation of Ketones
These gentle oxidizing agents listed below are employed to differentiate between aldehydes and ketones.
(i) Tollens' Test:
- When an aldehyde is warmed with freshly prepared ammoniacal silver nitrate solution (Tollens' reagent), it forms a shiny silver mirror.
- This occurs because aldehydes get oxidized to form carboxylate anions, and the reaction takes place in an alkaline environment.
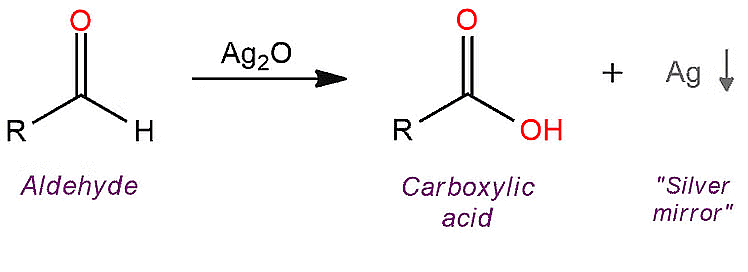 Tollen's Reagent Test
Tollen's Reagent Test
(ii) Fehling's Test:
- Fehling's reagent consists of two solutions—Fehling solution A (copper sulfate) and Fehling solution B (alkaline sodium potassium tartarate). These solutions are mixed before the test.
- When an aldehyde is heated with Fehling's reagent, a reddish-brown precipitate is observed. Aldehydes undergo oxidation to form corresponding carboxylate anions. However, aromatic aldehydes do not exhibit this reaction.
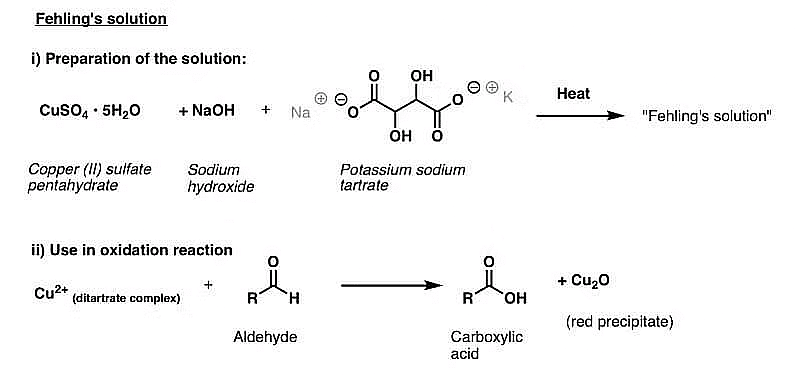 Fehling's Test
Fehling's Test
(iii) Haloform Reaction:
- Methyl ketones, which are aldehydes or ketones containing a methyl group linked to the carbonyl carbon, undergo oxidation through the haloform reaction.
- When treated with sodium hypohalite, these compounds are transformed into sodium salts of corresponding carboxylic acids.
- The resulting carboxylic acids have one carbon atom less than the original carbonyl compound, and the methyl group is converted into a haloform. Importantly, this oxidation doesn't impact a carbon-carbon double bond, if one is present in the molecule.
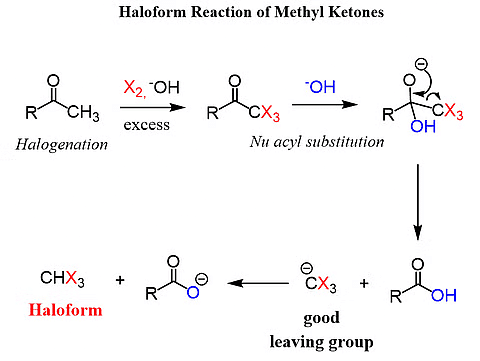 Haloform Reaction
Haloform Reaction
Additionally, the iodoform reaction using sodium hypoiodite can be applied to detect the presence of CH3CO or CH3CH(OH) groups, which are converted to CH3CO groups upon oxidation.
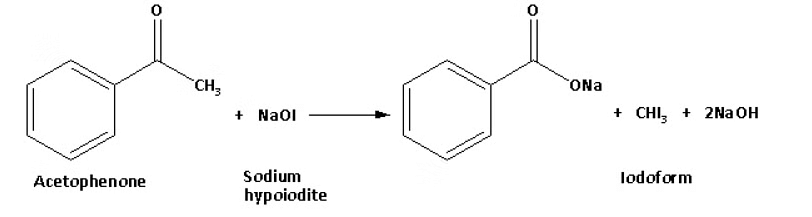 Example of Iodoform Reaction
Example of Iodoform Reaction
Reactions due to α-hydrogen
- To understand what alpha hydrogen means, we should start with understanding alpha carbon. The alpha carbon is the first carbon linked to the functional group. For aldehydes and ketones, the functional group is the carbonyl group, and it's this group that leads to the formation of alpha hydrogen.
- The hydrogen attached to the alpha-carbon is known as alpha-hydrogen. It's a bit acidic because of the carbonyl group and the way it's stabilized through resonance.
- The acidity of a-hydrogen atoms of carbonyl compounds is due to the strong electron-withdrawing effect of the carbonyl group and resonance stabilization of the conjugate base.
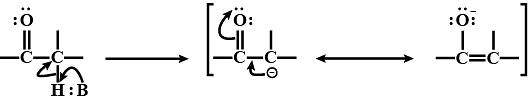 Resonance Structures of Carbonyl Group
Resonance Structures of Carbonyl Group
Aldol Condensation
- Aldehydes and ketones with at least one α-hydrogen can react in the presence of a weak alkali catalyst to produce β-hydroxy aldehydes (called aldol) or β-hydroxy ketones (called ketol). This reaction is known as the Aldol reaction, named after the two functional groups, aldehyde and alcohol, present in the resulting products.
- The aldol and ketol compounds can easily lose water, forming α, β-unsaturated carbonyl compounds in a process called Aldol condensation. Even though ketones produce ketols, we still use the term "aldol condensation" because of their similarity to aldehydes in this reaction.
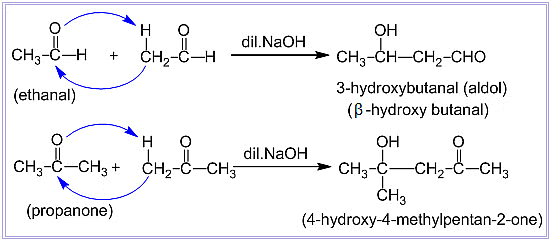 Examples of Aldol Condensation
Examples of Aldol Condensation
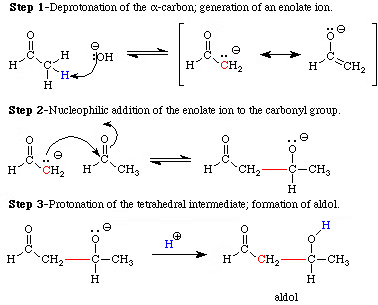 Reaction Mechanism of Aldol Condensation
Reaction Mechanism of Aldol Condensation
Cross Aldol Condensation
When aldol condensation involves two different aldehydes and/or ketones, it is referred to as cross aldol condensation. If both compounds have alpha-hydrogen atoms, the reaction yields a mixture of four products. This can be demonstrated through the aldol reaction of a mix of ethanal and propanal.
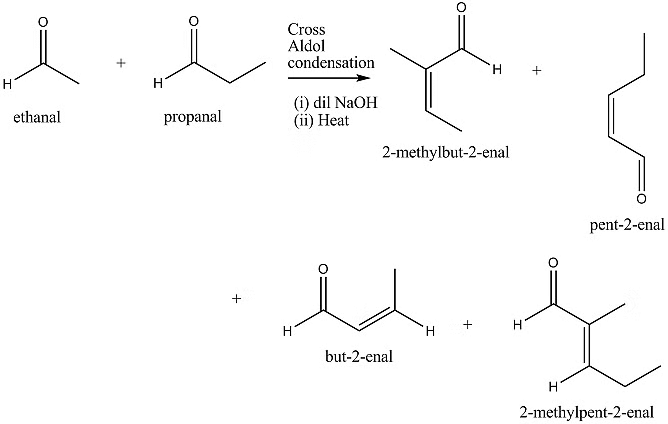 Cross Aldol Condensation
Cross Aldol Condensation
Other Reactions
Cannizzaro Reaction
Aldehydes without an alpha-hydrogen undergo a reaction called self-oxidation and reduction (disproportionation) when heated with concentrated alkali. In this process, one molecule of the aldehyde gets reduced to form alcohol, while another molecule is oxidized to produce the salt of a carboxylic acid.
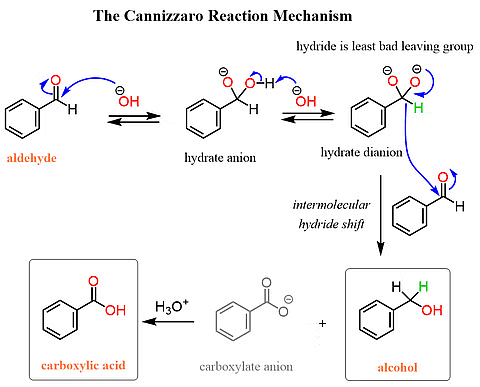 Cannizzaro ReactionElectrophilic Substitution Reactions
Cannizzaro ReactionElectrophilic Substitution Reactions
Aromatic aldehydes and ketones experience electrophilic substitution on the ring, where the carbonyl group serves as a deactivating and meta-directing group.
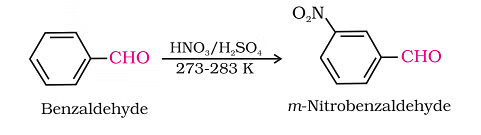 Electrophilic Substitution Reaction
Electrophilic Substitution Reaction
|
361 videos|822 docs|301 tests
|
FAQs on Carbonyl Groups: Physical and Chemical Properties - Chemistry for JEE Main & Advanced
| 1. What are the physical properties of carbonyl compounds? |  |
| 2. What is a nucleophilic addition reaction of carbonyl compounds? |  |
| 3. How do aldehydes and ketones undergo reduction reactions? |  |
| 4. What are the oxidation reactions of aldehydes and ketones? |  |
| 5. What are the reactions due to α-hydrogen in carbonyl compounds? |  |





















