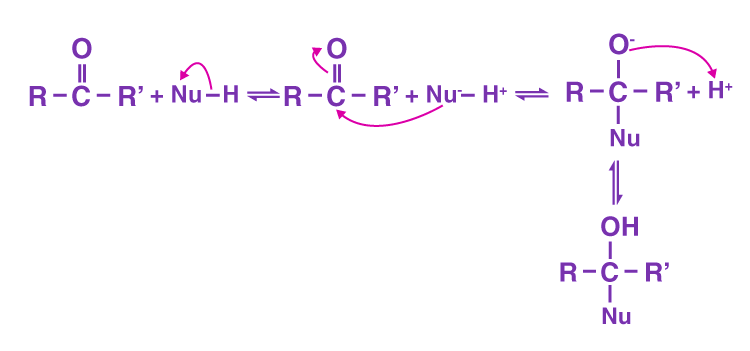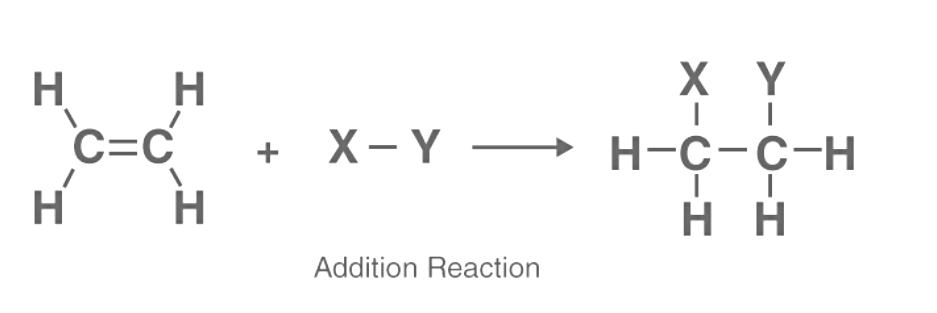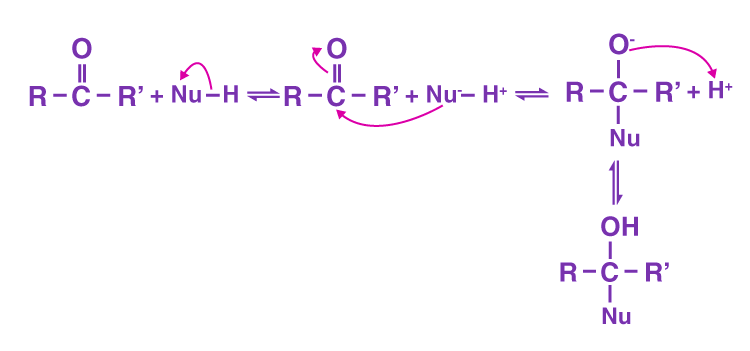Put plainly in the realm of organic chemistry, an addition reaction denotes a chemical process where two or more reactants combine to produce a singular, larger product.
Addition Reaction Explanation
However, only chemical compounds with multiple bond characteristics can engage in an addition reaction, typically involving the breaking of a double or triple bond to form requisite single bonds. Essentially, an addition reaction operates in reverse compared to a decomposition reaction, where a compound disintegrates into one or more elements or compounds. Examining an instance of an addition reaction, consider the hydrochlorination of propane (an alkene), illustrated by the following equation:
CH3CH = CH2 + HCl → CH3C+HCH3 + Cl- → CH3CHClCH3
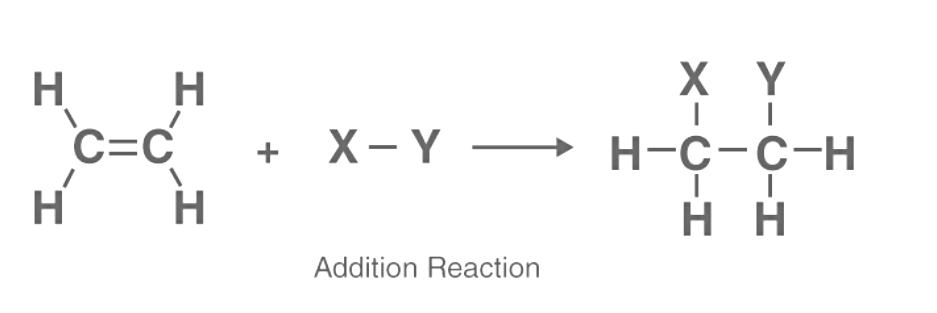
Types of Addition Reactions
- For polar addition reactions there are two classifications, namely:
- Electrophilic Addition reactions
- Nucleophilic Addition reactions
- For non-polar addition reactions, we have two classifications, namely:
- Free radical addition reactions
- Cycloadditions reactions
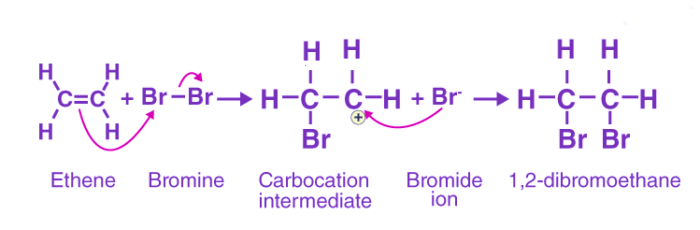
Electrophilic addition
An electrophilic addition reaction can be described as an addition reaction in which a reactant with multiple bonds as in a double or triple bond undergoes its π bond broken and two new σ bonds are formed.
Nucleophilic Addition
A nucleophilic addition reaction is an addition reaction where a chemical compound with an electron-deficient or electrophilic double or triple bond, a π bond, reacts with a nucleophile which is an electron-rich reactant with the disappearance of the double bond and creation of two new single, or σ, bonds.