IGCSE Year 10 > Year 10 Notes > Chemistry for GCSE/ > Preparation of Soluble Salts
Preparation of Soluble Salts
Preparing Soluble Salts
Salts
- A salt is a compound that is formed when the hydrogen atom in an acid is replaced by a metal
- For example, if we replace the hydrogen atom in Hydrochloric Acid (HCl) with a potassium atom, then the salt potassium chloride (KCl) is formed.
- Salts play a crucial role in various applications such as fertilizers, batteries, cleaning products, healthcare items, and fungicides.
Naming salts
- Salts are compounds produced by replacing the hydrogen atom in an acid with a metal.
- When the hydrogen in an acid like Hydrochloric Acid (HCl) is substituted with a metal such as potassium, a salt like potassium chloride (KCl) is created.
- The significance of salts lies in their diverse applications spanning fertilizers, batteries, cleaning agents, medicinal products, and fungicides.
- The name of a salt comprises two parts:
- The first part is derived from the metal, metal oxide, or metal carbonate involved in the reaction.
- The second part originates from the acid used in the reaction.
- The name of a salt is determined by the reactants involved.
- For instance, when hydrochloric acid reacts, salts ending in chloride are produced, containing the chloride ion (Cl -).
- Examples:
- When sodium hydroxide reacts with hydrochloric acid, sodium chloride is produced.
- When zinc oxide reacts with sulfuric acid, zinc sulfate is produced.
Preparing Salts
- Certain salts are obtainable through mining, while others necessitate laboratory synthesis.
- The selection of method is contingent upon the solubility characteristics of the salt undergoing preparation.
MULTIPLE CHOICE QUESTIONTry yourself: Which of the following is true about salts?
Preparing Soluble Salts
Method A: adding acid to a solid metal, insoluble base or insoluble carbonate
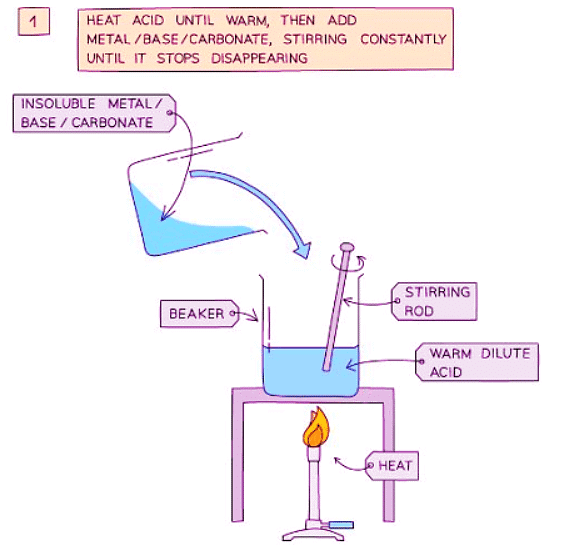
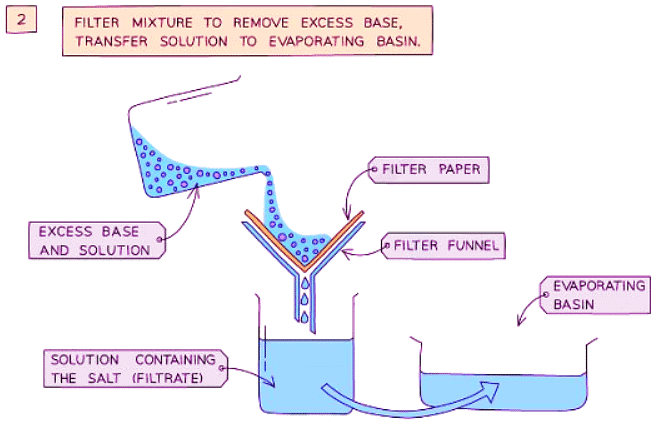
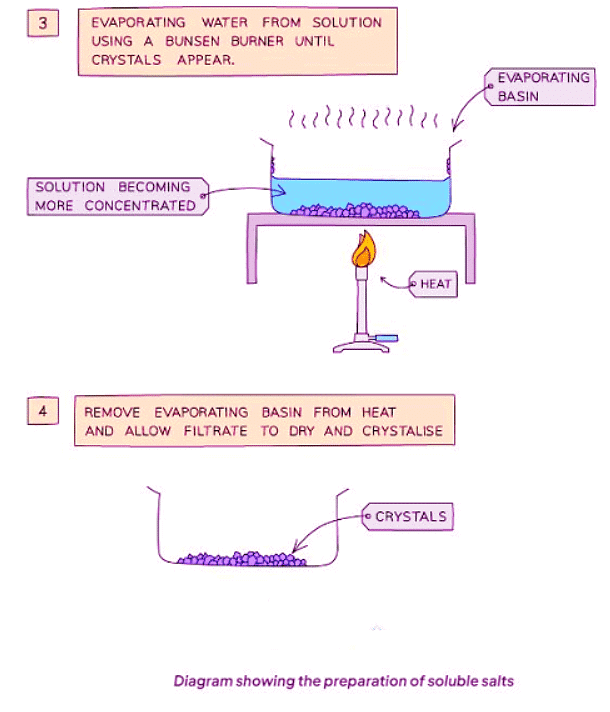
Method
- Add dilute acid into a beaker and heat using a Bunsen burner flame.
- Add the insoluble metal, base, or carbonate gradually to the warm dilute acid and stir until the base is in excess (i.e., until a suspension of the base forms in the acid).
- Filter the mixture into an evaporating basin to eliminate the excess base.
- Heat the solution to evaporate water and achieve saturation. Verify saturation by observing crystal formation on a cold, glass rod dipped into the solution.
- Allow the filtrate to dry and crystallize in a warm location.
- Remove excess solution and either air-dry the crystals or use filter paper to blot them dry.
Example: preparation of pure, hydrated copper(II) sulfate crystals using method A
Acid: Dilute sulfuric acid
Insoluble base: Copper(II) oxide
Method
- Add dilute sulfuric acid into a beaker and heat using a bunsen burner flame
- Add copper(II) oxide (insoluble base), a little at a time to the warm dilute sulfuric acid and stir until the copper (II) oxide is in excess (stops disappearing)
- Filter the mixture into an evaporating basin to remove the excess copper(II) oxide
- Leave the filtrate in a warm place to dry and crystallize
- Decant excess solution
- Blot crystals dry with filter paper
Equation of reaction: Copper(II) oxide + Sulfuric acid → Copper(II) sulphate + Water
CuO (s) + H2SO4 (aq) → CuSO4 (aq) + H2O (l)
Method B: Reacting a Dilute Acid and Alkali (Soluble Base)
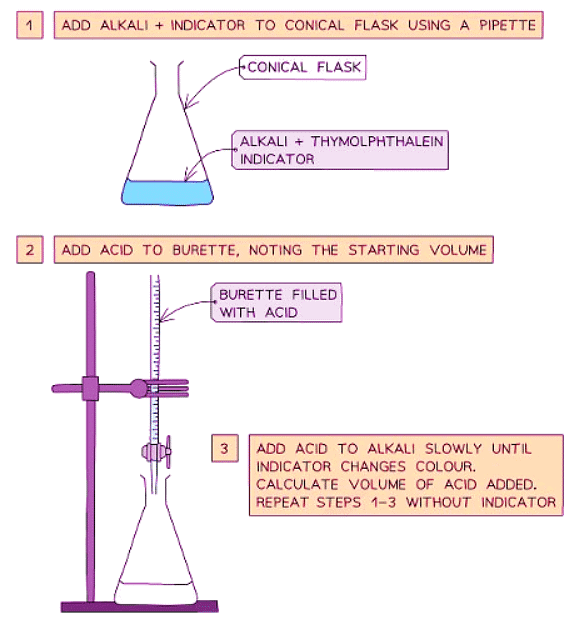
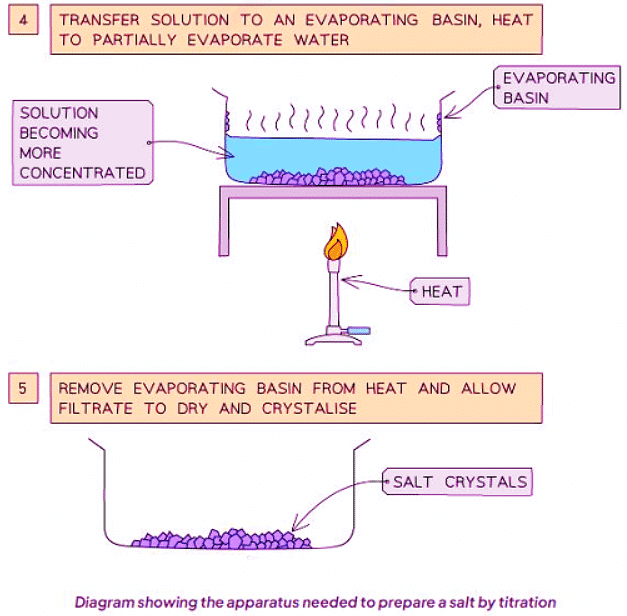
Method
- Use a pipette to measure the alkali into a conical flask and add a few drops of indicator (such as thymolphthalein or methyl orange).
- Add the acid into the burette.
- Record the starting volume of acid in the burette.
- Add the acid slowly from the burette to the conical flask until the indicator changes color appropriately.
- Record the final volume of acid in the burette.
- Calculate the volume of acid added by subtracting the initial volume from the final volume.
After determining the volume of acid needed, follow these steps:
- Add the calculated volume of acid to the same volume of alkali (without the indicator).
- Heat the resulting solution in an evaporating basin to partially evaporate, leaving a saturated solution (where crystals start to form).
- Allow the solution to cool and crystals to form, then decant the excess solution to obtain the crystals.
Steps for Preparing Soluble Salts
- Calculate the final volume of acid by subtracting the initial volume of acid from it.
- Add the obtained volume of acid to an equivalent volume of alkali without adding any indicator.
- Heat the resulting solution in an evaporating basin until partial evaporation occurs, leaving a saturated solution (visible by crystals forming on the basin's sides or on a glass rod).
- Allow the solution to cool and crystallize, then carefully decant the excess liquid and let the crystals dry.
MULTIPLE CHOICE QUESTIONTry yourself: What is the purpose of heating the solution in Method A when preparing soluble salts?
The document Preparation of Soluble Salts is a part of the Year 10 Course Chemistry for GCSE/IGCSE.
All you need of Year 10 at this link: Year 10
FAQs on Preparation of Soluble Salts
| 1. How can soluble salts be prepared? |  |
Ans. Soluble salts can be prepared by reacting an acid with a base, a metal, or a carbonate.
| 2. What are some common methods for preparing soluble salts? |  |
Ans. Some common methods for preparing soluble salts include precipitation, neutralization, and direct combination of elements.
| 3. Can soluble salts be prepared by mixing two soluble salts together? |  |
Ans. Yes, soluble salts can be prepared by mixing two soluble salts together through double displacement reactions.
| 4. Why is it important to ensure that the salt formed is soluble in the preparation of soluble salts? |  |
Ans. It is important to ensure that the salt formed is soluble to avoid any insoluble precipitates from forming, which could affect the purity of the final product.
| 5. What are some factors to consider when preparing soluble salts? |  |
Ans. Factors to consider when preparing soluble salts include the reactivity of the reactants, the solubility of the salt formed, and the method of preparation chosen.
Related Searches
Preparation of Soluble Salts, shortcuts and tricks, Semester Notes, Objective type Questions, Exam, ppt, Sample Paper, video lectures, past year papers, pdf , Summary, practice quizzes, Preparation of Soluble Salts, study material, mock tests for examination, Important questions, Viva Questions, Preparation of Soluble Salts, Previous Year Questions with Solutions, MCQs, Extra Questions, Free;





