IGCSE Year 10 > Year 10 Notes > Chemistry for GCSE/ > Group I Properties
Group I Properties
The Group I metals
- The Group I metals are also called the alkali metals as they form alkaline solutions with high pH values when reacted with water
- Group I metals are lithium, sodium, potassium, rubidium, caesium, and francium
- They all contain just one electron in their outer shell
Physical properties of the Group I metals
- Group I metals exhibit softness and are easily cut, with their softness and density increasing further down the group (though exceptions like sodium and potassium exist regarding density).
- Freshly cut surfaces of Group I metals display shiny, silvery appearances.
- They possess the ability to conduct both heat and electricity.
- Group I metals generally have low melting points and densities compared to other metals, with their melting points decreasing as one moves down the group; some may even melt under hot conditions.
MULTIPLE CHOICE QUESTIONTry yourself: Which property of Group I metals increases as one moves down the group?
The alkali metals lie on the far left-hand side of the Periodic Table:
Chemical properties of the Group I metals
- Group I metals exhibit high reactivity with both oxygen and water vapor in the air, necessitating their storage under oil to prevent reactions.
- When exposed to water, Group I metals undergo vigorous reactions, yielding alkaline metal hydroxide solutions along with hydrogen gas.
- The reactivity of Group I metals increases progressively as one moves down the group, prompting restrictions in schools where only the first three metals are permitted for demonstrations.
Reactions of the Group I metals with water:
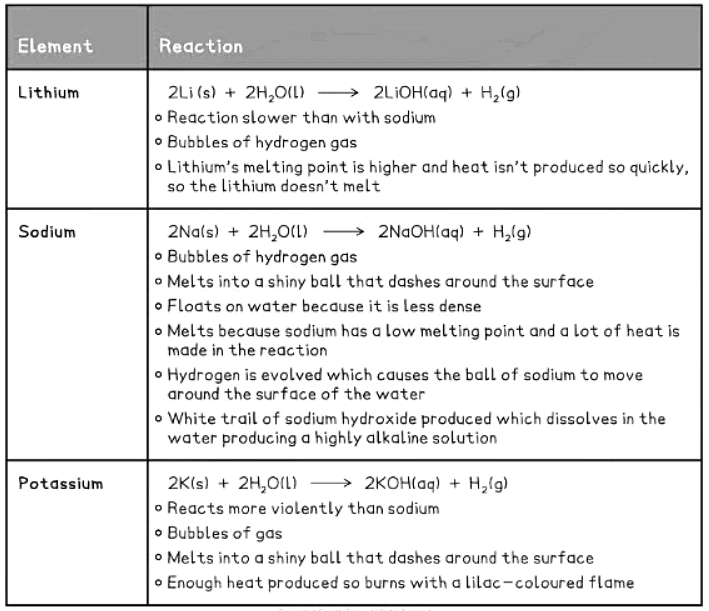
Predicting the Properties of Group I Elements
- Understanding how the top group I elements react helps in anticipating the behavior of elements lower in the Group I.
Properties of other Alkali Metals (Rubidium, Caesium, and Francium)
- As we move down the alkali metal group, including rubidium, caesium, and francium, their reactivity intensifies compared to lithium, sodium, and potassium.
- Among these metals, lithium is the least reactive at the top, while francium, found at the bottom, is exceptionally reactive.
- Predictions involving francium can be challenging due to its scarcity and radioactivity.
- For instance, when considering reactions with water:
- Predicting the Reaction with Water:

- You can also observe other properties such as boiling point, melting point, and density of Group I elements to predict whether other properties are likely to be larger or smaller descending down the group.
- Consider examining boiling point, melting point, and density of Group I elements to anticipate the variations in other properties as you move down the group.
MULTIPLE CHOICE QUESTIONTry yourself: Which of the following statements is true about the reactivity of Group I metals?
The document Group I Properties is a part of the Year 10 Course Chemistry for GCSE/IGCSE.
All you need of Year 10 at this link: Year 10
FAQs on Group I Properties
| 1. What are the Group I metals? |  |
Ans. Group I metals are the elements located in the first column of the periodic table, including lithium, sodium, potassium, rubidium, cesium, and francium.
| 2. What are the common properties of Group I metals? |  |
Ans. Common properties of Group I metals include low melting and boiling points, high reactivity with water, forming alkaline solutions, and having one electron in their outermost shell.
| 3. How do Group I metals react with water? |  |
Ans. Group I metals react vigorously with water to form metal hydroxides and hydrogen gas. The reactivity increases down the group from lithium to francium.
| 4. Why are Group I metals stored under oil? |  |
Ans. Group I metals are stored under oil to prevent them from reacting with moisture and oxygen in the air, which could lead to corrosion and degradation of the metal.
| 5. What are some practical applications of Group I metals? |  |
Ans. Group I metals have various applications, including in the manufacturing of batteries, fireworks, streetlights, and pharmaceuticals. Sodium and potassium compounds are also used in fertilizers and food preservation.
Related Searches
Viva Questions, mock tests for examination, Group I Properties, shortcuts and tricks, past year papers, MCQs, Sample Paper, study material, Objective type Questions, ppt, Free, Extra Questions, practice quizzes, Semester Notes, Summary, Important questions, pdf , Group I Properties, Previous Year Questions with Solutions, Group I Properties, Exam, video lectures;





