IGCSE Class 10 > Class 10 Notes > Physics for GCSE/ > Gases & Absolute Temperature
Gases & Absolute Temperature
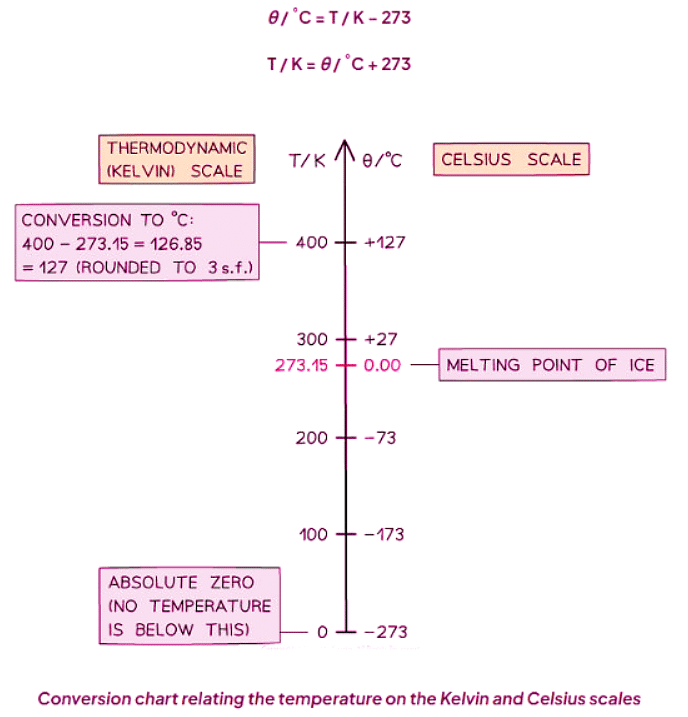
Absolute Temperature
- The Kelvin temperature scale commences at absolute zero.
- At this point, 0 K corresponds to -273 °C.
- A rise of 1 K equates to the same alteration as a rise of 1 °C.
- Temperatures below 0 K are unattainable, signifying that Kelvin temperatures cannot be negative.
- To convert between Celsius (θ) and Kelvin (T) scales, utilize the subsequent conversion:
The Gas Laws
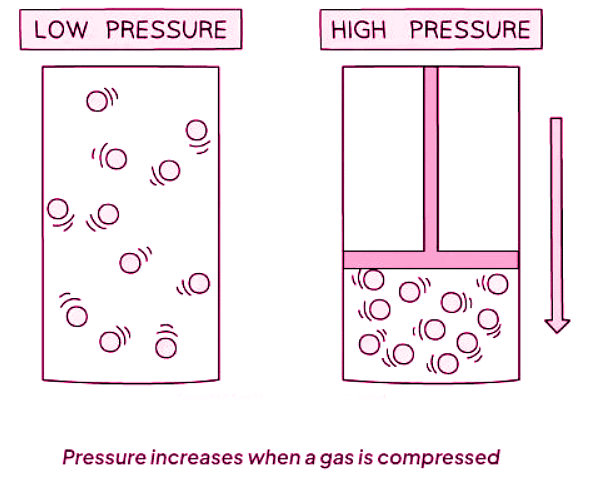
If the temperature of a gas remains constant, changes in pressure occur when the gas is compressed or expanded:
- Compressed: Decreasing the volume increases the pressure.
- Expanded: Increasing the volume decreases the pressure.
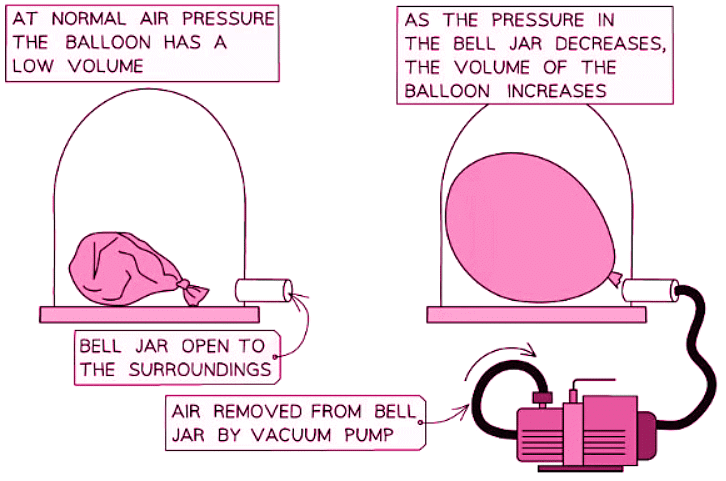
- Likewise, alterations in pressure can induce variations in volume.
- A vacuum pump has the capability to extract air from a sealed enclosure.
- The illustration below illustrates how the volume of a inflated balloon alters when the surrounding air pressure diminishes:
- When gas is compressed, its molecules will collide with the container walls more often.
- This results in a greater total force exerted on the walls, consequently raising the pressure.
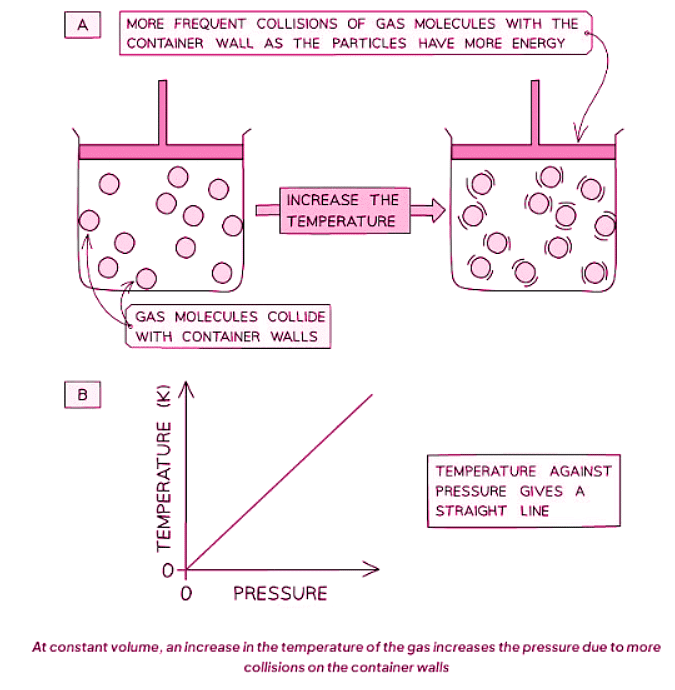
Pressure & Temperature (Constant Volume)
- The velocity of molecules rises with temperature escalation, and diminishes when temperature decreases.
- Given that average kinetic energy is contingent on velocity, an increase in temperature, while keeping volume constant, also augments the kinetic energy of molecules.
- Consequently, hotter gases exhibit higher average kinetic energy, whereas cooler ones manifest lower average kinetic energy.
- As gas heats up, its molecules accelerate, leading to more frequent collisions with container walls and consequent pressure escalation.
- Hence, at a constant volume, heightened temperature augments gas pressure, and conversely.
- Diagram A illustrates how molecules in a fixed volume collide more frequently with container walls as temperature rises.
- Diagram B portrays a linear graph since temperature and pressure are directly proportional.
MULTIPLE CHOICE QUESTIONTry yourself: What is the relationship between temperature and pressure in a gas at constant volume?
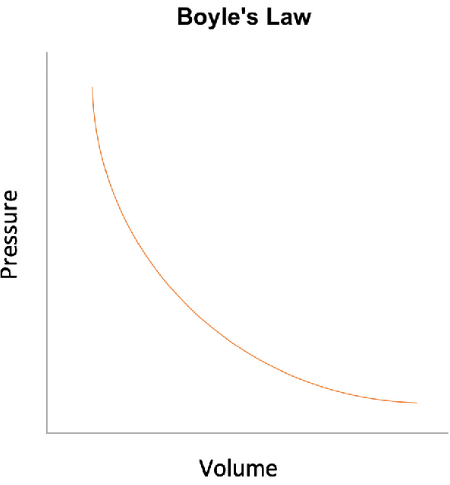
Boyle's Law
- Boyle's Law describes the relationship between the pressure and volume of an ideal gas when the temperature remains constant.
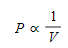 This means that as the pressure on a gas increases, its volume decreases, and vice versa.
This means that as the pressure on a gas increases, its volume decreases, and vice versa. - The relationship between pressure and volume for a fixed mass of gas at a constant temperature can be expressed as:
P1V1 = P2V2 - Here,
- P1 = initial pressure (Pa)
- P2 = final pressure (Pa)
- V1 = initial volume (m3)
- V2 = final volume (m3)
- It's important to note that the units of volume are typically in cubic meters (m3) and pressure in Pascals (Pa).
- Calculations involving different units, such as cm3 or MPa, can be directly converted as long as the final answer is given in the original units.
The document Gases & Absolute Temperature is a part of the Class 10 Course Physics for GCSE/IGCSE.
All you need of Class 10 at this link: Class 10
FAQs on Gases & Absolute Temperature
| 1. What is absolute temperature and how is it different from Celsius temperature? |  |
Ans. Absolute temperature is measured in Kelvin (K) and starts from absolute zero, where all molecular motion ceases. Celsius temperature, on the other hand, is measured in degrees Celsius (°C) and is based on the freezing and boiling points of water.
| 2. How do you convert Kelvin to Celsius temperature? |  |
Ans. To convert Kelvin to Celsius, you subtract 273 from the Kelvin temperature. For example, if the temperature is 300K, the Celsius equivalent would be 27°C (300K - 273 = 27°C).
| 3. How are gas volume and pressure related according to the gas laws? |  |
Ans. According to Boyle's Law, the volume of a gas is inversely proportional to its pressure when the temperature is constant. This means that as pressure increases, volume decreases, and vice versa.
| 4. Can you explain the relationship between temperature and pressure in gases? |  |
Ans. According to Charles's Law, the volume of a gas is directly proportional to its temperature when the pressure is constant. This means that as temperature increases, volume increases, and vice versa.
| 5. How does understanding gas behavior help in real-life applications? |  |
Ans. Understanding gas behavior is crucial in various industries such as manufacturing, healthcare, and transportation. It helps in gas storage, pressure regulation, and ensuring safety measures are in place.
Related Searches
Exam, Important questions, Sample Paper, ppt, past year papers, Summary, practice quizzes, Viva Questions, video lectures, Semester Notes, study material, MCQs, Objective type Questions, Free, shortcuts and tricks, Previous Year Questions with Solutions, Extra Questions, Gases & Absolute Temperature, pdf , Gases & Absolute Temperature, Gases & Absolute Temperature, mock tests for examination;





