CBSE Class 8 > Class 8 Notes > Year 8 Chemistry (Cambridge) > Solutions and solubility
Solutions and solubility
Introduction to Solutions
- Solutions are homogeneous mixtures composed of a solvent and one or more solutes.
- The solvent is the substance in which the solute dissolves, forming a uniform mixture.

Concentration of Solutions
- Molarity (M): Molarity is defined as the number of moles of solute per liter of solution (mol/L).
- Example: A solution with 0.5 M concentration means there are 0.5 moles of solute in 1 liter of solution.
MULTIPLE CHOICE QUESTIONTry yourself: What is the definition of molarity?
Factors Affecting Solubility
- Temperature: Generally, solubility increases with temperature for solids and decreases for gases.
- Example: Sugar dissolves faster in hot tea compared to cold tea.
- Pressure: Affects mainly gases; higher pressure increases solubility.
- Example: Carbon dioxide is more soluble in soda under pressure.
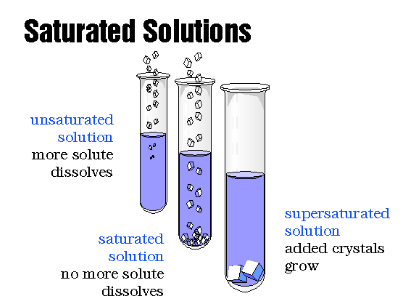
Solubility and Saturated Solutions
- Saturated Solution: Contains the maximum amount of solute that can dissolve at a given temperature.
- Example: A solution of sugar in water where no more sugar can dissolve at a specific temperature.
- Supersaturated Solution: Holds more solute than normally possible at a given temperature, achieved by careful cooling of a saturated solution.
- Example: Creating a supersaturated sugar solution by heating and then slowly cooling it.
Solubility Units
- Molality (m): Molality is the number of moles of solute per kilogram of solvent.
- Example: 1 molal (1 m) solution has 1 mole of solute dissolved in 1 kilogram of solvent.
MULTIPLE CHOICE QUESTIONTry yourself: What is a saturated solution?
Factors Affecting Dissolving Rate
- Stirring: Increases the rate by bringing fresh solvent into contact with the solute.
- Temperature: Generally, higher temperatures increase the rate of dissolution for solids in liquids.
- Particle Size: Smaller solute particles dissolve faster due to increased surface area.
Conclusion
Understanding solutions and solubility helps in various applications from chemistry labs to everyday life, such as cooking and cleaning.
The document Solutions and solubility is a part of the Class 8 Course Year 8 Chemistry (Cambridge).
All you need of Class 8 at this link: Class 8
FAQs on Solutions and solubility
| 1. What's the difference between a solution and a mixture in chemistry? |  |
Ans. A solution is a homogeneous mixture where a solute dissolves completely in a solvent, forming a single phase with uniform properties throughout. A mixture, however, can be heterogeneous, meaning its components remain visibly separate or don't fully dissolve. Solutions are transparent and stable, while mixtures may appear cloudy or separate over time.
| 2. How do you know if a substance is soluble or insoluble in water? |  |
Ans. Solubility refers to how much of a substance can dissolve in a solvent at a given temperature. Soluble substances like salt and sugar dissolve completely and disappear into the liquid, forming a clear solution. Insoluble substances like sand or chalk don't dissolve and remain as solid particles visible in the liquid, creating a cloudy or suspended appearance.
| 3. Why does temperature affect how much sugar dissolves in water? |  |
Ans. Increasing temperature provides more energy to solute particles, helping them break free from their crystal structure and disperse into the solvent more effectively. At higher temperatures, solubility increases for most solid solutes because the kinetic energy of particles rises. This is why hot tea dissolves sugar faster than cold water-heat accelerates the dissolution process and allows greater quantities to dissolve.
| 4. What happens to the mass when you dissolve salt in water? |  |
Ans. The total mass remains constant when salt dissolves in water-this follows the law of conservation of mass. The salt's mass doesn't disappear; it's simply distributed throughout the solution as dissolved ions invisible to the naked eye. The solution weighs the same as the combined mass of the salt and water added together.
| 5. How do you calculate the concentration of a solution for your chemistry exam? |  |
Ans. Concentration measures how much solute is dissolved in a given amount of solvent or solution. The most common method for Class 8 is using mass per volume: concentration = mass of solute (grams) ÷ volume of solution (litres). Students can also express it as a percentage by dividing solute mass by total solution mass and multiplying by 100 to compare solution strengths effectively.
Related Searches
Sample Paper, Important questions, Semester Notes, Solutions and solubility, Free, MCQs, practice quizzes, Solutions and solubility, Solutions and solubility, pdf , study material, Previous Year Questions with Solutions, Viva Questions, ppt, video lectures, Exam, shortcuts and tricks, Extra Questions, past year papers, Summary, mock tests for examination, Objective type Questions;






