Chapter Notes: Methods of Separation in Everyday Life
Separation of Substances
In our daily lives, we often need to separate useful substances from mixtures so that each part can be used in the best way. Below are simple ways to separate mixtures into their components.
- Mixture: A combination of two or more substances in which each substance retains its own properties. For example, air is a mixture of different gases. Water from a tap is usually a mixture because it contains dissolved salts and other substances.
- Pure substance: A material in which all particles are identical and have the same properties. For example, distilled water is a pure substance because it contains only water molecules.

We separate substances to obtain useful components, to remove impurities, and to prepare materials for cooking, medicine or industry.
For example, Ria and her brother Aryan visit their grandparents' farm. They notice their grandparents sorting vegetables. Curious, they ask why. Their Dadi explains, "We clean and sort them to make sure they're fresh for cooking." Ria and Aryan help wash and sort the vegetables, learning how to remove dirt and bad pieces before cooking. This shows how separation is useful in everyday life.
Different Methods of Separation

Handpicking is the simplest method of separation. It is used when unwanted material is present in small amounts and is clearly different in size, shape or colour from the useful material. The unwanted part is removed by hand.
Examples: removing stones, pebbles, husk or damaged grains from wheat, rice or pulses; taking whole black peppercorns out of a mixed dish by hand.
A good example of handpicking is when Malli separates whole black peppers from the vegetable pulao using his hands. This shows how handpicking is used to remove unwanted substances from a mixture.

2. Threshing
Threshing separates grain from stalks. Stalks are dried and then beaten so that seeds come loose.
- Manual threshing: Small bundles of stalks are beaten on a hard surface to release the grains.
- Threshing by animals: Stalks are spread around a fixed pole and animals like bullocks walk over them; the hooves help separate grains from stalks.
- Threshing machine: Machines powered by diesel engines or electricity are used to thresh grains quickly on farms and in larger operations.
Try yourself: Which method is used to separate grains from harvested stalks by beating them?
Threshing machines: How they work
- Threshing machines separate grains from stalks and remove some of the husk.
- Some machines perform both threshing and winnowing together, making the process faster.
- By using threshers, the process of separating grains becomes faster and more efficient.

3. Winnowing
Winnowing separates lighter particles from heavier ones with the help of wind or air.
This process separates lighter particles from heavier ones using wind. It is often used to separate grain from husk.
Farmers drop a mixture of wheat and husk from a height. The wind carries the husk away, forming a heap nearby, while the heavier wheat grains fall straight down to form another heap.
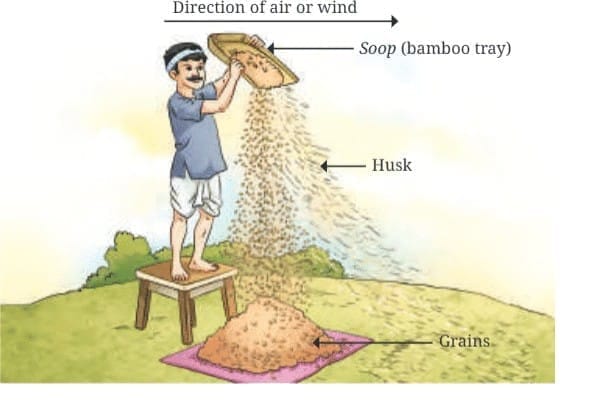
Winnowing is still commonly used by farmers where natural wind is available or by using a fan or winnowing device to create an airflow.
4. Sieving
This method is used when the particles are too small to be picked by hand or when there are too many. A sieve with appropriately sized holes is used. Larger particles stay on the sieve, while smaller ones pass through.
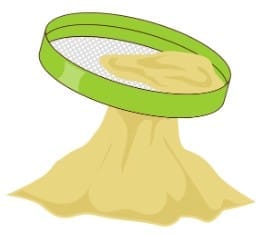
Smaller particles pass through the holes while larger particles remain on the sieve.
Examples: separating bran from wheat flour, or separating larger stones from sand.
If the holes in a sieve are larger than the particles being sieved, then both fine and larger particles will pass through. Choosing the correct hole size is important to obtain the desired separation.
5. Evaporation
Evaporation is the process of converting a liquid into vapour, leaving behind the dissolved solid. It is used when a solid has been dissolved in a liquid.

Example: Salt can be obtained from a salt solution by letting water evaporate under the sun or by gentle heating; the salt crystals remain after the water is gone.
How is salt obtained from seawater?
Seawater is collected in shallow pans and left in the sun. As water evaporates, salt crystals are left behind and collected.
Ayurveda: The Ancient Indian Healing System
- Ayurveda is a traditional Indian system of health and medicine that focuses on overall well-being.
- In Ayurveda, herbs and plant parts such as roots, leaves, flowers, and seeds are used to treat various health issues.
- The plant materials are often dried in the shade . This process helps remove excess moisture while preserving the medicinal properties of the plants.

6. Sedimentation and Decantation
- Sedimentation: This is when insoluble particles settle at the bottom. For example, in muddy water, soil and sand are impurities that settle down after the water is left to stand.
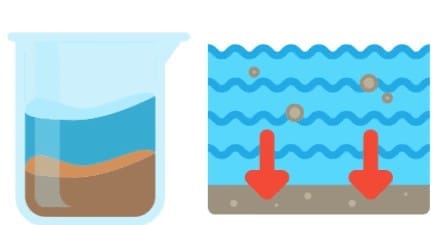
- Decantation: Used after sedimentation, this method separates the heavier sand particles from water. The clear water is carefully poured off, leaving the sand behind.
Try yourself: Which method is used to separate heavier particles from lighter particles using wind or blowing air?
7. Filtration
Filtration is a technique that separates fine, insoluble solid particles from liquids. The mixture is passed through a filter, which allows the liquid to flow through while trapping the solid particles.
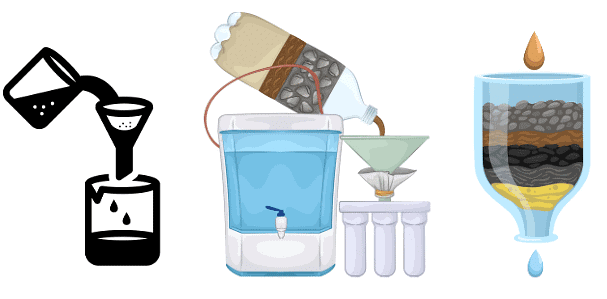
Example: Using a strainer to remove tea leaves from brewed tea.
What do you observe when filtering muddy water using filter paper?
When muddy water is poured through filter paper, the mud particles are trapped by the paper while clear water passes through and is collected.
Different materials used as filters
- Filter paper, cotton, muslin or gauze
- Charcoal and sand layered in a pot for simple water purification
The evolution of tea bags
- Tea bags were first made from soft materials like silk as it could hold the tea leaves while allowing hot water to pass through.
- Silk was chosen because it was strong and didn't break apart when it came into contact with hot water.
- Later on, people began using gauze or muslin for making tea bags.
- Eventually, the use of filter paper became common, and today, most tea bags are made from this material.

8. Churning
Churning is the process that separates lighter components from heavier ones by vigorous stirring or shaking. It is used to separate fat (butter) from milk solids.

- Example: Churning curd or cream separates butter (fat) from buttermilk.
9. Magnetic Separation
Magnetic separation uses a magnet to remove magnetic materials from non-magnetic ones.
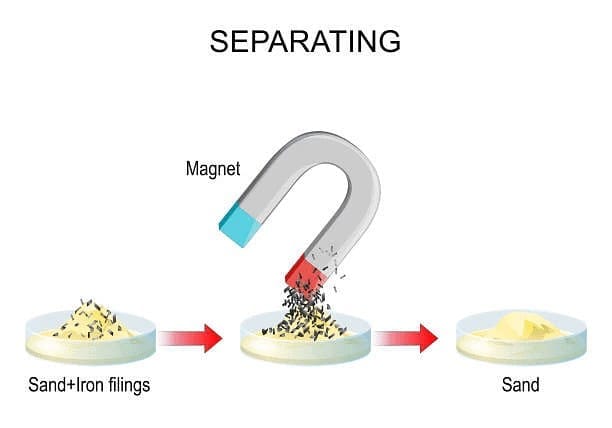
- Example: Using a magnet to take out iron nails from a mixture of sawdust and nails.
- These days, recyclers use magnets to remove iron items from a pile of waste. In various industries, waste materials often include scrap iron, which is extracted using magnets attached to cranes. This scrap iron can be recycled and reused.
Separation of scrap iron from waste
- Many wastes include scrap iron which is attracted by magnets.
- Magnetic substances (for example, iron) are separated from non-magnetic ones using magnets.
- The extracted scrap iron can be processed for reuse.

Key Points
- Handpicking: Remove unwanted items manually using size, colour or shape differences.
- Threshing: Separate grains from stalks by beating.
- Winnowing: Use wind or air to separate lighter husk from heavier grains.
- Sieving: Use a sieve to separate particles by size.
- Evaporation: Evaporate liquid to obtain dissolved solids.
- Sedimentation and Decantation: Let heavy particles settle and pour off the clear liquid.
- Filtration: Use a filter medium to separate fine insoluble solids from liquids.
- Churning: Agitate a mixture to separate lighter components (for example, butter from curd).
- Magnetic separation: Use magnets to separate magnetic materials from non-magnetic materials.
Practical Applications
- Removing small stones from pulses before cooking.
- Churning curd to make butter.
- Picking out green chillies from cooked dalia or poha.
- Removing seeds from watermelon.
- Separating pebbles from sand.
FAQs on Chapter Notes: Methods of Separation in Everyday Life
| 1. What are the different methods of separation and how do they work in everyday life? |  |
| 2. How does filtration separate solid particles from liquid mixtures? |  |
| 3. When should I use evaporation instead of filtration to separate mixtures? |  |
| 4. Why do we use sedimentation and decantation together for water purification at home? |  |
| 5. What's the difference between distillation and evaporation as separation methods? |  |