Very Short Question Answers: A Journey through States of Water | Short & Long Answer Questions for Class 6 PDF Download
Q1: What is water primarily composed of?
Ans: Water is composed of hydrogen and oxygen.
Q2: Are ice and water the same substance?
Ans: Yes, they are different states of the same substance.
Q3: What happens to water on a hot pan?
Ans: It converts to steam, which is water vapour.
Q4: What is the process of water turning into vapour called?
Ans: The process is called evaporation.
Q5: Why do puddles disappear on warm days?
Ans: Due to evaporation and water seeping into the ground.
Q6: Why do water droplets form on a cold bottle?
Ans: Due to condensation of water vapour from the air.
Q7: What is condensation?
Ans: Condensation is when water vapour turns into liquid water.
Q8: What are the three states of water?
Ans: Solid, liquid, and gas.
Q9: How can we change water's state?
Ans: By adding or removing heat.
Q10: What is melting?
Ans: Melting is the transition from solid (ice) to liquid (water).
Q11: What does an Atmospheric Water Generator do?
Ans: It produces drinkable water from humid air.
Q12: Why do clothes dry faster on windy days?
Ans: Wind increases the rate of evaporation.
Q13: What factors affect evaporation?
Ans: Temperature, surface area, air movement, and humidity.
Q14: How does evaporation cool us down?
Ans: Evaporation absorbs heat, cooling the surroundings.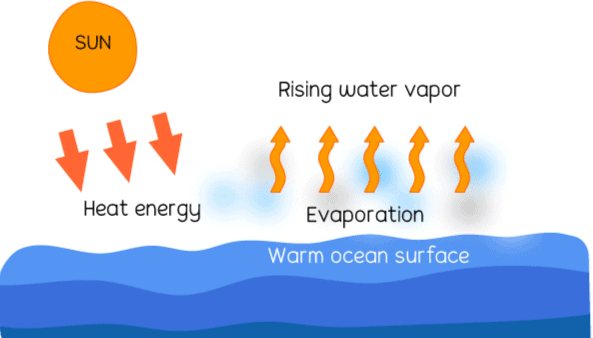
Q15: What role does condensation play in the water cycle?
Ans: It forms clouds and returns water to the Earth's surface.
Q16: Why does water vapour rise in the atmosphere?
Ans: It is less dense than the surrounding air.
Q17: What is the cooling effect of evaporation observed in?
Ans: Sweating and cooling of earthen pots.
Q18: What is freezing?
Ans: Freezing is the transition from liquid (water) to solid (ice).
Q19: How do clouds form rain?
Ans: Through condensation of water vapour into droplets.
Q20: Why is water essential for living things?
Ans: It is crucial for survival and maintaining health.
FAQs on Very Short Question Answers: A Journey through States of Water - Short & Long Answer Questions for Class 6
| 1. What are the three states of water? |  |
| 2. How does water change from one state to another? |  |
| 3. What is the water cycle? |  |
| 4. Why is water important for life? |  |
| 5. How can we conserve water in our daily lives? |  |

















