NCERT Solutions: Heat
Exercises
Q1. State similarities and differences between the laboratory thermometer and the clinical thermometer.
Ans:
Similarities:
- Both thermometers are used to measure temperature.
- Both laboratory and clinical thermometers have long, narrow, uniform glass tubes.
- They both contain mercury that expands or contracts with temperature changes.
- Both have a scale to read the temperature and both are graduated generally on the Celsius scale.
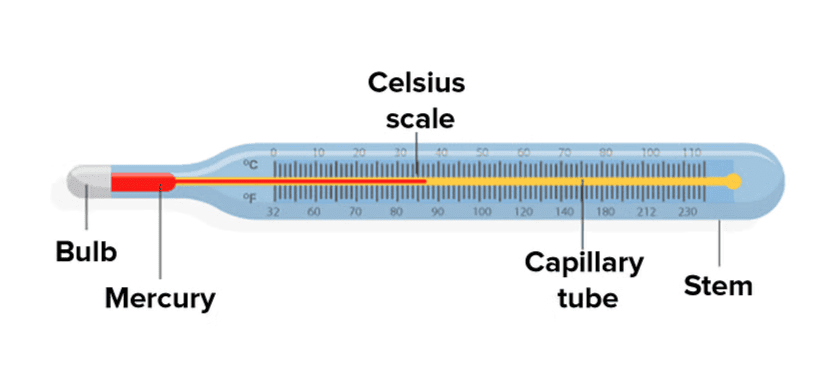 Clinical Thermometer
Clinical Thermometer Laboratory Thermometers
Laboratory ThermometersDifferences in Thermometers:
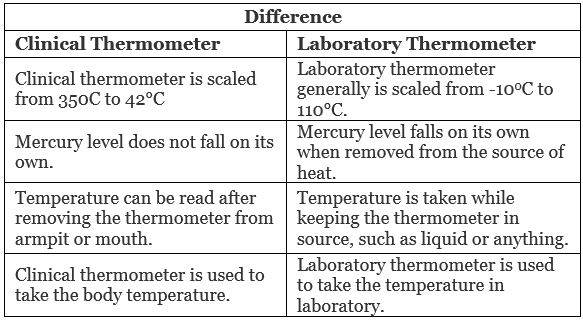
Q2. Give two examples each of conductors and insulators of heat.
Ans:
Conductors:
- Copper
- Aluminium
Insulators:
- Rubber
- Glass
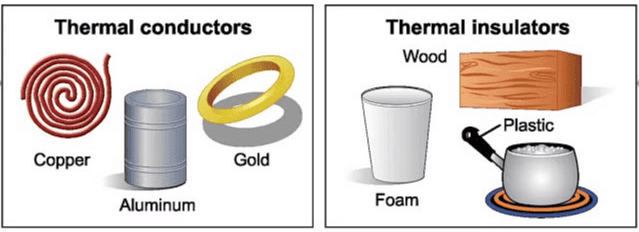 Thermal Conductors & Insulators
Thermal Conductors & InsulatorsQ3. Fill in the blanks:
(a) The hotness of an object is determined by its ________.
Ans: Temperature.
(b) Temperature of boiling water cannot be measured by a ________ thermometer.
Ans: Clinical thermometer
(c) Temperature is measured in degree ________.
Ans: Celsius
(d) No medium is required for transfer of heat by the process of __________.
Ans: Radiation
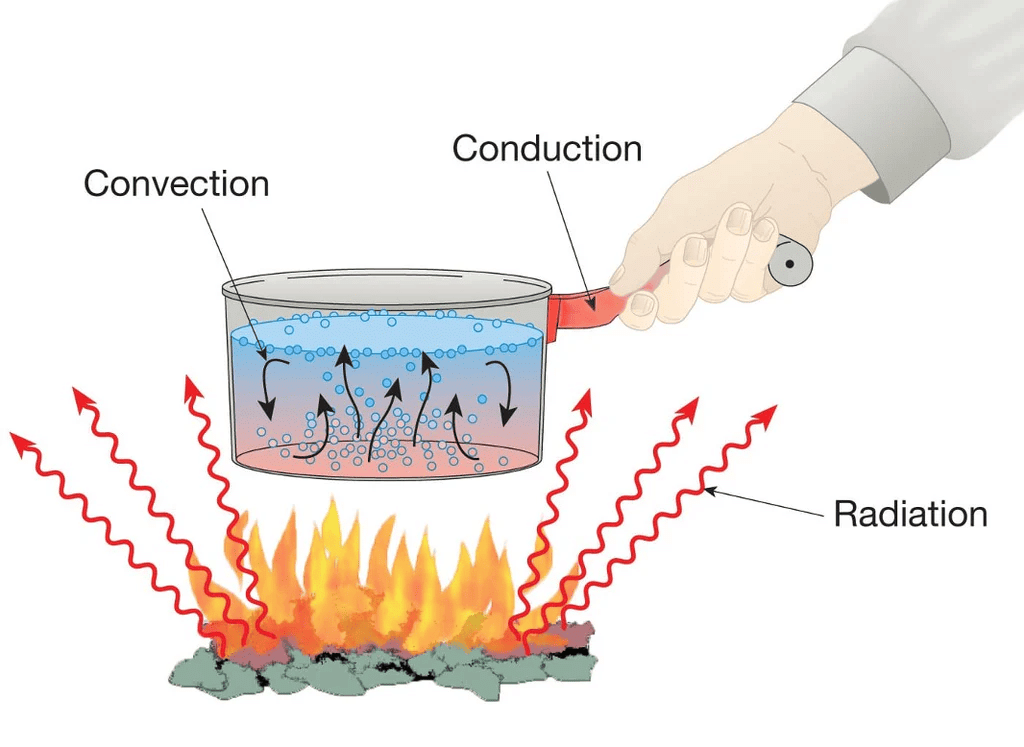 Heat Transfer
Heat Transfer(e) A cold steel spoon is dipped in a cup of hot milk. It transfers heat to its other end by the process of ________.
Ans: Conduction.
(f) Clothes of ________colours absorb heat better than clothes of light colours.
Ans: Dark
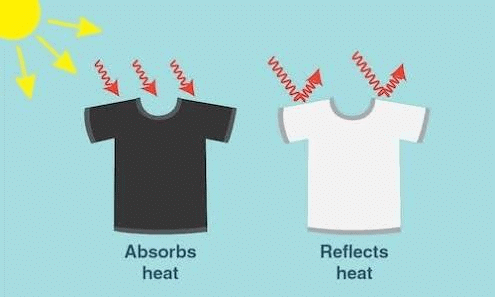 Dark colour clothes absorb Heat
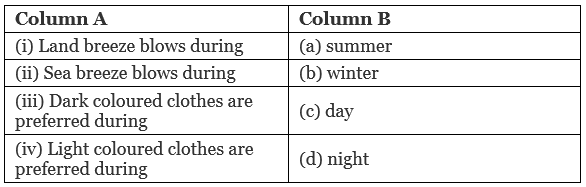
Dark colour clothes absorb HeatQ4. Match the following:
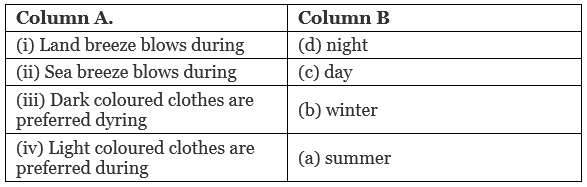
Ans:
Q5. Discuss why wearing more layers of clothing during winter keeps us warmer than wearing just one thick piece of clothing?
Ans:
- Multiple layers trap several thin pockets of air between them.
- Air is a poor conductor of heat, so these trapped pockets reduce heat loss from the body.
- One thick piece of clothing traps less air overall and therefore is less effective at preventing heat escape than several layers.
Q6. Look at Fig. (in NCERT Text Book). Mark where the heat is being transferred by conduction, by convection and by radiation.

Ans:
Q7. In places of hot climate it is advised that the outer walls of houses be painted white. Explain.
Ans:
- White colour reflects most of the sunlight and therefore reflects most of the heat falling on it.
- This reflection reduces the amount of heat absorbed by the walls and helps keep the house interior cooler.
- Hence, painting outer walls white is a simple way to lower indoor temperature in hot climates.
Q8. One litre of water at 30°C is mixed with one litre of water at 50°C. The temperature of the mixture will be
(a) 80°C
(b) more than 50°C but less than 80°C
(c) 20°C
(d) between 30°C and 50°C
Ans: (d)
Q9. An iron ball at 40°C is dropped in a mug containing water at 40°C. The heat will
(a) flow from iron ball to water.
(b) not flow from iron ball to water or from water to iron ball.
(c) flow from water to iron ball.
(d) increase the temperature of both.
Ans: (b)
Q10. A wooden spoon is dipped in a cup of ice cream. Its other end
(a) becomes cold by the process of conduction.
(b) becomes cold by the process of convection.
(c) becomes cold by the process of radiation.
(d) does not become cold.
Ans: (d)
Q11. Stainless steel pans are usually provided with copper bottoms. The reason for this could be that
(a) copper bottom makes the pan more durable.
(b) such pans appear colourful.
(c) copper is a better conductor of heat than stainless steel.
(d) copper is easier to clean than stainless steel.
Ans: (c)
FAQs on NCERT Solutions: Heat
| 1. What is the difference between heat and temperature in Class 7 Science? |  |
| 2. How does conduction of heat work and what are some real examples? |  |
| 3. What's the difference between radiation, conduction, and convection of heat? |  |
| 4. Why do dark coloured surfaces absorb more heat than light coloured ones? |  |
| 5. What is specific heat capacity and why does water have such high specific heat capacity? |  |






