NCERT Solutions: Separation of Substances
Exercises
Q1. Why do we need to separate different components of a mixture? Give two examples.
Ans: Before using a substance, we need to separate harmful or non-useful substances that may be mixed with it. Sometimes, we separate even useful components if we need to use them separately.
For example:
(i) We used to separate slightly larger-sized impurities like pieces of dirt, stone, and husk from wheat, rice, or pulses by the handpicking method.
(ii) Rice or pulses are usually washed before cooking. When we add water to these, the impurities like dust and soil particles get separated.
 Hand-picking stones from rice
Hand-picking stones from rice
Q2. What is winnowing? Where is it used?
Ans: Winnowing is the method of separating components of a mixture containing heavier and lighter components by wind or by blowing air. It is used to separate husk particles from seeds of grain.
Q3. How will you separate husk or dirt particles from a given sample of pulses before cooking.
Ans: Husk or bigger pieces of dirt particles can be removed from a sample of pulses by handpicking.
Q4. What is sieving? Where is it used?
Ans: Sieving is the process of filtering components of a mixture of different sizes. Sieving allows fine particles to pass through the holes of the sieve while the bigger impurities remain on the sieve. Sieving is used in flour mills to separate broken particles of grains from flour. It is also used at construction sites to separate lumps and smaller stones from the mixture of sand and cement.
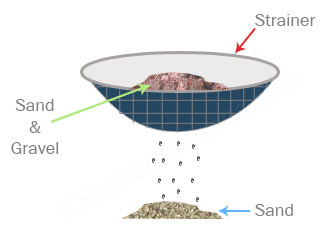 Sieving
Sieving
Q5. How will you separate sand and water from their mixture?
Ans: To separate sand and water from their mixture, we follow the following steps:
- Leave the mixture to stand undisturbed for some time in a container.
- Sand settles at the bottom of the container. It is called sedimentation.
- Gently pour the water into another container (called decantation).
- We may also use filter paper to remove fine particles of sand (called filtration)
Q6. Is it possible to separate sugar mixed with wheat flour? If yes, how will you do it?
Ans: Yes, it is possible to separate sugar mixed with wheat flour by the following method
- Mix sugar and wheat flour in water
- Stir the solution to allow the sugar to dissolve
- Now filter the mixture
- Filtrate contains a sugar solution, and the residue will be wheat flour
 Filtration
Filtration
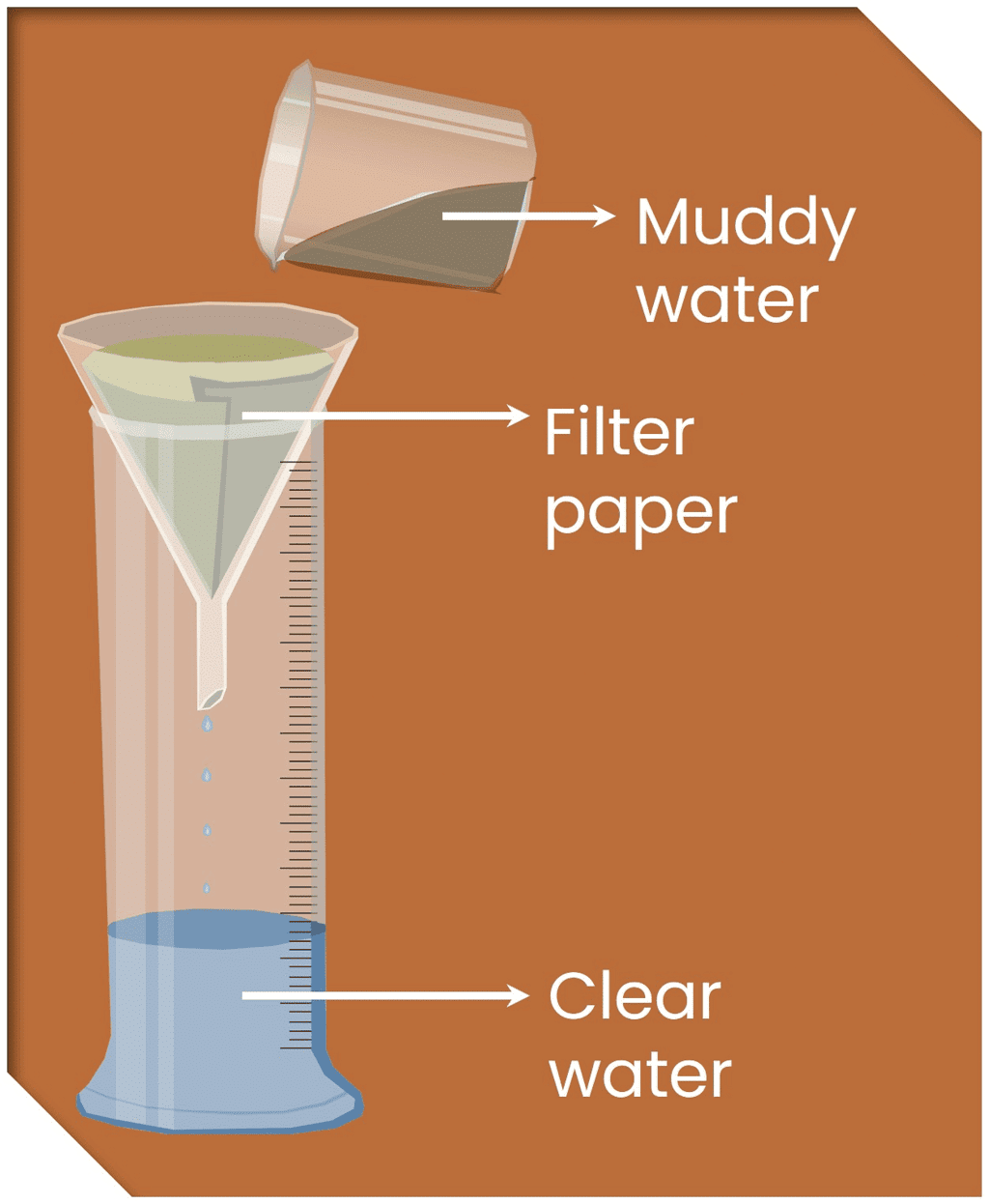
Q7. How would you obtain clear water from a sample of muddy water?
Ans: The following steps are required to obtain clear water from muddy water:
- Allow muddy water to stand undisturbed in a container.
- After some time, mud settles at the bottom of the container. This process is called sedimentation.
- The upper layer is clear water.
- Pour the clear water gently into another container. This process is called decantation.
- To remove finer impurities, we can filter this water again with the help of filter paper. This process is called filtration.
Q8. Fill up the blanks
(a) The method of separating seeds of paddy from its stalks is called ___________.
Ans: Threshing
(b) When milk, cooled after boiling, is poured onto a piece of cloth the cream (malai) is left behind on it. This process of separating cream from milk is an example of ___________.
Ans: Churning
(c) Salt is obtained from seawater by the process of ___________.
Ans: Evaporation
(d) Impurities settled at the bottom when muddy water was kept overnight in a bucket. The clear water was then poured off from the top. The process of separation used in this example is called ___________.
Ans: Sedimentation and decantation
Q9. True or false?
(a) A mixture of milk and water can be separated by filtration.
Ans: False
(b) A mixture of powdered salt and sugar can be separated by the process of winnowing.
Ans: False
(c) Separation of sugar from tea can be done with filtration.
Ans: False
(d) Grain and husk can be separated with the process of decantation.
Ans: False
Q10. Lemonade is prepared by mixing lemon juice and sugar in water. You wish to add ice to cool it. Should you add ice to the lemonade before or after dissolving sugar? In which case would it be possible to dissolve more sugar?
Ans: You should dissolve sugar in water before adding ice to the lemonade. Sugar dissolves more readily in warm water than in cold water. By dissolving sugar in water first, you maximize the amount of sugar that can be dissolved. Once the sugar is dissolved, you can then add ice to cool the lemonade without affecting the amount of sugar dissolved, as the sugar is already in the solution.
FAQs on NCERT Solutions: Separation of Substances
| 1. What are the different methods to separate mixtures in CBSE Class 6 Science? |  |
| 2. How do you separate salt from seawater using evaporation? |  |
| 3. What's the difference between evaporation and condensation in separation techniques? |  |
| 4. When should I use filtration versus decantation to separate mixtures? |  |
| 5. Why is magnetic separation useful and which materials can it separate? |  |






