Mixture and Solution

Introduction
- All the matter around us is not always pure; many substances are composed of two or more kinds of particles.
- If you examine soil and sugar under a magnifying glass, soil shows particles of clay, pieces of grass, small insects, etc.; such a substance is an impure substance or a mixture.
- Sugar, which shows only one kind of particle, is an example of a pure substance.
- Most materials around us are mixtures of two or more pure components. Examples: Milk, sea water, many minerals.
Types of Matter
On the basis of chemical nature, matter is classified into two broad types:
- Pure substances
- Mixtures (impure substances)
Pure Substances
- A homogeneous material that contains particles of only one kind and which has a definite set of properties is called a pure substance.
- A pure substance cannot be separated into other substances by physical methods.
- Examples: Oxygen, sulphur, iron.
- A substance composed of two or more different kinds of particles combined in a fixed proportion by weight is also regarded as a pure substance (a chemical compound).
- Examples: Sodium chloride (NaCl), magnesium oxide (MgO), carbon dioxide (CO2), copper sulphate (CuSO4).
 Copper Sulphate
Copper Sulphate- Not every homogeneous mixture is a pure substance. For example, a common salt solution in water is homogeneous but is not a pure substance because it contains two different substances: salt and water.
Mixtures or Impure Substances
- When two or more substances (elements, compounds or both) are mixed together in any proportion such that they do not undergo any chemical change but retain their individual characteristics, the resulting material is called a mixture.
- Examples: Brass (mixture of copper and zinc), crude oil (mixture of many hydrocarbons).
 A Mixture
A MixtureTypes of Mixtures
Depending on the nature of components and their distribution, mixtures are classified into two main types:
- Homogeneous mixture (Solution): Constituents are mixed uniformly; the mixture has the same composition throughout. The individual components cannot be seen even under a microscope. Examples: Salt solution, sugar solution, copper sulphate solution, alloys such as brass and bronze.
- Heterogeneous mixture: Constituents are not mixed uniformly and can be seen as distinct parts using the naked eye or a microscope. Examples: Sand and iron filings, sand and water.
Try yourself: Physical properties of a mixture:
Solutions

- A solution is a homogeneous mixture of two or more substances with uniform composition at the particle level.
- Solutions can be solid, liquid or gaseous. A solid solution example is an alloy; air is a gaseous solution of gases (about 78% nitrogen and 21% oxygen by volume).
- The substances present in a solution are called the components of the solution.
- Solutions have two basic components:
- Solvent: The component that dissolves the other component(s). It is usually the larger component of the solution. Example: Water is the solvent in a sugar solution.
- Solute: The component that dissolves in the solvent. It is usually the smaller component. Example: Sugar in sugar solution; carbon dioxide in soda water; iodine in alcohol (tincture of iodine).
Characteristics of a Solution
- A solution is homogeneous.
- Solute particles in a true solution are extremely small (less than 1 nm in diameter).
- Particles of a solution cannot be seen under a microscope.
- Solute particles pass through filter paper, so a solution cannot be separated by ordinary filtration.
- Solutions are stable; solute particles do not separate out on standing.
- A true solution does not scatter light because its particles are very small.
Concentration of a Solution
- The concentration of a solution is the amount of solute present in a given quantity of the solution-often expressed as the mass of solute present in 100 g of solution (mass percentage).
- Depending on the amount of solute, a solution may be described as dilute, concentrated or saturated. Different substances have different solubilities in the same solvent at the same temperature.
- One common way to express concentration is the percentage method.
- Mass percentage (for a solid solute dissolved in a liquid): mass percentage of solute = (mass of solute / mass of solution) × 100.
- Here, mass of solution = mass of solute + mass of solvent.
- Mass by volume percentage: Often used where mass of solute (in grams) is expressed per 100 mL (or per 1 L) of solution. Units may be g/mL or g/L depending on the context.
Classification of Solutions by Amount of Solute
- Saturated solution: At a given temperature, a solution that contains as much solute as it can dissolve is called saturated. Example: At 30°C, 36 g of common salt dissolves in 100 g of water; the solution containing 36 g salt in 100 g water is saturated at 30°C.
- Unsaturated solution: When the amount of solute present is less than the saturation level, the solution is unsaturated. Example: 30 g salt in 100 g water at 30°C is unsaturated.
- Supersaturated solution: A solution that contains more solute than is present in a saturated solution at the same temperature is supersaturated. Such solutions are unstable and excess solute may crystallise out on seeding or cooling.
Solubility of a Solute
- Solubility is the amount of solute (in grams) that dissolves in 100 g of water at a specified temperature.
- Solubility in water (at 20°C) - some examples:
- Copper sulphate: 21 g
- Potassium nitrate: 32 g
- Potassium chloride: 34 g
- Sodium chloride: 36 g
- Ammonium chloride: 37 g
- Sugar (sucrose): 204 g
Factors Affecting Solubility
- Temperature: For most solid solutes in water, solubility increases with temperature. For gases, solubility in water generally decreases as temperature increases.
- Pressure: Pressure has little effect on solubility of solids and liquids, but the solubility of gases in liquids is directly proportional to the partial pressure of the gas above the liquid
Alloys
- Alloys are homogeneous mixtures of two or more metals, or a metal and a non-metal, prepared to obtain desired properties. They are mixtures because components retain their properties to some extent and composition can vary.
- Example: Brass, an alloy of copper and zinc (commonly around 70% copper and 30% zinc).

Brass Keychain
Suspensions
- A suspension is a heterogeneous mixture in which small particles of a solid are spread throughout a liquid without dissolving.
- Particles in a suspension tend to settle on standing and can be separated by filtration because their size is larger than the pores of filter paper.
- Examples: Chalk powder in water, muddy water.
Properties of a Suspension
- Suspensions are heterogeneous.
- Particle size is large (greater than about 100 nm).
- Particles can usually be seen easily and scatter light.
- Particles settle down on standing (sedimentation), so suspensions are unstable.
Colloidal Solutions (Colloids)
- A colloid is a heterogeneous mixture in which particle sizes lie between those of true solutions and suspensions (roughly 10-9 m to 10-6 m). Such particles neither dissolve nor settle on standing.
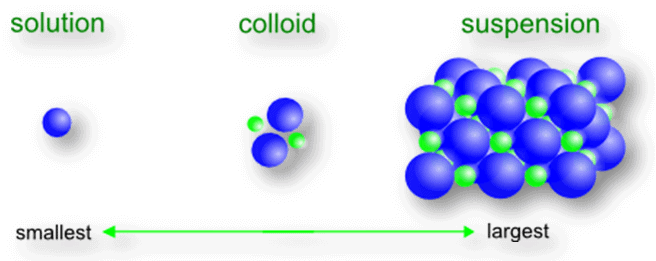
- Colloids consist of two parts: the dispersed phase (the small particles) and the dispersion medium (the medium in which the particles are dispersed).
- The dispersed phase is present in small proportion and the dispersion medium is present in excess.
Properties of Colloids
- Colloidal particles are too small to be seen individually with the naked eye.
- They do not settle down on standing; colloids are fairly stable.
- Colloids cannot be separated by ordinary filtration; centrifugation or other special methods are used.
- Colloidal dispersions are not fully transparent but are translucent.
- Colloidal particles scatter light; the path of a light beam becomes visible when it passes through a colloid.
Common Examples of Colloids

Brownian Movement of Colloids
- Colloidal particles show a random zig-zag motion called Brownian movement.
- This motion is caused by continuous bombardment of colloidal particles by the molecules of the dispersion medium.
Tyndall Effect
The phenomenon in which a beam of light becomes visible due to scattering by colloidal particles is called the Tyndall effect.
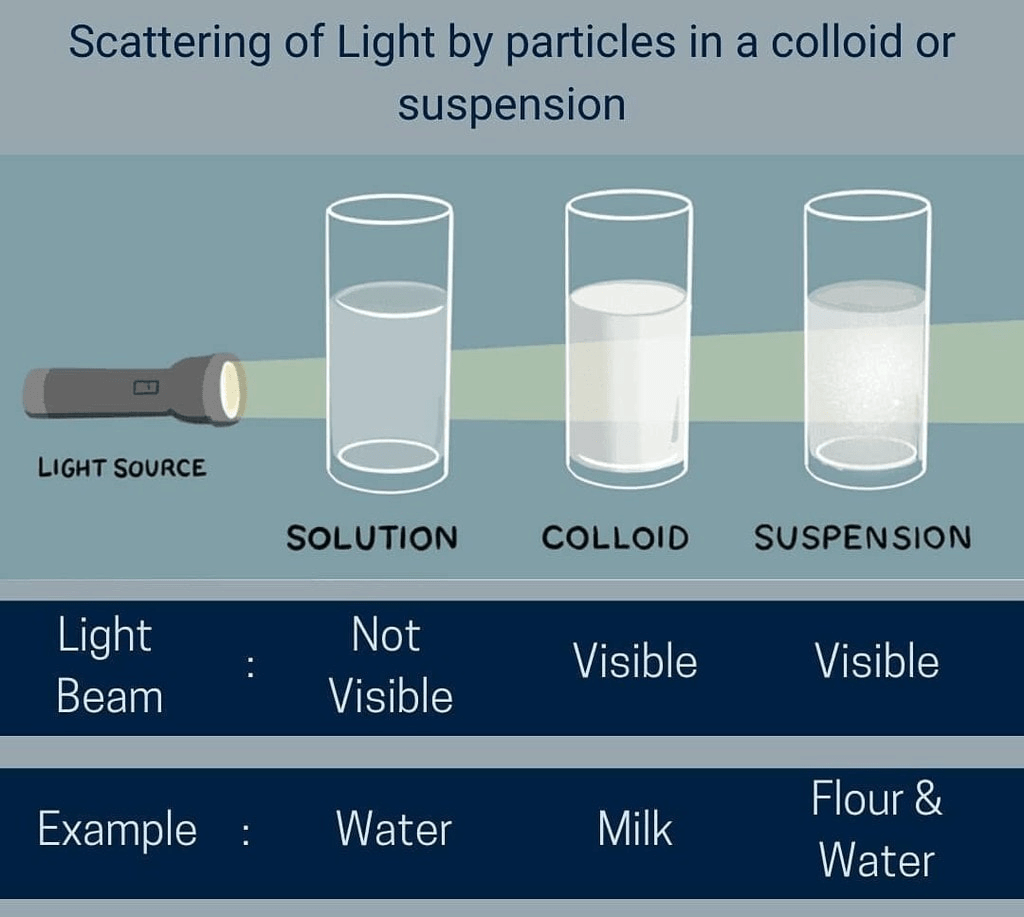
Example: The path of a strong beam of sunlight entering a dusty room appears visible because dust and smoke particles scatter light. Sunlight passing through mist in a forest also shows the Tyndall effect.
Difference Between True Solutions and Colloidal Solutions

Difference Between Colloidal Solutions and Suspensions

Types of Pure Substances

On the basis of chemical composition, pure substances are classified as elements and compounds.
Elements
- Robert Boyle first used the term element in 1661.
- Elements are the basic building blocks of matter; every substance is made of one or more elements.
- The input material states that 115 elements were discovered so far, of which 92 occur in nature and 23 have been synthesised in laboratories.
- Eleven elements are gases at room temperature; two elements are liquids at room temperature: mercury and bromine.
- Antoine-Laurent Lavoisier (1743-1794) defined an element as a basic form of matter that cannot be broken down into simpler substances by chemical means.
- Elements can also be defined as substances made up of atoms with the same atomic number.
- Examples: Hydrogen, oxygen, nitrogen, zinc, mercury.
- Elements are classified as metals, non-metals and metalloids.
Metals
- Most metals have a silver-grey colour and a shiny surface (lustre); some and their alloys may be yellowish or reddish (for example copper).
- Metals conduct heat and electricity well.
- They are malleable (can be hammered into thin sheets) and ductile (can be drawn into wires).
- They are generally sonorous (produce sound when struck).
- Examples: Gold, silver, copper, iron, sodium, potassium. Mercury is a metal that is liquid at room temperature.
Non-Metals
- Non-metals may be solids, liquids or gases at room temperature and display a variety of colours.
- They are poor conductors of heat and electricity (most of them).
- They are generally neither malleable nor ductile and are not sonorous.
- Examples: Hydrogen, oxygen, iodine, carbon.
Metalloids
- Metalloids (or semimetals) have intermediate properties between metals and non-metals.
- Examples: Boron, silicon, germanium.
 Silicon
SiliconCompounds
- A compound is a pure substance composed of two or more elements chemically combined in a definite ratio; compounds can be broken down into elements only by chemical means.
- The elements in a compound are called constituents or components of the compound.
- Example: Water is a compound of hydrogen and oxygen combined in a weight ratio of 1 : 8. Water can be decomposed into hydrogen and oxygen by electrolysis.
- Compounds can be classified into acids, bases and salts (chemical classification used in later chapters).
The product formed by mixing 1 g of sulphur powder and 2 g of iron filings/turnings is called a mixture.
- The constituents (iron filings and yellow sulphur particles) can be seen with the naked eye - a property of a mixture.
- The iron filings and sulphur can be separated by physical methods; for example, sulphur dissolves in carbon disulphide but iron does not, so the constituents can be separated - therefore the product is a mixture.
- The distribution of iron and sulphur in the mixture is not uniform; at some places iron filings are more, at others sulphur is more.
- Mixing iron and sulphur does not involve absorption or release of significant energy.
- Each component (iron and sulphur) retains its chemical and physical properties in the mixture.
The product formed on heating 1 g of sulphur powder and 2 g of iron turnings is called a compound.
- On heating, iron and sulphur react chemically to form iron sulphide; the yellow sulphur and iron filings no longer remain visible.
- Iron sulphide cannot be separated into iron and sulphur by physical means.
- The composition of iron sulphide is the same throughout (it is homogeneous at the macroscopic level).
- Heat energy is evolved when iron reacts with sulphur; the product remains hot and glowing even after heating is stopped.
- The properties of iron sulphide are entirely different from those of iron and sulphur.
Difference Between Mixtures and Compounds

Try yourself: Two substances A and B when bought together form a substance C with the evolution of heat. The properties of C are entirely different from those of A and B. the substance C is:
FAQs on Mixture and Solution
| 1. What are the different types of matter? |  |
| 2. What are solutions? |  |
| 3. What are alloys? |  |
| 4. What are suspensions? |  |
| 5. What are colloidal solutions or colloids? |  |







