Allotropes of Carbon & Hydrocarbons
Carbon occurs in several distinct forms in nature. These forms differ not because the atoms themselves are different but because the carbon atoms are bonded together in different ways. The different structural arrangements of carbon atoms give rise to materials with very different physical properties, even though their chemical behaviour is often similar.
What are Allotropes of Carbon?
The phenomenon of the existence of different structural forms of the same element is called allotropy. The different forms of an element are called allotropes. Allotropes of an element have different physical properties but often similar chemical properties, because the differences arise from different arrangements of atoms.
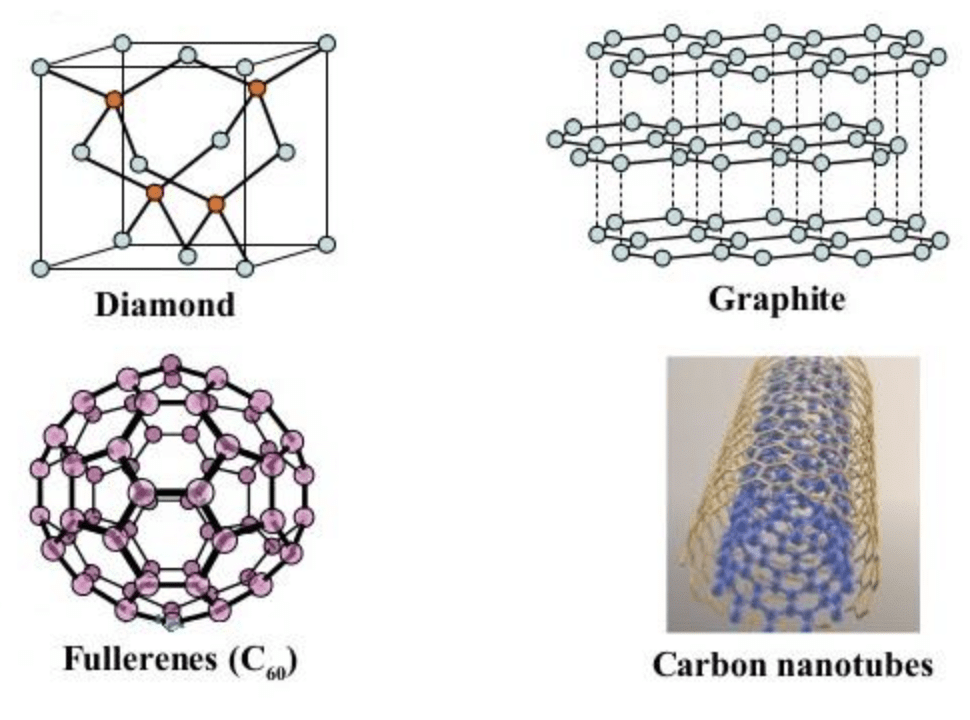 Allotropes of Carbon
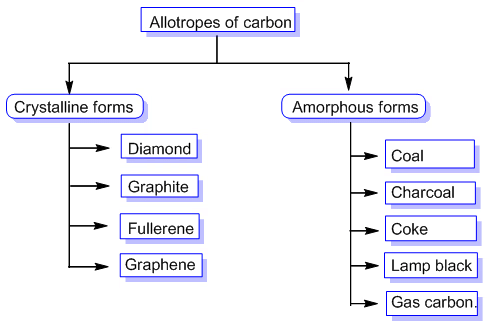
Allotropes of CarbonThe allotropes of carbon are commonly classified into two broad groups:
- Amorphous carbon allotropes: These forms do not have a well-defined long-range crystal structure. The arrangement of carbon atoms is disordered. Common examples include carbon black, activated carbon and charcoal.
- Crystalline carbon allotropes: These have a well-defined and repeating crystal lattice. Different arrangements produce markedly different properties. Important crystalline allotropes are diamond, graphite, fullerenes and carbon nanotubes.
Try yourself: What is allotropy?
We now examine the more widely known allotropes of carbon and explain their structures, properties and common uses.
1. Diamond
Diamond is a crystalline allotrope of carbon. The atomic symbol is C.

Structure of Diamond
Each carbon atom in diamond is covalently bonded to four other carbon atoms in a regular tetrahedral arrangement. These tetrahedra connect in three dimensions to give a very rigid and strong network. The strong covalent bonds throughout the lattice make diamond extremely hard and give it a high melting point.
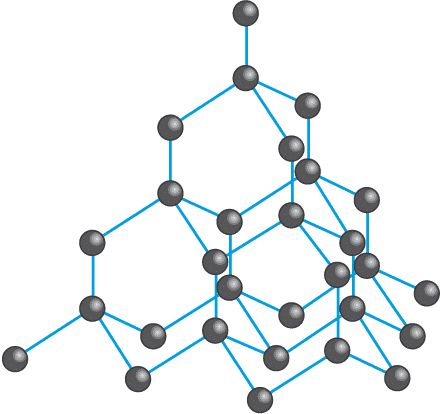
Physical Properties of Diamond
- Pure diamond is a transparent, colourless solid.
- It is the hardest known natural material and has a very high melting point.
- Diamond is an electrical insulator (does not conduct electricity) but is an excellent conductor of heat.
- Polished diamond shows high brilliance because of its strong refraction and reflection of light.
Uses of Diamond
- Industrial cutting and grinding: diamond-tipped saws and drills are used for sawing and shaping hard materials such as marble and stone.
- Glass cutting and precision machining: small diamond chips are used to score or cut glass and to draw fine wires of hard metals.
- Precision instruments: diamond is used in high-precision bearings and thermal sensors; protective windows in high-temperature and high-pressure conditions.
- Medical instruments: diamond-edged tools are used in some delicate surgical procedures (for example, in certain types of eye surgery).
- Gemstones: cut and polished diamonds are used in jewellery because of their brilliance and durability.
Try yourself: Which of the following properties is not a characteristic of diamond due to its tetrahedral arrangement of carbon atoms?
2. Graphite
Graphite is often called "black lead" because it leaves a dark mark on paper; the word graphite is derived from the Greek graphein meaning "to write". Graphite is a naturally occurring crystalline form of carbon.
Structure of Graphite
In graphite each carbon atom is covalently bonded to three other carbon atoms in the same plane, forming hexagonal rings. These hexagonal rings form layers (planes) of carbon atoms. One valence electron from each carbon atom is delocalised over the plane, which allows electrical conduction along the layers. The layers themselves are held together by weak van der Waals forces and can slide over one another easily.
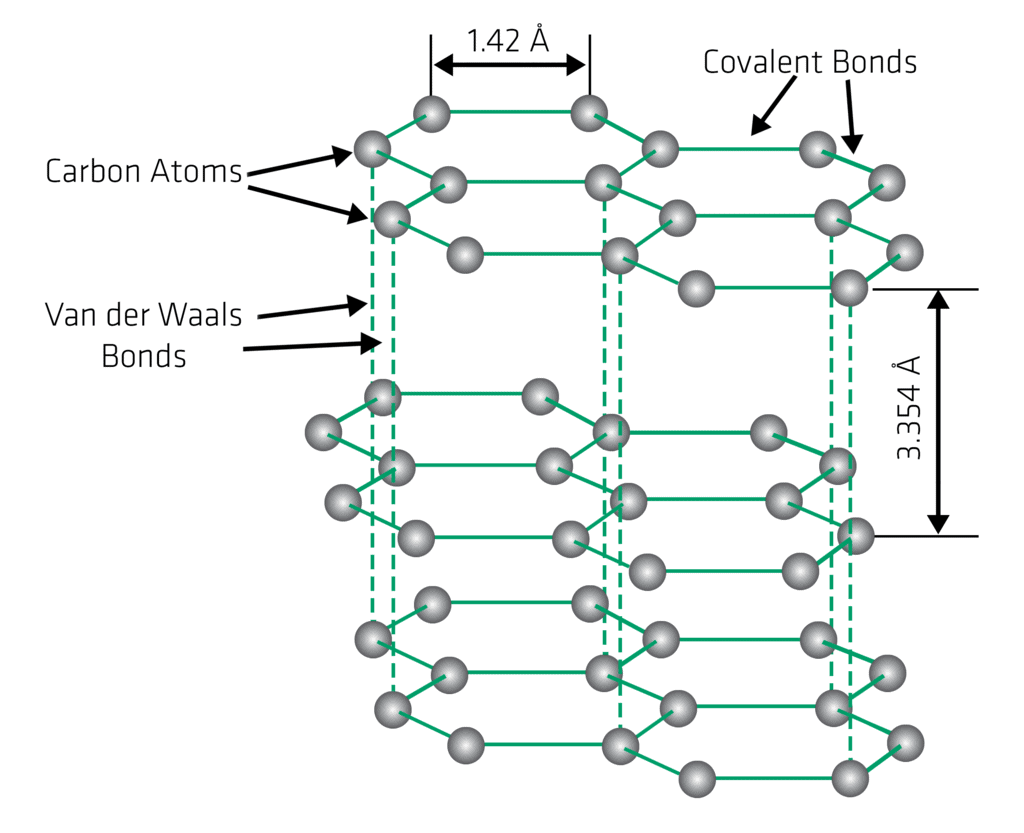
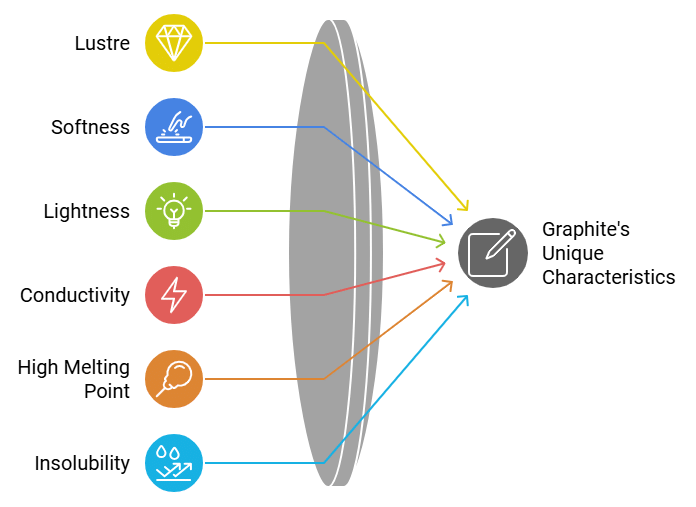
Physical Properties of Graphite
- Graphite is a dark grey to black, opaque solid with a metallic (shiny) lustre.
- It is soft and slippery because layers slide over each other; this property makes it useful as a dry lubricant.
- Graphite is lighter than diamond in density.
- Graphite is a good conductor of electricity (due to delocalised electrons within layers).
- It has a very high melting point and is insoluble in common solvents.
Uses of Graphite
- Electrodes in dry cells and in electric arc furnaces.
- Dry lubricant for machinery where oils and greases cannot be used.
- Manufacture of crucibles for melting metals at high temperatures.
- Lead pencils (the "lead" is a mixture of graphite and clay).
- Graphite is used as a neutron moderator in some types of nuclear reactors.
- Manufacture of certain types of paints, black dyes and industrial components.
- Starting material in processes to synthesise artificial diamond under high temperature and pressure.
3. Fullerenes
Fullerenes were discovered in 1985 by Robert F. Curl Jr., Harold Kroto and Richard E. Smalley. The first and most famous fullerene is the molecule containing sixty carbon atoms, C60, commonly called Buckminsterfullerene (or buckyball).
- The name honours the architect and inventor R. Buckminster Fuller, because the spherical shape resembles his geodesic domes.
- Other fullerenes include C70, C74 and C78, but C60 is the most stable and well studied.
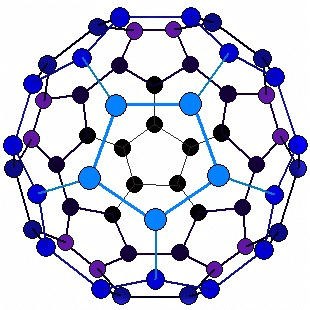
Structure of Fullerene (C60)
- The C60 molecule is nearly spherical and has a hollow, cage-like structure made up of 12 pentagonal faces and 20 hexagonal faces, giving a total of 60 carbon atoms at the vertices.
- The structure resembles a soccer ball, with carbon atoms at each vertex connected by covalent bonds in a pattern of pentagons and hexagons.
Preparation of Fullerene
- By electrically heating a graphite rod in an inert atmosphere such as helium.
- By vapourising graphite using a high-energy laser and then condensing the vapour in an inert gas stream.
Properties of Fullerene
- Fullerene (C60) is soluble in organic solvents such as benzene, producing a characteristic deep violet solution.
- Crystalline fullerenes exhibit semiconductor properties.
- Compounds of fullerenes with alkali metals (called fullerides) can show superconductivity under certain conditions.
Uses of Fullerene
- Research into superconducting and semiconducting materials.
- Potential use as lubricants, catalysts and in materials reinforcement.
- Applications in nanotechnology, for example as components in composite materials and molecular electronics.
Hydrocarbons
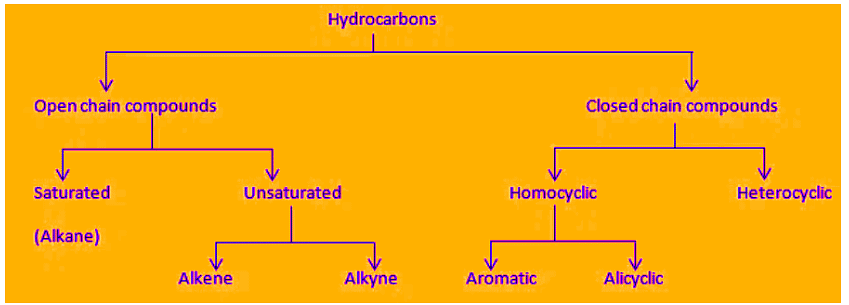
Hydrocarbons are organic compounds that contain only carbon and hydrogen atoms. They form the basic framework of organic chemistry and are classified by the types of bonds between carbon atoms.
 Classification of Hydrocarbons
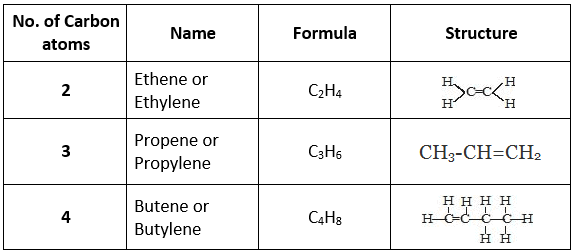
Classification of Hydrocarbons1. Saturated Hydrocarbons (Alkanes)
- The hydrocarbons that contain only single carbon-carbon bonds are called saturated hydrocarbons.
- They are also known as alkanes.
- The general formula for alkanes is CnH2n+2, where n is the number of carbon atoms.
 Some examples of saturated hydrocarbons
Some examples of saturated hydrocarbons2. Unsaturated Hydrocarbons
- Unsaturated hydrocarbons contain double or triple carbon-carbon bonds.
- They are divided into two main types: alkenes (with C=C double bonds) and alkynes (with C≡C triple bonds).
(i) Alkenes (Olefins)
Alkenes contain a carbon-carbon double bond (C=C). The general formula is CnH2n, where n is the number of carbon atoms. Alkenes show characteristic reactions such as addition reactions (e.g., hydrogenation, halogenation) because the double bond is a region of higher electron density and is more reactive than a single bond.
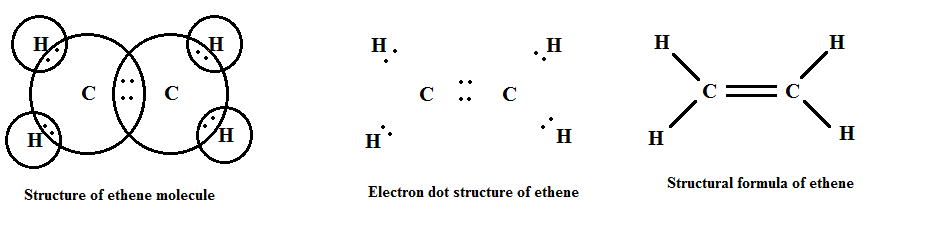
 Some examples of alkenes
Some examples of alkenes(ii) Alkynes
Alkynes contain a carbon-carbon triple bond (C≡C). The general formula for alkynes is CnH2n-2, where n is the number of carbon atoms. Alkynes also undergo addition reactions and have distinct chemical behaviour compared with alkanes and alkenes.
Try yourself: Which among the following are unsaturated hydrocarbons?
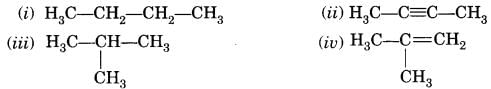
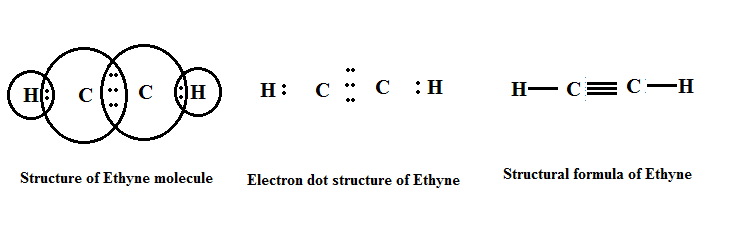
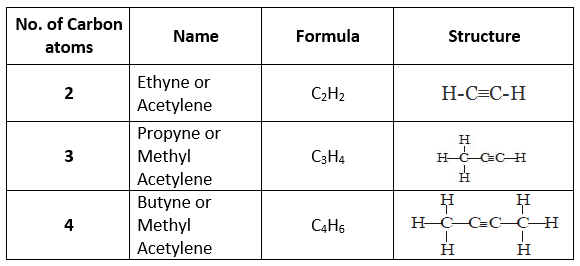
Some examples of alkynes include ethyne (C2H2), propyne (C3H4) and but-2-yne (C4H6). Alkynes are used as fuel gases (for example, ethyne or acetylene in welding), as starting materials for organic synthesis and as reagents in chemical industry.
Summary
Carbon exhibits allotropy: the same element forms different materials (such as diamond, graphite and fullerenes) depending on how the carbon atoms bond together. These allotropes differ widely in hardness, electrical conductivity, solubility and other physical properties. Hydrocarbons, compounds of carbon and hydrogen only, are classified as saturated (alkanes) or unsaturated (alkenes and alkynes) depending on the types of C-C bonds; each class has its characteristic general formula, properties and uses.
FAQs on Allotropes of Carbon & Hydrocarbons
| 1. What are the main allotropes of carbon? |  |
| 2. How do the physical properties of diamond and graphite differ? |  |
| 3. What are hydrocarbons and how are they classified? |  |
| 4. What are some common uses of hydrocarbons? |  |
| 5. How do the environmental impacts of hydrocarbons affect their usage? |  |






