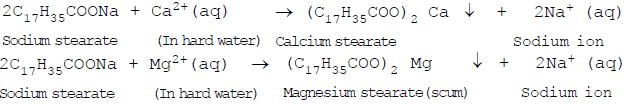Cleansing Action of Soaps & Detergents
Soap
Soap is the sodium or potassium salt of a long-chain fatty acid. Soaps are produced by the hydrolysis of fats or oils with an alkali and show cleansing action in water. A typical ionic representation of soap contains a long non-polar hydrocarbon chain and a polar ionic group such as -COONa. Soaps are alkaline in nature and a soap solution turns red litmus blue.
Examples:
- Sodium stearate (C17H35COONa).
- Sodium palmitate (C15H31COONa).
Structure
A soap molecule has two distinct parts with different affinities for water:
- a long hydrocarbon chain that is non-polar and water-repelling (hydrophobic), and
- a polar ionic end (for example -COO-Na+) that is water-loving (hydrophilic).
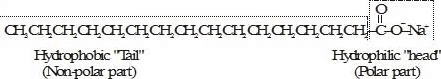
The hydrophilic end makes the molecule soluble in water, while the hydrophobic tail prefers oily or greasy substances. This dual nature is the basis of the cleansing action of soap.
A soap molecule may be represented as:
Preparation of Soap
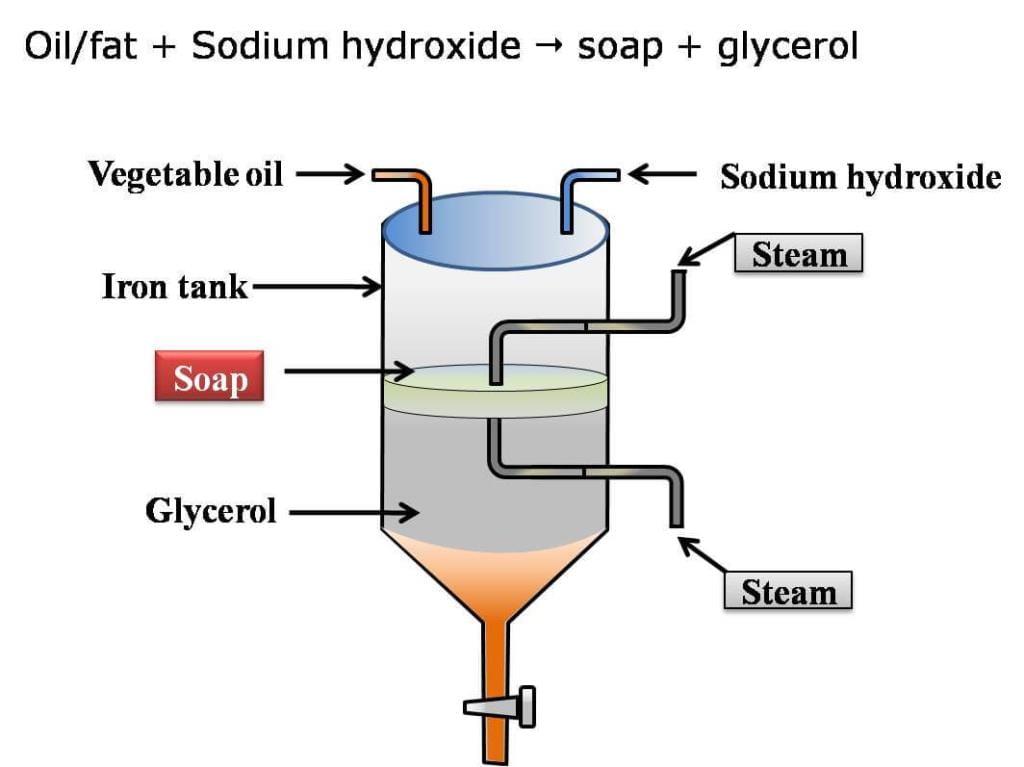
Soaps are prepared by heating animal fats or vegetable oils (for example olive oil, castor oil, palm oil) with a strong base such as sodium hydroxide or potassium hydroxide. This reaction is called saponification. In saponification, ester bonds in fats (triglycerides) are broken by alkali to give glycerol and the salt of a fatty acid (soap).
Chemical equation (general form): Fat or oil + Sodium hydroxide → Soap + Glycerol
Cleansing Action of Soap
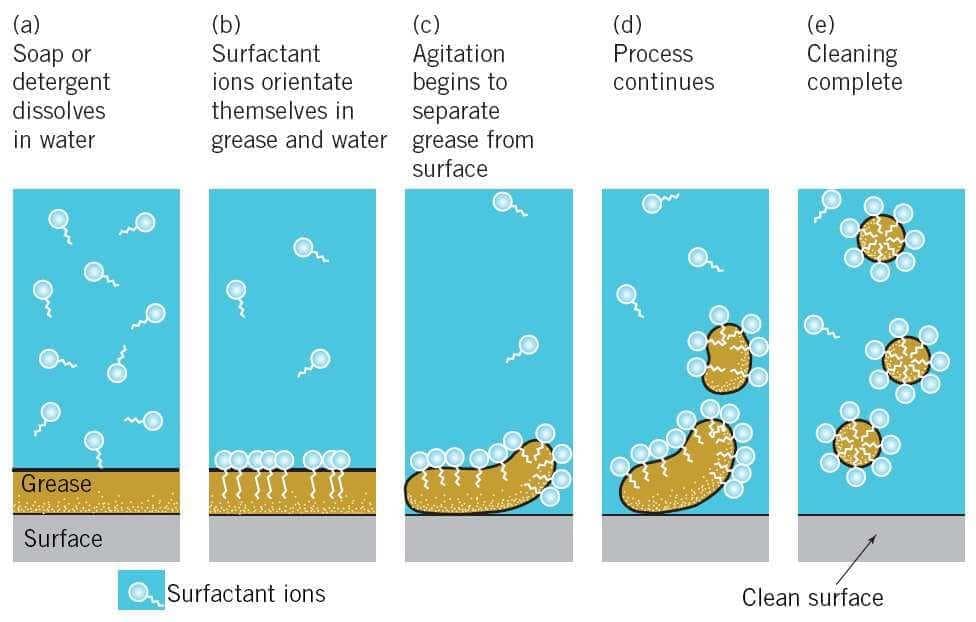
When soap is added to water containing oily dirt, the soap molecules arrange themselves around tiny oil droplets to form spherical aggregates called micelles. In a micelle:
- the hydrophobic (hydrocarbon) tails point towards the interior and dissolve the oily dirt, and
- the hydrophilic ionic heads remain on the outside in contact with water.
By surrounding the oil droplet, micelles make the oil droplets dispersible in water; this dispersal is an emulsion. The micelle-bound oil can then be rinsed away with water, thereby cleaning the surface.
Disadvantages of Soap
1. Reduced effectiveness in hard water. Hard water contains calcium (Ca2+) and magnesium (Mg2+) ions. These ions react with the carboxylate ions (RCOO-) of soap to form insoluble calcium or magnesium salts, commonly called scum. The formation of scum wastes soap and reduces cleansing action.

Representative reaction showing formation of insoluble calcium soap:
The scum formed adheres to clothes, utensils and skin and interferes with the formation of foam and with further cleansing.
2. Ineffectiveness in acidic medium. In an acidic medium soap (carboxylate salts) reacts with hydrogen ions to form the corresponding free fatty acids (undissociated). Fatty acids are weakly ionised and are insoluble in water, so micelle formation is hindered and cleansing action is lost.
Fatty acids precipitate out as a curdy white mass in acidic conditions, so soap cannot be used effectively in acidic solutions.
Detergents
Detergents are synthetic cleaning agents sometimes called "soapless soaps" because they do not contain fatty acid salts formed by saponification. Instead of a carboxylate group, detergents contain groups such as the sulphonic group (-SO3H) or alkyl hydrogen sulphate groups. Their salts (for example sodium salts) are used as cleaning agents.
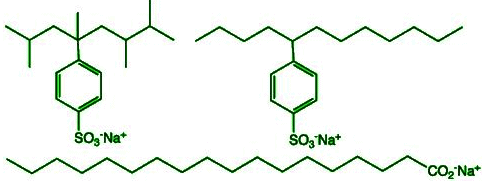

Properties of Detergents
Key properties that make detergents useful:
- Detergents have both hydrophobic (long hydrocarbon) and hydrophilic (ionic) parts, so they form micelles and remove grease in the same way as soaps.
- Detergents do not form insoluble salts with Ca2+ and Mg2+ ions, so they remain effective in hard water and produce lather.
- Detergents can be used in acidic media and in sea water where soaps are ineffective.
- Some detergents dissolve faster in water and give better cleansing action than ordinary soaps.
- Certain synthetic detergents are not readily biodegradable and may cause environmental problems such as persistent foaming in rivers and lakes.

Detergents have better cleansing action than soaps: Detergents are generally ammonium or sulphonate salts of long chain hydrocarbons. The charged ends of these compounds do not form insoluble precipitates with the calcium and magnesium ions in hard water. Thus, they remain effective in hard water. Detergents are usually used to make shampoos and products for cleaning clothes.
Differences between Soaps and Synthetic Detergents

Practical Notes and Applications
- Use soap for general cleaning in areas supplied with soft water for best efficiency.
- Use detergents for washing in hard water areas, for washing machines, in shampoos and for cleaning in acidic or sea-water conditions.
- Prefer biodegradable detergents where environmental discharge may affect rivers or lakes.
FAQs on Cleansing Action of Soaps & Detergents
| 1. What are the main differences between soaps and synthetic detergents? |  |
| 2. How do soaps clean surfaces? |  |
| 3. Why are synthetic detergents preferred over soaps in some situations? |  |
| 4. Can soaps and synthetic detergents harm the environment? |  |
| 5. What are the advantages of using soap over synthetic detergents? |  |