NEET Exam > NEET Notes > Chemistry Class 11 > Nomenclature of Elements with Atomic Number > 100
Nomenclature of Elements with Atomic Number > 100 | Chemistry Class 11 - NEET PDF Download
| Table of contents |

|
| Elements with Atomic Number > 100 (IUPAC) |

|
| Notation for IUPAC nomenclature of elements. |

|
| Nomenclature of Elements |

|

|
Elements with Atomic Number > 100 (IUPAC)
According to IUPAC, elements with atomic number > 100 are represented by three latter symbols.
- These symbols are based on the first letter of numbers from 0 to 9. The names of these numbers are derived from Greek and Latin languages.
- The Latin words for various digits of the atomic number are written together in the order of which makes the atomic number and suffix 'ium' is added at the end. In case of bi and tri one 'i' is omitted.
Notation for IUPAC nomenclature of elements.
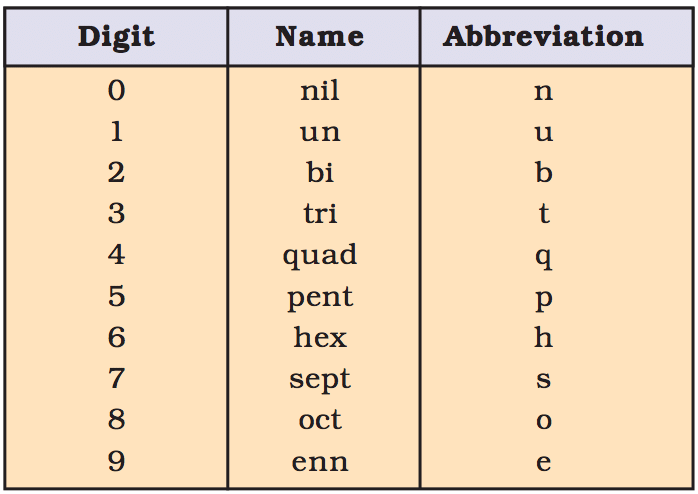
Nomenclature of Elements
Atomic Number | Name | Symbol | IUPAC Official Name | IUPAC Symbol |
104 | Unnilquadium | Unq | Rutherfordium | Rf |
105 | Unnilpentium | Unp | Dubnium | Db |
106 | Unnilhexium | Unh | Seaborgium | Sg |
107 | Unnilseptium | Uns | Bohrium | Bh |
108 | Unniloctium | Uno | Hassnium | Hs |
109 | Unnilennium | Une | Meitnerium | Mt |
110 | Ununnilium | Uun | Darmstadtium | Ds |
111 | Ununnium | Uuu | * | * |
112 | Ununbium | Uub | * | * |
113 | Ununtrium | Uut | + | |
114 | Ununquadium | Uuq | * | * |
115 | Ununpentium | Uup | + | |
116 | Ununhexium | Uuh | + | |
117 | Ununseptium | Uus | + | |
118 | Ununoctium | Uuo | + |
- Official IUPAC name yet to be announced + Elements yet to be discovered
- IUPAC recommended this nomenclature to be followed until their names are officially recognised.
The document Nomenclature of Elements with Atomic Number > 100 | Chemistry Class 11 - NEET is a part of the NEET Course Chemistry Class 11.
All you need of NEET at this link: NEET
|
114 videos|263 docs|74 tests
|
FAQs on Nomenclature of Elements with Atomic Number > 100 - Chemistry Class 11 - NEET
| 1. What is the IUPAC nomenclature for elements with atomic number greater than 100? |  |
Ans. The IUPAC nomenclature for elements with atomic number greater than 100 is based on a systematic naming system where the element is named using its atomic number as a prefix followed by the suffix "-ium". For example, element 101 is named "unnilunium" and element 102 is named "unnilbium".
| 2. How many elements with atomic number greater than 100 have been discovered so far? |  |
Ans. As of now, a total of six elements with atomic number greater than 100 have been discovered. These elements are 101- Mendeleevium (Md), 102- Nobelium (No), 103- Lawrencium (Lr), 104- Rutherfordium (Rf), 105- Dubnium (Db), and 106- Seaborgium (Sg).
| 3. Who is responsible for assigning names to elements with atomic number greater than 100? |  |
Ans. The International Union of Pure and Applied Chemistry (IUPAC) is responsible for assigning names to elements with atomic number greater than 100. They follow a systematic approach to ensure consistency and uniformity in the nomenclature of elements.
| 4. What are the criteria for naming elements with atomic number greater than 100? |  |
Ans. The criteria for naming elements with atomic number greater than 100 include using the element's atomic number as a prefix, followed by the suffix "-ium". The names should be unique and not conflicting with any existing names or symbols. The names are also subjected to approval by the IUPAC.
| 5. Can the names of elements with atomic number greater than 100 be changed in the future? |  |
Ans. Yes, the names of elements with atomic number greater than 100 can be changed in the future. The IUPAC periodically reviews and reevaluates the names of elements, and if necessary, they can propose changes to ensure accuracy and consistency in the nomenclature. However, any proposed changes would go through a thorough scientific and public scrutiny process before being officially adopted.
Related Searches





















