Types of Redox Reactions
Combination Reactions
Definition: A combination (or synthesis) reaction is one in which two or more substances combine to form a single product. The general form is:
- A + B → C
Such a reaction is a redox reaction when there is a change in oxidation number of one or more elements between reactants and product. In many practical cases one reactant is in the elemental state (oxidation number 0) and the other is in a compound or another element. All combustion reactions in which elemental dioxygen reacts are examples of redox combination reactions.
Typical examples:
- 2Mg + O2 → 2MgO. Oxidation numbers: Mg (0 → +2), O (0 → -2). Magnesium is oxidised and oxygen is reduced.
- N2 + 3H2 → 2NH3. Oxidation numbers: N (0 → -3), H (0 → +1). Nitrogen is reduced, hydrogen is oxidised.
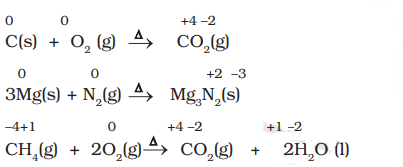 Examples of Combustion Reactions
Examples of Combustion ReactionsDecomposition Reactions
Definition: A decomposition reaction is the reverse of a combination reaction; a compound breaks down into two or more simpler substances. The general form is:
- AB → A + B
Whether a decomposition reaction is a redox process depends on changes in oxidation numbers of the elements involved. Some decomposition reactions are redox, while others are not.
Examples and notes:
- 2KClO3 → 2KCl + 3O2. This is a redox decomposition. Potassium remains in the +1 oxidation state while oxygen changes from -1 (in chlorate) to -2 (in O2? note: in O2 oxygen is 0) and 0 respectively; oxygen is both oxidised and reduced among products.
- CaCO3 → CaO + CO2. This is not a redox reaction because the oxidation states of Ca (+2), C (+4) and O (-2) remain unchanged.
It is important to examine oxidation numbers for each element to decide whether a given decomposition is redox. Some elements may keep the same oxidation number (for example, K in the decomposition of potassium chlorate remains +1), while other elements in the same reaction change their oxidation states.
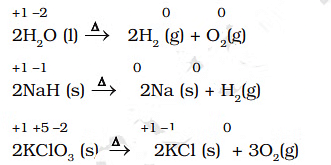 Examples of Decomposition Reactions
Examples of Decomposition Reactions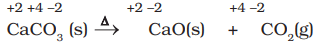
Displacement Reactions
Definition: In a displacement (single-replacement) reaction an element in the uncombined state displaces another element from a compound. The general form is:
- X + YZ → XZ + Y
Displacement reactions are redox reactions because one species is oxidised (loses electrons) and the other is reduced (gains electrons). Displacement reactions are normally classified into metal displacement and non-metal displacement.
Metal displacement
A metal in the elemental form can displace a less reactive metal from its compound. The tendency to displace is guided by the activity series of metals: a metal higher in the series displaces ions of a metal lower in the series from solutions of their salts.
Examples:
- Zn + CuSO4 → ZnSO4 + Cu. Oxidation numbers: Zn (0 → +2), Cu (+2 → 0). Zinc is oxidised and acts as the reducing agent; Cu2+ is reduced to Cu.
- Fe + CuSO4 → FeSO4 + Cu. Oxidation numbers: Fe (0 → +2), Cu (+2 → 0).
 Some Examples of Metal Displacement Reactions
Some Examples of Metal Displacement ReactionsMetal displacement reactions are used in metallurgy to obtain pure metals from their ores or salts by reduction with a more reactive metal.
Non-metal displacement
Non-metal displacement reactions include displacement among halogens and hydrogen displacement by metals.
Halogen displacement:
- Cl2 + 2Br- → 2Cl- + Br2. Chlorine (0) is reduced to Cl- (-1) and bromide (-1) is oxidised to bromine (0). A more reactive halogen displaces a less reactive halide ion from solution.
Hydrogen displacement:
- All alkali metals and some alkaline earth metals (Ca, Sr, Ba) displace hydrogen from cold water, giving the corresponding hydroxide and hydrogen gas. Example: 2Na + 2H2O → 2NaOH + H2.
- Less active metals such as magnesium and iron react with steam to produce hydrogen: Mg + H2O(steam) → MgO + H2.
- Many metals displace hydrogen from dilute acids. Example: Zn + 2HCl → ZnCl2 + H2. Some metals (for example cadmium, tin) displace hydrogen from acids but do not react with steam.
- Very unreactive metals such as silver and gold do not displace hydrogen from acids and often occur in the native state.
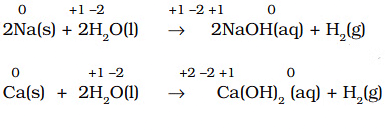
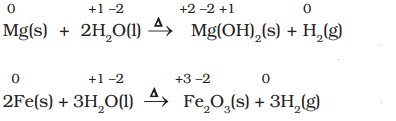
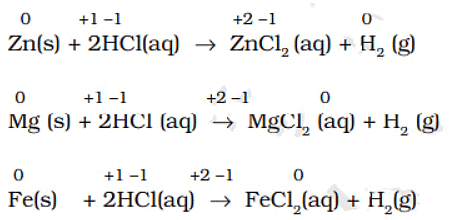
Disproportionation Reactions
Definition: A disproportionation reaction is a special type of redox reaction in which the same element in one oxidation state undergoes simultaneous oxidation and reduction to give two different products in higher and lower oxidation states.
Key points:
- One element is both oxidised and reduced in the same reaction.
- Disproportionation requires that the element be able to exist in at least three oxidation states so that an intermediate state can transform into both a higher and a lower state.
- The reacting form of the element is in an intermediate oxidation state prior to disproportionation.
Example: Decomposition of hydrogen peroxide:
- 2H2O2 → 2H2O + O2. Oxidation numbers of oxygen in H2O2 are -1; in H2O it is -2 and in O2 it is 0. Thus oxygen is both reduced (-1 → -2) and oxidised (-1 → 0) in the same reaction.
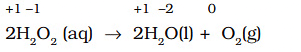
How to identify redox behaviour quickly
- Assign oxidation numbers to all elements in reactants and products; any change indicates electron transfer and therefore a redox process.
- If an element in the reactants is in elemental form (oxidation number 0) and converts to an ion or compound, that element is being oxidised or reduced depending on the sign change.
- In displacement reactions, refer to the activity series for metals and reactivity order for halogens to predict whether displacement will occur.
Summary
Combination, decomposition, displacement and disproportionation are the principal types of redox reactions encountered at the Class 11 level. For each reaction, examining oxidation numbers, identifying which species lose or gain electrons, and recognising the role of reducing and oxidising agents clarifies whether the process is a redox change and helps balance and interpret the reaction correctly.
FAQs on Types of Redox Reactions
| 1. What is a combination reaction? |  |
| 2. What is a decomposition reaction? |  |
| 3. What is a displacement reaction? |  |
| 4. What is a disproportionation reaction? |  |
| 5. What are the types of redox reactions? |  |






