NEET Exam > NEET Notes > Chemistry Class 11 > Introduction to Organic Chemistry
Introduction to Organic Chemistry
General Introduction
- The branch of chemistry which deals with Carbon and its derivatives is called Organic chemistry.
- Organic chemistry began to emerge as a science about 200 years ago. By the late eighteenth century, substances were divided into two classes called organic and inorganic compounds.
Try yourself: What percent of our body is made up of carbon?MULTIPLE CHOICE QUESTION
- Organic compounds were obtained from plants and animals.
- Organic compounds were more difficult to work within the laboratory and decompose easily.
- The hydrides of carbon (hydrocarbons) and their derivatives are called organic compounds. The branch of chemistry which deals with these compounds is called organic chemistry.
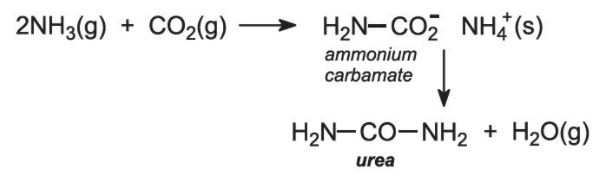
- Berzelius (1808) defined organic chemistry as the chemistry of substances found in living matter and gave the vital force theory.
- The synthesis of urea, the first organic compound synthesised in the laboratory, by Wohler who gave the death blow to the vital force theory.
- Acetic acid is the first organic compound synthesized from its elements.
Tetravalency of Carbon: Shapes of Organic Compounds
(a) Catenation
- It is the tendency of self combination and is maximum in carbon.
- A carbon atom can combine with other carbon atoms by single, double, or triple bonds.
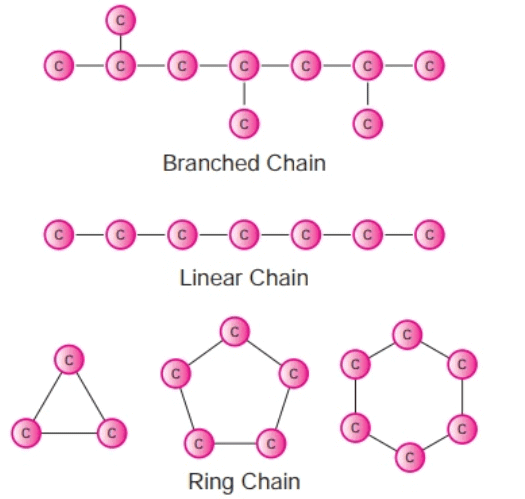
- Thus, Carbon forms more compounds than the other elements.
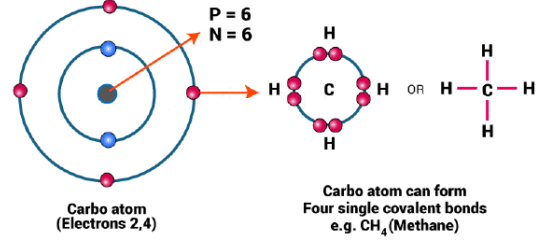
(b) Tetravalency and Small Size
- Carbon is tetravalent, is capable of bonding with four other C atoms or some other monovalent atoms.
- Carbon can form a compound with oxygen, hydrogen, chlorine, sulphur, nitrogen and phosphorus.
- These compounds have specific properties depending upon the nature of the element or group attached to the carbon.
- Furthermore, these compounds are exceptionally stable because of the small size of carbon.
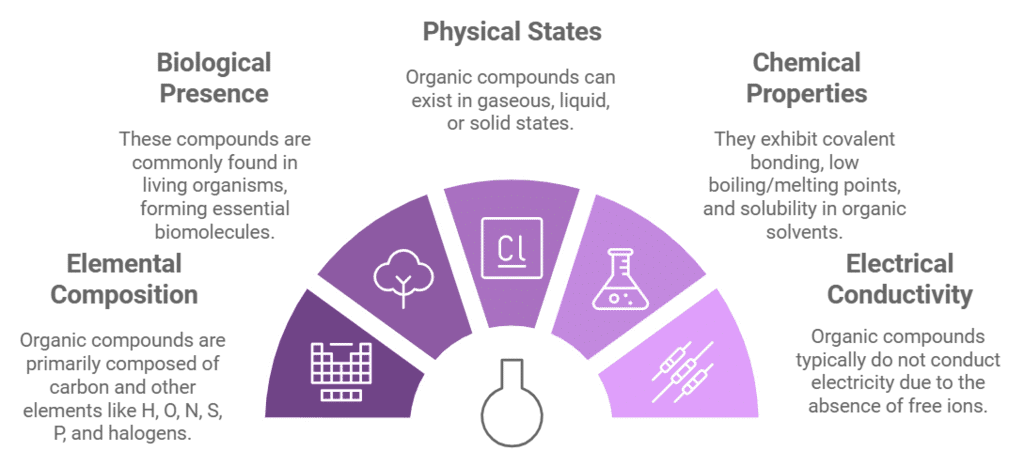
General Characteristics of Organic Compounds
- These are the compounds of carbon with H, O, N, S, P, F, CI, Br and I.
- These are generally found in living organisms. e.g., carbohydrates, proteins etc.
- These may be gases, liquids or solids.
- Being covalent in nature, these have a low boiling point and melting point and soluble in organic solvents.
- These are generally volatile and inflammable.
- They do not conduct electricity because of the absence of free ions.
- They possess distinct colour and odour.
MULTIPLE CHOICE QUESTION
Try yourself: Carbon is the _____ most abundant element in the universe
Structural Representations of Organic Compounds
- Complete Structural Formula - Full structural equations show all the atoms in a molecule, the types of bonds that bind them, and how they are interconnected.
- Condensed Structural Formula - The Condensed structural formula is used to save space, structural formulas are conveniently abbreviated as condensed structural formulas.
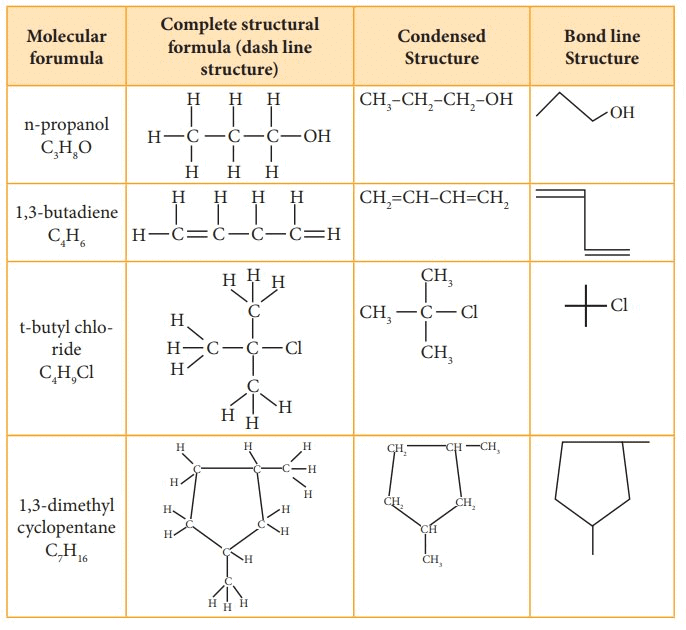
- Bond Line Structural Formula - A bond-line structure is a less cluttered drawing than a condensed structural formula. However, to understand the simplified bond-line structure, the reader has to mentally add many more features to comprehend the overall structure.
The document Introduction to Organic Chemistry is a part of the NEET Course Chemistry Class 11.
All you need of NEET at this link: NEET
FAQs on Introduction to Organic Chemistry
| 1. What is the difference between organic and inorganic chemistry for JEE? |  |
Ans. Organic chemistry studies carbon-containing compounds with covalent bonds, while inorganic chemistry covers all other elements and their compounds. For JEE, organic chemistry focuses on structure, reactions, and mechanisms of hydrocarbons and functional groups, making it essential for both Main and Advanced levels. Understanding this distinction helps students prioritise topics and recognise reaction patterns specific to carbon chemistry.
| 2. Why do we need to learn hybridisation and bonding in organic chemistry before tackling reactions? |  |
Ans. Hybridisation determines how atoms bond and influences molecular shape, reactivity, and stability-all fundamental to predicting organic reactions. sp, sp², and sp³ hybridisation directly explain why certain carbons are more reactive or why molecules adopt specific geometries. Without grasping these concepts, students struggle to understand why specific reactions occur and how to predict products, making it a prerequisite for mechanism-based problem solving.
| 3. What are functional groups and why do I need to memorise them for JEE Main? |  |
Ans. Functional groups are specific atom arrangements (like -OH, -COOH, -NH₂) that determine a molecule's chemical properties and reactivity. Rather than memorising, students should understand how each group influences reactivity and which reactions it undergoes. Recognising functional groups quickly helps identify reaction types during exams and predict products without memorising every possible reaction.
| 4. How do I identify whether a carbon is primary, secondary, or tertiary in organic molecules? |  |
Ans. Primary carbons bond to one other carbon, secondary carbons to two, and tertiary carbons to three-count carbon neighbours only. This classification directly affects reactivity in substitution and elimination reactions; tertiary carbons are most reactive in SN1 reactions, while primary carbons favour SN2 mechanisms. Quick classification during problem-solving prevents mistakes in predicting reaction pathways and product stability.
| 5. What's the easiest way to understand IUPAC nomenclature without getting confused by all the rules? |  |
Ans. Start by identifying the longest carbon chain (parent name), then locate and number substituents from the end nearest to functional groups or first branch point. Practice naming simple alkanes and alkenes before tackling complex molecules with multiple functional groups. Use flashcards and mind maps to memorise prefixes and suffixes systematically, building complexity gradually rather than memorising random examples.
Related Searches
Extra Questions, Sample Paper, ppt, Previous Year Questions with Solutions, Introduction to Organic Chemistry, Objective type Questions, study material, shortcuts and tricks, Summary, mock tests for examination, MCQs, video lectures, past year papers, practice quizzes, Exam, Introduction to Organic Chemistry, Viva Questions, Important questions, Introduction to Organic Chemistry, Semester Notes, pdf , Free;






