Carbohydrates | Biology Class 11 - NEET PDF Download
What are Carbohydrates?
Carbohydrates are macronutrients and are one of the three main ways by which our body obtains its energy.
They are called carbohydrates as they comprise carbon, hydrogen and oxygen at their chemical level. Carbohydrates are essential nutrients that include sugars, fibres and starches. They are found in grains, vegetables, fruits and milk and other dairy products. They are the basic food groups that play an important role in a healthy life.
The food containing carbohydrates are converted into glucose or blood sugar during the process of digestion by the digestive system.
Our body utilizes this sugar as a source of energy for the cells, organs and tissues. The extra amount of energy or sugar is stored in our muscles and liver for further requirements. The term ‘carbohydrate’ is derived from the French term 'hydrate de carbone' meaning ‘hydrate of carbon‘. The general formula of this class of organic compounds is Cn(H2O)n.
Classification of Carbohydrates
The carbohydrates are further classified into simple and complex which is mainly based on their chemical structure and degree of polymerization.
Simple Carbohydrates (Monosaccharides, Disaccharides and Oligosaccharides)
Simple carbohydrates have one or two sugar molecules. In simple carbohydrates, molecules are digested and converted quickly resulting in a rise in the blood sugar levels. They are abundantly found in milk products, beer, fruits, refined sugars, candies, etc. These carbohydrates are called empty calories, as they do not possess fiber, vitamins and minerals.
Plants, being producers, synthesize glucose (C6H12O6) using raw materials like carbon dioxide and water in the presence of sunlight. This process of photosynthesis converts solar energy to chemical energy. Consumers feed on plants and harvest energy stored in the bonds of the compounds synthesized by plants.
1. Monosaccharides
Glucose is an example of a carbohydrate monomer or monosaccharide. Other examples of monosaccharides include mannose, galactose, fructose, etc. The structural organization of monosaccharides is as follows:
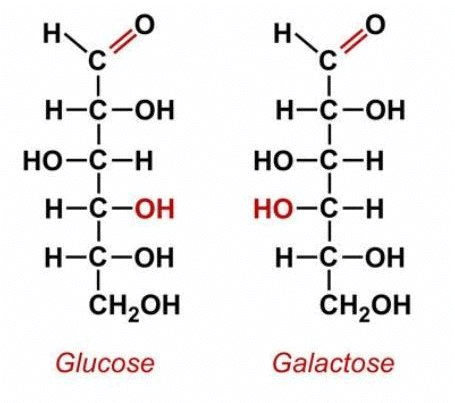
Monosaccharides may be further classified depending on the number of carbon atoms:
- Trioses (C3H6O3): These have three carbon atoms per molecule.
Example: Glyceraldehyde - Tetroses (C4H6O4): These monosaccharides have four carbon atoms per molecule.
Example: Erythrose. - Pentoses,
- Hexoses, and
- Heptoses
2. Disaccharides
Two monosaccharides combine to form a disaccharide. Examples of carbohydrates having two monomers include- Sucrose, Lactose, Maltose, etc.
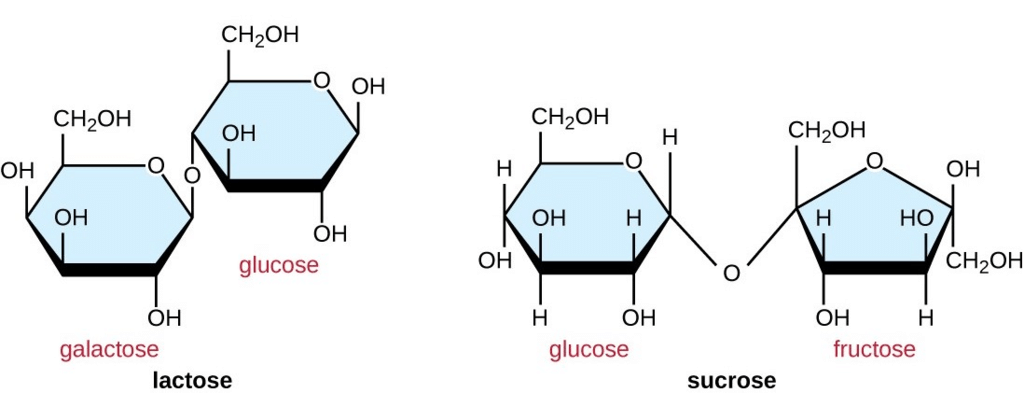
3. Oligosaccharides
Carbohydrates formed by the condensation of 2-9 monomers are called oligosaccharides. By this convention, trioses, pentoses, hexoses are all oligosaccharides.
Complex Carbohydrates (Polysaccharides)
Complex carbohydrates have two or more sugar molecules, hence they are referred to as starchy foods. In complex carbohydrates, molecules are digested and converted slowly compared to simple carbohydrates. They are abundantly found in lentils, beans, peanuts, potatoes, peas, corn, whole-grain bread, cereals, etc.
Polysaccharides are complex carbohydrates formed by the polymerization of a large number of monomers. Examples of polysaccharides include starch, glycogen, cellulose, etc. which exhibit extensive branching and are homopolymers – made up of only glucose units.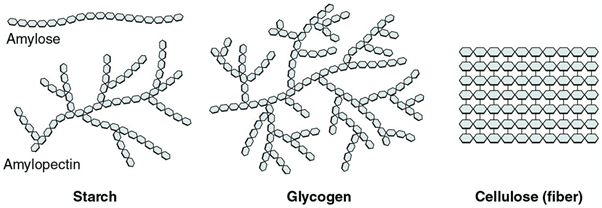
- Starch is composed of two components- amylose and amylopectin. Amylose forms the linear chain and amylopectin is a much-branched chain.
- Glycogen is called animal starch. It has a structure similar to starch but has more extensive branching.
- Cellulose is a structural carbohydrate and is the main structural component of the plant cell wall. It is a fibrous polysaccharide with high tensile strength. In contrast to starch and glycogen, cellulose forms a linear polymer.
Functions of Carbohydrates
The main function of carbohydrates is to provide energy and food to the body and to the nervous system. Carbohydrates are known as one of the basic components of food, including sugars, starch, and fibre which are abundantly found in grains, fruits and milk products. Carbohydrates are also known as starch, simple sugars, complex carbohydrates and so on.
It is also involved in fat metabolism and prevents ketosis. Inhibits the breakdown of proteins for energy as they are the primary source of energy. An enzyme by the name of amylase assists in the breakdown of starch into glucose, finally producing energy for metabolism.
Sources of Carbohydrates
- Simple sugars are found in the form of fructose in many fruits.
- Galactose is present in all dairy products.
- Lactose is abundantly found in milk and other dairy products.
- Maltose is present in cereal, beer, potatoes, processed cheese, pasta, etc.
- Sucrose is naturally obtained from sugar and honey containing small amounts of vitamins and minerals.
These simple sugars that consist of minerals and vitamins exist commonly in milk, fruits, and vegetables. Many refined and other processed foods like white flour, white rice, and sugar, lack important nutrients and hence, they are labelled “enriched.” It is quite healthy to use vitamins, carbohydrates and all other organic nutrients in their normal forms.
Carbohydrate Foods
Eating too much sugar results in an abnormal increase in calories, which finally leads to obesity and in turn low calories leads to malnutrition. Therefore, a well-balanced diet needs to be maintained to have a healthy life. That is the reason a balanced diet is stressed so much by dietitians.
Let us look into the differences between good and bad carbohydrates.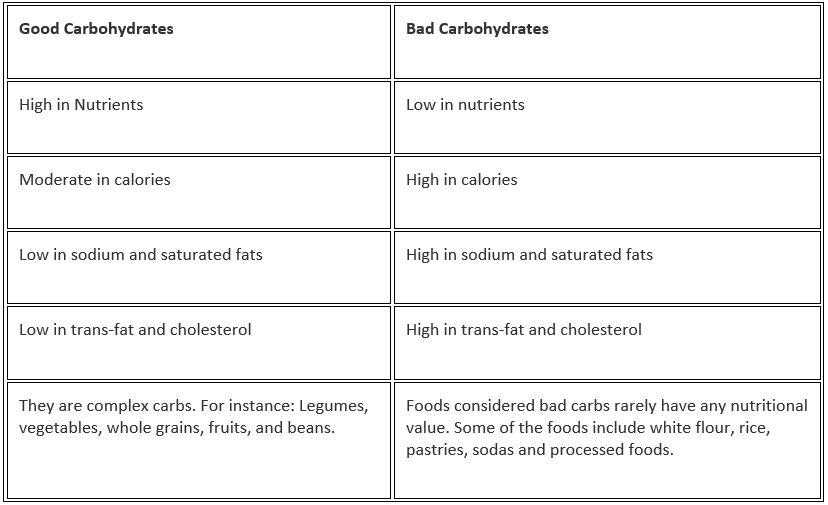
Examples of Carbohydrates
Following are the important examples of carbohydrates:
- Glucose
- Galactose
- Maltose
- Fructose
- Sucrose
- Lactose
- Starch
- Cellulose
- Chitin
Derivatives of Monosaccharides
(1) Amino Sugars
Formed by the displacement of hydroxyl group from second carbon atom by amino group e.g. Glucosamine, Galactosamine.
(2) Sugar Alcohol
Aldehyde group (–CHO) of the sugar is changed to primary alcohol (–CH2OH). Sorbitol and Mannitol are respectively formed from glucose and mannose.
(3) Sugar Acids
They are formed by the oxidation of terminal –CHO or –CH2OH group of sugar to produce carboxyl group –COOH e.g. Glucoronic acid, Galacturonic acid. (4) Glycoside – They are compounds formed by condensation reaction between a sugar (eg. glucose) and hydroxyl group of another substance which may be a sugar, a sterole, methanol in presence of dry HCl. They are acetal which can be hydrolysed by strong reagents like HCN, NH2OH, C6H5NHNH2. They cannot be hydrolysed in acidic condition. Streptomycin is a glycoside.
Oligo – Saccharides
Oligo – Saccharides are those carbohydrates which on hydrolysis yield 2 to 10 monosaccharide units (monomers). In oligosaccharides, monosaccharides are linked together by glycosidic bonds. Aldehyde or ketone group of one monosaccharide reacts with alcoholic group of another monosaccharide to form glycosidic bond. One molecule of H2O eliminates during glycosidic bond formation (dehydration synthesis). Direction of glycosidic bonds is usually 1'.4''.
Types of Oligosaccharides
(i) Disaccharides – composed of two monosaccharide units e.g. Maltose, Sucrose, Lactose, Trehalose.
All disaccharides are water soluble and sweet in taste, so they are known as sugar.
Maltose: is commonly called malt sugar. It is intermediate compound in starch digestion. Maltose has 1'-4'' glycosidic linkage between α-D glucose and α-D glucose.
Lactose: is milk sugar with b-1'-4'' glycosidic linkage between glucose and galactose. It is least sweetest sugar.
Maximum % of lactose = Human milk » 7%
Sucrose: In plants transport of sugar is present in form of sucrose. Sucrose is also known as invert sugar. Sucrose is called Cane Sugar or Table Sugar or Commercial Sugar. Sucrose is composed of α-D Glucose and fructose.
Trehalose: is present in haemolymph of insects. It has glycosidic linkage between two anomeric carbon (α-glucose and β-Glucose).
(ii) Trisaccharides – e.g Raffinose (Galactose + Fructose)
(iii) Tetrasaccharides – e.g Stachyose (Gal. + Gal. + Glu. + Fructose)
(iv) Pentasaccharides – e.g Barbascose (Gal + Gal + Glu + Glu + Fructose)
Raffinose and Stachyose occur in phloem and may be employed for translocation carbohydrates.
Polysaccharides
- Poly saccharides composed of large number of monosaccharide units.
- Suffix '–an' added in their names and they are known as glycans.
- Pentose polysaccharides are called pentosans for e.g. xylan, arabinoxylan.
- Araban (from L-arabinose), xylan (from D-xylose), all these are found in cell wall.
- Hexose polysaccharides are called ''hexans''. for e.g. mannan (from mannose) cellulose, starch etc.
- Polysaccharides are insoluble in water and do not taste sweet.
- All polysaccharides are non-reducing.
- According to function, they are classified as nutritive and structural.
- On structural basis polysaccharides are of two types.
(I) Homopolysaccharides
Composed of same monomers. Biologically important homopolysaccharides are as follows :(a) Cellulose : - Linear polymer of β-D-glucose units (6000 to 10,000) . It has b 1'-4'' linkage. Partial digestion yields a cellobiose units (Disaccharide). Cellulose is main component of plant cell wall. In wood, cellulose is 50% and in cotton, it is 90%. Most abundant organic molecule on earth.
In urochordates, animals their occur cellulose like material and it is called ''Tunicine'' It is also called animal cellulose. It is also used to form Rayon fibre (Artificial silk).
(b) Starch – It is main stored food in plants. Starch is polymer of α-D-glucose units. Starch consists of two types of chains:
(i) Amylose – 250-300 glucose units are arranged in an unbranched chain by a 1'-4'' linkage.
(ii) Amylopectin – A branched chain molecule. Approximately 30 glucose units are linked by α-1' 4'' and α-1', 6'' linkage.
- Amylose gives blue colour with iodine.
- Amylopectin gives red colour with iodine.
- Starch present in potato contains 20% amylose and 80% amylopectin.
(c) Glycogen – Storage form of carbohydrate in animals, Storage region of glycogen is liver and muscles. Storage of glycogen: liver > muscle. Glycogen is also called as animal starch. Glycogen is highly branched polymer of α-D-glucose.
- Glycogen is formed by the 1'-4'' bond linkage at long chain and 1', 6'' bond linkage at branching point.
- Glycogen gives red colour with iodine.
- Glycogen is store food of fungi.
(d) Chitin – Linear polymer of N-acetyl-D-glucosamine with β-1', 4''-linkage.
- N-acetyl D-glucosamine is an amino acyl (-NH-CO-CH3) derivative of β-D-glucose.
- Chitin is an important component of exoskeleton of Arthropods and cell walls of fungi.
- Second most abundant organic molecule on earth.
- It is also called Fungal cellulose.
(e) Inulin – Linear polymer of fructose units linked with β-1', 2'' bonds. Insulin is found in roots of Dahalia and Artichoke. It is water soluble polysaccharide and it is used to know the glomerular filteration rate. It is smallest storage polysaccharide.
(f) Dextrin – Dextrin is an intermediate substance in the digestion of glycogen and starch. By hydrolysis of dextrin, glucose and maltose are formed. It also occurs as stored food in yeast and bacteria.
(II) Heteropolysaccharide
Composed of different monosaccharide units.
(a) Hyaluronic acid – Found in vitreous humour, umbillical cord, joints and connective tissue in the form of lubricating agent. It also occurs in animal cell coat as binding material (Animal cement).
- Hyaluronic acid is made up of D-glucouronic acid and N-acetyl – D-glucosamine arranged in alternate orders. These different monosaccharides have β-1'-4'' bonds and such disaccharides have β -1'- 4" bonds.
(b) Chondriotin – D-glucuronic acid + N-acetyl galactosamine.
- Chondriotin occurs in connective tissue.
- Sulphate ester of chondriotin is main structural component of cartilages, tendons and bones.
(c) Heparin – It is anticoagulant of blood. Heparin is made up of D-glucuronic acid and N-sulphate glucosamine arranged in alternate order.
(d) Pectins – Methylated galacturonic acid + galactose + arabinose.
- Pectin found in cell wall where it binds cellulose fibrils in bundles.
- Salts of pectin i.e Ca and Mg-pectates form middle lamella in plants.
- It is also called Plant cement.
(e) Hemicellulose – Mannose + Galactose + Arabinose + Xylulose.
- Store material – Phytalophus (Ivory palm). Hemicellulose which is obtained from this plant is white, hard and shiny and it is used to form billiard ball and artificial ivory.
Mucopolysaccharides
Slimy polysaccharides with capacity to bind proteins and water are called mucopolysaccharides. In plants, mucilage is a common mucopolysaccharide formed of galactose and mannose units.
Hyaluronic acid, chondriotin, heparin are other examples.
Special Points :
1. Peptidoglycan – Present in cell wall of bacteria.
– Composed of N-acetyl Glucosamine + N-acetyl muramic acid + peptide chain of 4-5 amino acids.
2. Agar-Agar – It is a mucopolysaccharide which is obtained from some red algae – Gracilaria, Gelidium, Chondrus. Its is composed of D-galactose and L-galactose unit and after every 10th unit a sulphate group is present it is us.
Frequently Asked Questions
Q1. What are carbohydrates?
Ans: Carbohydrates are biomolecules comprising carbon, hydrogen and oxygen atoms. They are an important source of energy. They are sugars, starch and fibres found in fruits and vegetables.
Q2. How are carbohydrates classified?
Ans: Carbohydrates are classified into the following:
- Simple carbohydrates
- Complex carbohydrates
Q3. How are carbohydrates important to our body?
Ans: Carbohydrates provide energy to the body. It breaks down into glucose and enters our bloodstream. The body cells utilize glucose to produce ATP.
Q4. Name a few sources of carbohydrates.
Ans: Carbohydrates are obtained from a variety of sources such as bread, milk, potatoes, cookies, corn, etc.
Q5. How are the carbohydrates digested?
Ans: Carbohydrates start being digested in the mouth by the action of salivary amylase. They are not completely broken down in the stomach but in the intestine.
Q6. What are simple carbohydrates? Give examples.
Ans: Simple carbohydrates are the ones that are quickly broken down by the body to be converted into energy. Fruits, milk and milk products are the main sources of simple carbohydrates.
Q7. How are complex carbohydrates different from simple carbohydrates?
Ans: Complex carbohydrates are the ones in which the sugar molecules are strung in long, complex chains. Peas, beans, vegetables and grains are important sources of carbohydrates.
Q8. What are the three types of simple carbohydrates?
Ans: Three types of simple carbohydrates include:
- Monosaccharides
- Disaccharides
- Polysaccharides
Q9. Name some bad carbohydrates that are harmful to the body.
Ans: The bad carbs include:
- White bread
- Sugary drinks
- Pastries
- Candies and chocolates
|
150 videos|398 docs|136 tests
|
FAQs on Carbohydrates - Biology Class 11 - NEET
| 1. What are carbohydrates? |  |
| 2. How are carbohydrates classified? |  |
| 3. What are the functions of carbohydrates in the body? |  |
| 4. What are some sources of carbohydrates? |  |
| 5. Which foods are high in carbohydrates? |  |

















