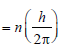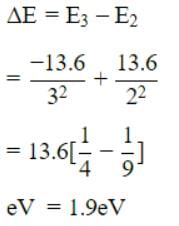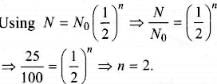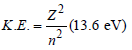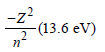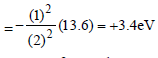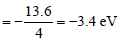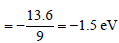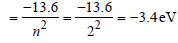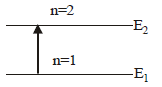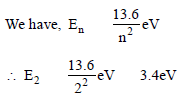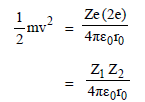31 Year NEET Previous Questions: Atoms - Free MCQ Test with solutions [Updated]
MCQ Practice Test & Solutions: 31 Year NEET Previous Year Questions: Atoms (40 Questions)
You can prepare effectively for NEET Physics Class 12 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "31 Year NEET Previous Year Questions: Atoms". These 40 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 80 minutes
- - Number of Questions: 40
Sign up on EduRev for free to attempt this test and track your preparation progress.
What is the radius of iodine atom (At. no. 53,mass no. 126) [1988]
Detailed Solution: Question 1
The ionisation energy of hydrogen atom is 13.6eV, the ionisation energy of helium atom wouldbe [1988]
Detailed Solution: Question 2
To explain his theory, Bohr used [1989]
Detailed Solution: Question 3
The ground state energy of H-atom 13.6 eV. Theenergy needed to ionize H-atom from its secondexcited state. [1991]
Detailed Solution: Question 4
In terms of Bohr radius a0, the radius of the second Bohr orbit of a hydrogen atom is given by [1992]
Detailed Solution: Question 5
The ionization energy of hydrogen atom is 13.6eV. Following Bohr’s theory, the energycorresponding to a transition between 3rd and4th orbit is [1992]
Detailed Solution: Question 6
Which source is associated with a line emissionspectrum ? [1993]
Detailed Solution: Question 7
In Rutherford scattering experiment, what willbe the correct angle for α-scattering for an impactparameter, b = 0 ? [1994]
Detailed Solution: Question 8
The radius of hydrogen atom in its ground stateis 5.3 × 10–11 m. After collision with an electron itis found to have a radius of 21.2 × 10–11 m. Whatis the principal quantum number n of the finalstate of the atom [1994]
Detailed Solution: Question 9
The spectrum obtained from a sodium vapourlamp is an example of [1995]
Detailed Solution: Question 10
When a hydrogen atom is raised from the groundstate to an excited state, [1995]
Detailed Solution: Question 11
If the threshold wavelength for a certain metal is 2000 Å, then the work-function of the metal is
Detailed Solution: Question 12
When hydrogen atom is in its first excited level,its radius is [1997]
Detailed Solution: Question 13
In the Bohr model of a hydrogen atom, the centripetal force is furnished by the coulomb attraction between the proton and the electron. If a0 is the radius of the ground state orbit, m is the mass, e is the charge on the electron and ε0 is the vacuum ermittivity, the speed of the electron is [1998]
Detailed Solution: Question 14
Who indirectly determined the mass of theelectron by measuring the charge of the electron? [2000]
Detailed Solution: Question 15
When an electron jumps from the fourth orbit tothe second orbit, one gets the [2000]
Detailed Solution: Question 16
Which of the following transitions in a hydrogenatom emits the photon of highest frequency? [2000]
Detailed Solution: Question 17
An electron changes its position from orbit n = 2 to the orbit n = 4 of an atom. The wavelength of the emitted radiations is (R = Rydberg’s constant)
Detailed Solution: Question 18
The energy of hydrogen atom in nth orbit is En, then the energy in nth orbit of single ionised helium atom will be [2001]
Detailed Solution: Question 19
J.J. Thomson’s cathode-ray tube experimentdemonstrated that [2003]
Detailed Solution: Question 20
In which of the following systems will the radiusof the first orbit (n = 1) be minimum ? [2003]
Detailed Solution: Question 21
Detailed Solution: Question 22
Energy E of a hydrogen atom with principal quantum number n is given by E = – 13.6/n2 eV. The energy of photon ejected when the electronjumps from n = 3 state to n = 2 state of hydrogenis approximately [2004]
Detailed Solution: Question 23
The half life of radium is about 1600 years. Of 100 g of radium existing now, 25 g will remainunchanged after [2004]
Detailed Solution: Question 24
The total energy of an electron in the firstexcited state of hydrogen atom is about –3.4eV. Its kinetic energy in this state is [2005]
Detailed Solution: Question 25
Ionization potential of hydrogen atom is 13.6eV.Hydrogen atoms in the ground state are excitedby monochromatic radiation of photon energy12.1 eV. According to Bohr’s theory, the spectrallines emitted by hydrogen will be [2006]
Detailed Solution: Question 26
The total energy of electron in the ground stateof hydrogen atom is – 13.6 eV. The kinetic energyof an electron in the first excited state is [2007]
Detailed Solution: Question 27
The ground state energy of hydrogen atom is 13.6eV. When its electron is in the first excited state, its excitation energy is [2008]
Detailed Solution: Question 28
In a Rutherford scattering experiment when a projectile of charge Z1 and mass M1 approaches a target nucleus of charge Z2 and mass M2, the distance of closest approach is r0. The energy of the projectile is [2009]
Detailed Solution: Question 29
The ionization energy of the electron in thehydrogen atom in its ground state is 13.6 eV.The atoms are excited to higher energy levels toemit radiations of 6 wavelengths. Maximumwavelength of emitted radiation corresponds tothe transition between [2009]
Detailed Solution: Question 30
74 videos|347 docs|82 tests |


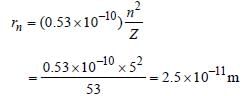

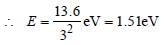
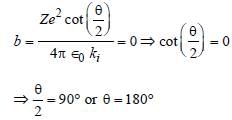
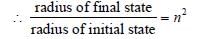
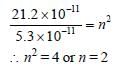
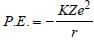 and
and 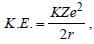 where,
where,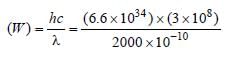

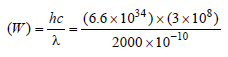
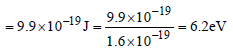

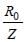
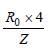
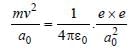
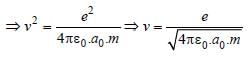
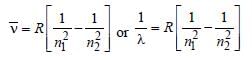
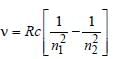
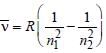 , where n1 = 2, n2 = 4
, where n1 = 2, n2 = 4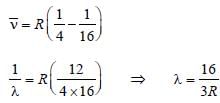
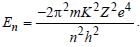 For helium
For helium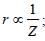 Z(=3) is maximum for Li2+.
Z(=3) is maximum for Li2+.