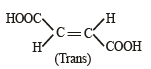31 Year NEET Previous Questions: Some Basic Principles & Techniques - 1
MCQ Practice Test & Solutions: 31 Year NEET Previous Year Questions: Some Basic Principles & Techniques - 1 (45 Questions)
You can prepare effectively for NEET Chemistry Class 11 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "31 Year NEET Previous Year Questions: Some Basic Principles & Techniques - 1". These 45 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 45 minutes
- - Number of Questions: 45
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which one of the following reactions does not belong to "Lassaigne's test"? [NEET 2025]
Detailed Solution: Question 1
Match List-I with List-II [NEET 2025]
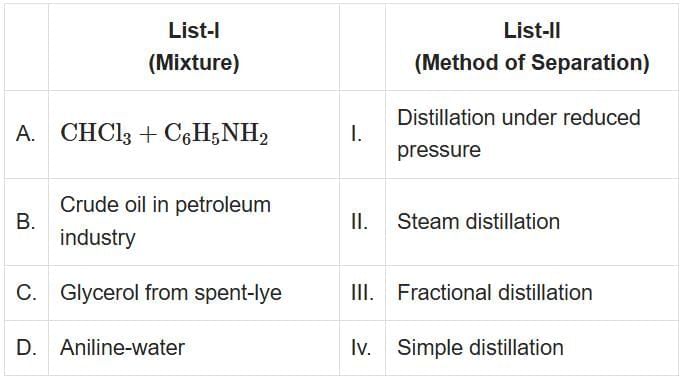
Choose the correct answer from the option given below:

Detailed Solution: Question 2
The most stable carbocation among the following is: [NEET 2024]
Detailed Solution: Question 3
On heating, some solid substances change from solid to vapour state without passing through liquid state. The technique used for the purification of such solid substances based on the above principle is known as [NEET 2024]
Detailed Solution: Question 4
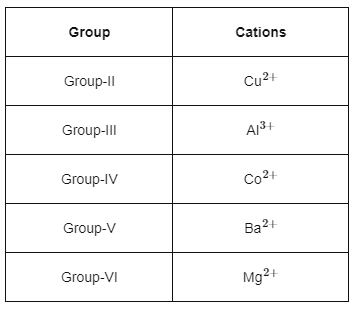
Given below are certain cations. Using inorganic qualitative analysis, arrange them in increasing group number from 0 to VI. [NEET 2024]
A. Al3+
B. Cu2+
C. Ba2+
D. Co2+
E. Mg2+
Choose the correct answer from the options given below:
A. Al3+
B. Cu2+
C. Ba2+
D. Co2+
E. Mg2+
Choose the correct answer from the options given below:
Detailed Solution: Question 5
The number of σ bonds, π bonds and lone pair of electrons in pyridine, respectively are: [NEET 2023]
Detailed Solution: Question 6
In Lassaigne’s extract of an organic compound, both nitrogen and sulphur are present, which gives blood red colour with Fe3+ due to the formation of [NEET 2023]
Detailed Solution: Question 7
Amongst the given options which of the following molecules/ion acts as a Lewis acid? [NEET 2023]
Detailed Solution: Question 8
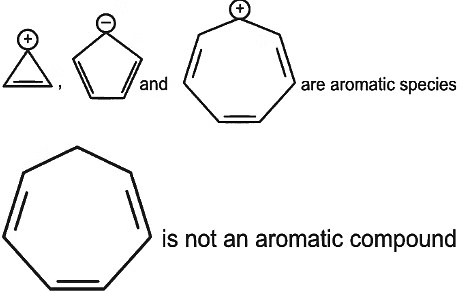
Consider the following compounds/species: [NEET 2023]
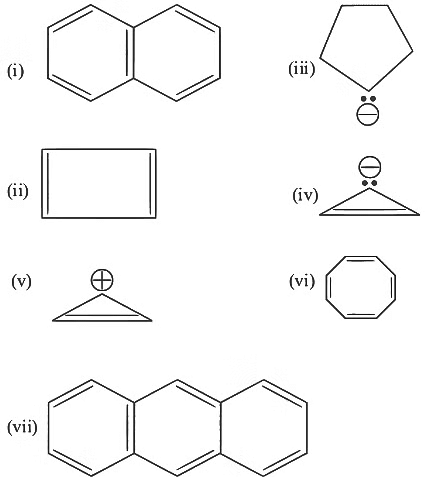
The number of compounds/species which obey Huckel's rule is ___________.

Detailed Solution: Question 9
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : Chlorine is an electron withdrawing group but it is ortho, para directing in electrophilic aromatic substitution.
Reason (R) : Inductive effect of chlorine destabilises the intermediate carbocation formed during the electrophilic substitution, however due to the more pronounced resonance effect, the halogen stabilises the carbocation at ortho and para positioins.
In the light of the above statements, choose the most appropriate answer from the options given below : [NEET 2022 Phase 2]
Assertion (A) : Chlorine is an electron withdrawing group but it is ortho, para directing in electrophilic aromatic substitution.
Reason (R) : Inductive effect of chlorine destabilises the intermediate carbocation formed during the electrophilic substitution, however due to the more pronounced resonance effect, the halogen stabilises the carbocation at ortho and para positioins.
In the light of the above statements, choose the most appropriate answer from the options given below : [NEET 2022 Phase 2]
Detailed Solution: Question 10
Predict the order of reactivity of the following four isomers towards SN2 reaction. [NEET 2022 Phase 2]
(I) CH3CH2CH2CH2Cl
(II) CH3CH2CH(Cl)CH3
(III) (CH3)2CHCH2Cl
(IV) (CH3)3CCl
(I) CH3CH2CH2CH2Cl
(II) CH3CH2CH(Cl)CH3
(III) (CH3)2CHCH2Cl
(IV) (CH3)3CCl
Detailed Solution: Question 11
What is the hybridization shown by C1 and C2 carbons, respectively in the given compound?
OHC − CH = CH − CH2COOCH3 [NEET 2022 Phase 2]
OHC − CH = CH − CH2COOCH3 [NEET 2022 Phase 2]
Detailed Solution: Question 12
The Kjeldahl's method for the estimation of nitrogen can be used to estimate the amount of nitrogen in which one of the following compounds? [NEET 2022 Phase 1]
Detailed Solution: Question 13
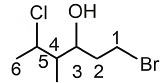
The correct IUPAC name of the following compound is: [NEET 2022 Phase 1]
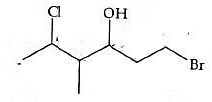

Detailed Solution: Question 14
The correct IUPAC name of the following compound is: [NEET 2022 Phase 1]


Detailed Solution: Question 15
The incorrect statement regarding chirality is [(NEET 2022 Phase 1]
Detailed Solution: Question 16
Which compound amongst the following is not an aromatic compound? [NEET 2022 Phase 1]
Detailed Solution: Question 17
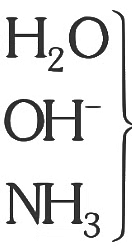
Nitrogen detection in an organic compound is carried out by Lassaigne’s test. The blue colour formed corresponds to which of the following formulae? [NEET Kar. 2013]
Detailed Solution: Question 18
Which of the following compounds undergoes nucleophilic substitution reaction most easily ?
Detailed Solution: Question 19
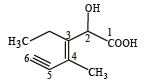
Structure of the compound whose IUPAC name is 3-ethyl-2-hydroxy-4-methylhex-3-en-5-ynoic acid is : [NEET 2013]
Detailed Solution: Question 20
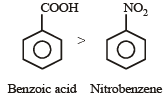
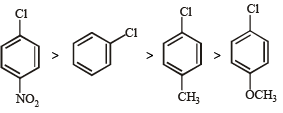
Some meta-directing substituents in aromatic substitution are given. Which one is most deactivating? [NEET 2013]
Detailed Solution: Question 21
Which of the following compounds will not undergo Friedal-Craft’s reaction easily : [NEET 2013]
Detailed Solution: Question 22
The structure of isobutyl group in an organic compound is : [NEET 2013]
Detailed Solution: Question 23
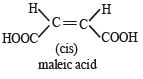
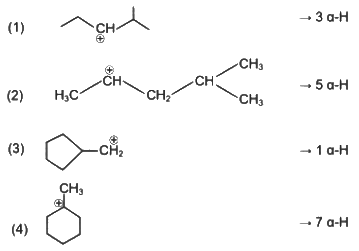
Arr ange the following in increasing order of stability [NEET Kar. 2013]
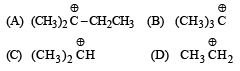
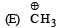
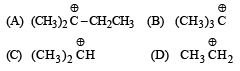
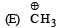
Detailed Solution: Question 24
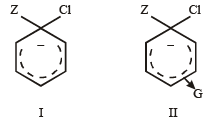
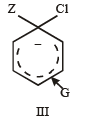
Given
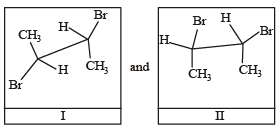
I and II are [NEET Kar. 2013]

Detailed Solution: Question 25
Homolytic fission of the following alkanes forms free radicals CH3 – CH3, CH3 – CH2 – CH3, (CH3)2 CH – CH3, CH3 – CH2 – CH (CH3)2. Increasing order of stability of the radicals is [NEET Kar. 2013]
Detailed Solution: Question 26
What is the hybridisation state of benzyl carbonium [NEET Kar. 2013]


Detailed Solution: Question 27
Which of the following acids does not exhibit optical isomerism ? [2012]
Detailed Solution: Question 28
Which nomenclature is not according to IUPAC system? [2012]
Detailed Solution: Question 29
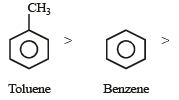
Among the following compounds the one that is most reactive towards electrophilic nitration is: [2012]
Detailed Solution: Question 30
98 videos|243 docs|71 tests |



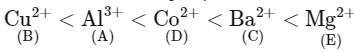

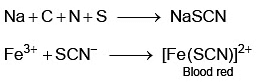
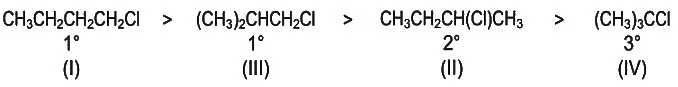
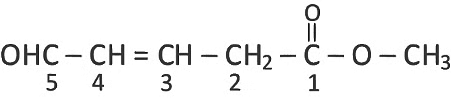
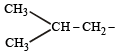 (iso-butyl group)
(iso-butyl group)