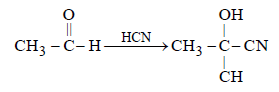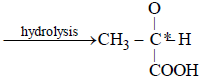31 Year NEET Previous Year Questions: Aldehydes, Ketones & Carboxylic Acids - 1 - NEET MCQ
30 Questions MCQ Test - 31 Year NEET Previous Year Questions: Aldehydes, Ketones & Carboxylic Acids - 1
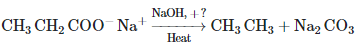
Consider the above reaction and identify the missing reagent/chemical. [2021]
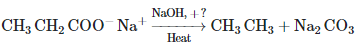
Consider the above reaction and identify the missing reagent/chemical. [2021]
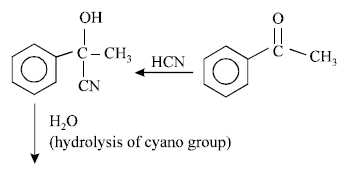
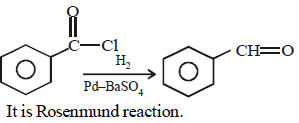
Reaction by which Benzaldehyde cannot be prepared : [NEET 2013]
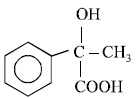
Which of the following compounds will give ayellow precipitate with iodine and alkali ?[2012 M]
Consider the reaction :
RCHO + NH2NH2 → RCH = N – NH2
What sort of reaction is it ? [2012 M]
Consider the following reaction :
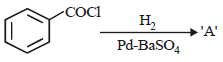
The product ‘A’ is : [2012 M]
CH3CHO and C6H5CH2CHO can be distinguished chemically by: [2012]
The correct order of decreasing acid strength oftrichloroacetic acid (A), trifluoroacetic acid (A),acetic acid (C) and formic acid (D) is : [2012]
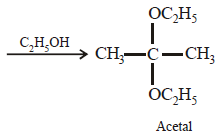
Acetone is treated with excess of ethanol in the presence of hydrochloric acid. The product obtained is : [2012]
Predict the product in the given reaction. [2012]
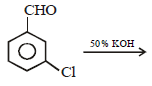
An organic compound ‘A’ on treatment with NH3 gives ‘B’ which on heating gives ‘C’, ‘C’ whentreated with Br2 in the presence of KOH produces ethylamine. Compound ‘A’ is:[2011 M]
Match the compounds given in List-I with List-II and select the suitable option using the code given below : [2011 M]


The order of reactivity of phenyl magnesium bromide (PhMgBr) with the following compounds [2011 M]
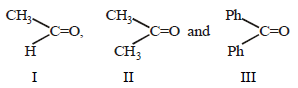
Clemmensen reduction of a ketone is carried outin the presence of which of the following ? [2011]
Which one of the following compounds will be most readily dehydrated? [2010]
Following compounds are given:
(a) CH3CH2OH
(b) CH3COCH3
(c) 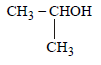
(d) CH3OH
Which of the above compound(s), on being warmed with iodine solution and NaOH, will give iodoform? [2010]
Which of the following reactions will not resultin the formation of carbon-carbon bonds?
In a set of reactions, ethylbenzene yielded a product D. [2010]
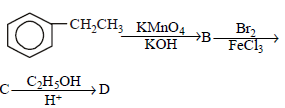
Propionic acid with Br2|P yields a dibromo product. Its structure would be: [2009]
The relative reactivities of acyl compounds towardsnucleophilic substitution are in the order of : [2008]
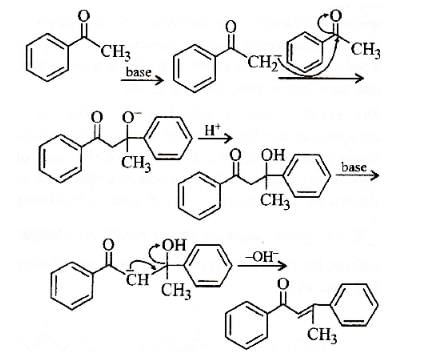
Acetophenone when reacted with a base, C2H5ONa, yields a stable compound which has the structure. [2008]
The product formed in Aldol condensation is [2007]
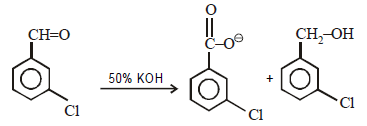
Which one of the following on treatment with 50% aqueous sodium hydroxide yields the corresponding alcohol and acid? [2007]
Which of the following represents the correctorder of the acidity in the given compounds? [2007]
Reduction of aldehydes and ketones intohydrocarbons using zinc amalgam and conc. HClis called [2007]
In a set of reactions propionic acid yielded a compound D. [2006]
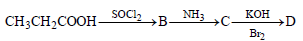
The structure of D would be
Self condensation of two moles of ethyl acetatein presence of sodium ethoxide yields [2006]
Nucleophilic addition reaction will be mostfavoured in [2006]
A carbonyl compound reacts with hydrogencyanide to form cyanohydrin which on hydrolysisforms a racemic mixture of α-hydroxy acid. Thecarbonyl compound is [2006]
In a set of reactions acetic acid yielded a product D.
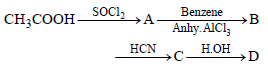
The structure of D would be: [2005]
The order of stability of the following tautomeric compounds is :
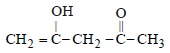
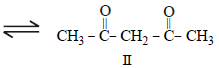
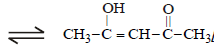



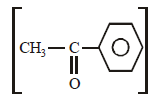 and 2-Hydroxypropane
and 2-Hydroxypropane 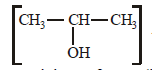 both give a yellow
both give a yellow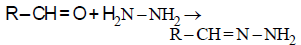
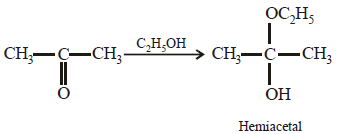
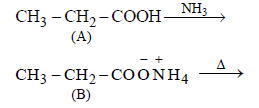
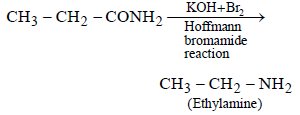
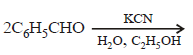
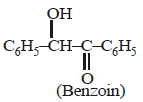
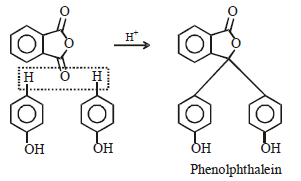
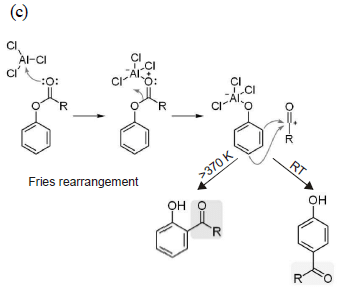
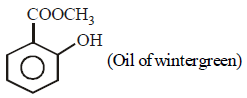
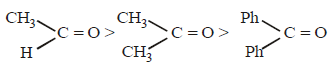
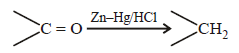
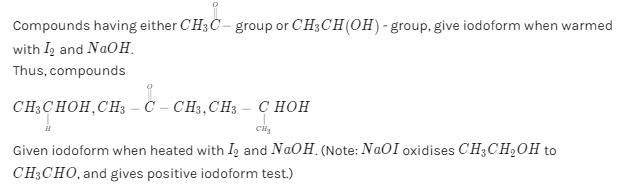
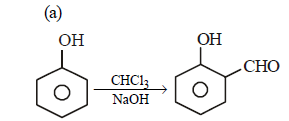
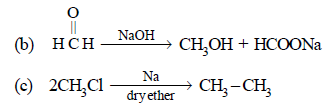
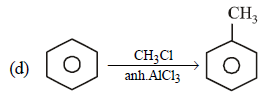
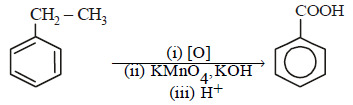
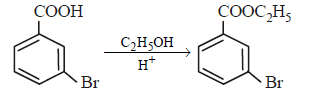
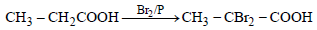
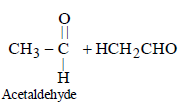
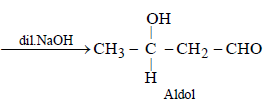
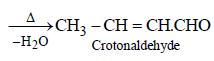
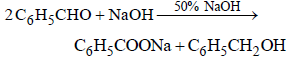
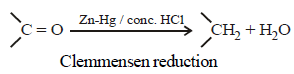
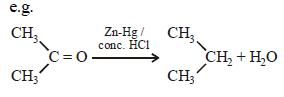
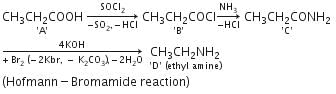 Hence, compound 'D' is CH3-CH2-NH2
Hence, compound 'D' is CH3-CH2-NH2