IUPAC Nomenclature of Coordination Compounds - NEET Chemistry Class 12
MCQ Practice Test & Solutions: Test: IUPAC Nomenclature of Coordination Compounds (25 Questions)
You can prepare effectively for NEET Chemistry Class 12 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: IUPAC Nomenclature of Coordination Compounds". These 25 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 35 minutes
- - Number of Questions: 25
Sign up on EduRev for free to attempt this test and track your preparation progress.
Direction (Q. Nos. 1-10) This section contains 10 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONLY ONE is correct.
Q. According to IUPAC nomenclature, sodium nitroprusside is named as
Detailed Solution: Question 1
The formula of the complex hexamminecobalt (III) chloride sulphate is
Detailed Solution: Question 2
The IUPAC name of the compound [Cr(NH3)5(NCS)][ZnCI4] is
Detailed Solution: Question 3
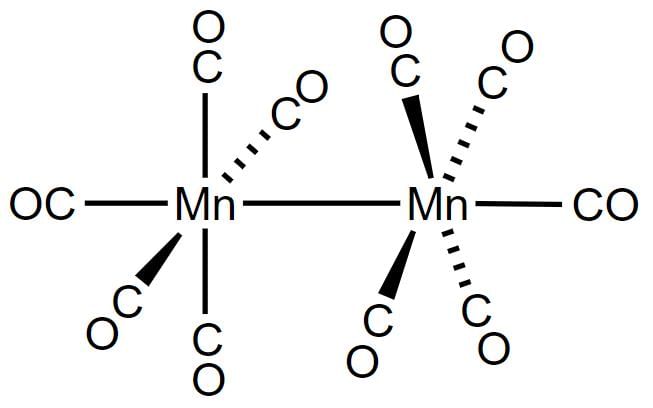
The IUPAC name of the complex [(CO)5Mn - Mn(CO)5] is
Detailed Solution: Question 4
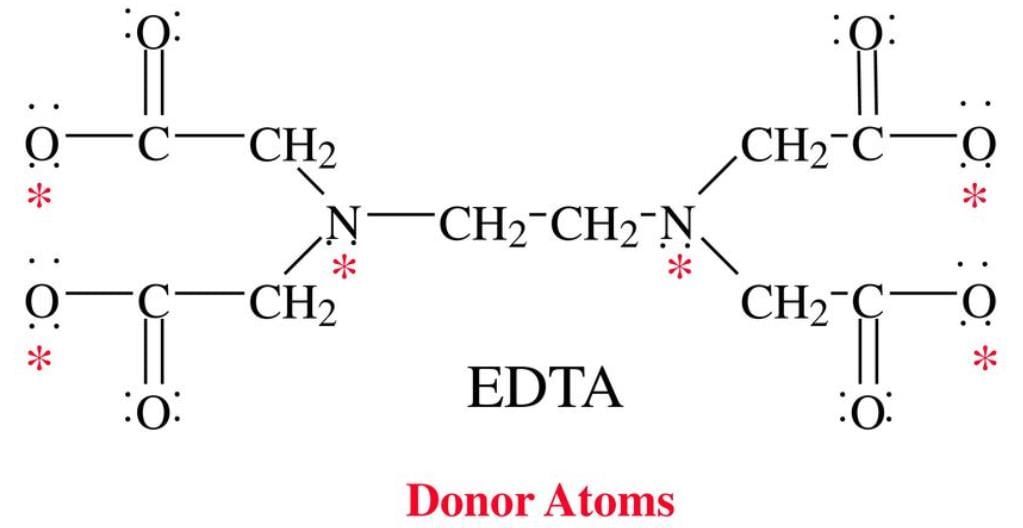
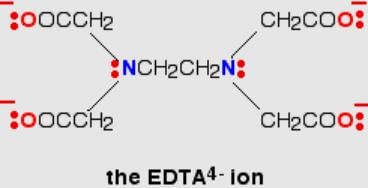
The donor sites of EDTA ligand are
Detailed Solution: Question 5
The IUPAC name of [Ni(PPh3)2CI2]2+ is
Detailed Solution: Question 6
Formula of brown ring complex is
Detailed Solution: Question 7
The IUPAC name of the compound K[SbCl5Ph] is
Detailed Solution: Question 8
Detailed Solution: Question 9
What is the IUPAC name of compound NaBH4?
Detailed Solution: Question 10
Direction (Q. Nos. 11-15) This section contains 5 multiple choice questions. Each question has four choices (a), (b), (c) and (d), out of which ONE or MORE THAN ONE are correct.
Q.
Which of the following complexes have correct name?
Detailed Solution: Question 11
Which is/are the correctly named?
Detailed Solution: Question 12
The correct name of ligand(s) in IUPAC name is/are
Detailed Solution: Question 13
The ionisation isomer of [Cr(H2O)4Cl(NO2)Cl] is
Detailed Solution: Question 14
The ligand(s) which gives chelate complexes is/are
Detailed Solution: Question 15
Comprehension Type
Direction (Q. Nos. 16 and 17) This section contains a paragraph, describing theory, experiments, data, etc. Two questions have been related to the paragraph have been given. Each question has only one correct answer among the four given options (a), (b), (c) and (d).
Passage
In naming complexes, cation is named first followed by anion. Non-ionic or neutral complexes are given a one word name. The cationic and neutral complex ions are named by writing ‘the number and name of the ligands followed by the name of the central metal ion with its oxidation number in roman numerals within the parenthesis. If the complex is anionic, name of the metals ends with ‘ate’. When the complex contain two or more metal atoms linked with the help of bridging ligand then each bridging ligand is started with μ.
Q.
Name of [CoBr(NH3)4 NO2] is
Detailed Solution: Question 16
In naming complexes, cation is named first followed by anion. Non-ionic or neutral complexes are given a one word name. The cationic and neutral complex ions are named by writing ‘the number and name of the ligands followed by the name of the central metal ion with its oxidation number in roman numerals within the parenthesis. If the complex is anionic, name of the metals ends with ‘ate’. When the complex contain two or more metal atoms linked with the help of bridging ligand then each bridging ligand is started with μ.
Q.
The IUPAC name of [Ni(NH3)4] [NiCI4] is
Detailed Solution: Question 17
Matching List Type
Direction (Q. Nos. 18 and 19) Choices for the correct combination of elements from Column I and Column II are given as options (a), (b), (c) and (d), out of which one is correct.
Q.
Match the Column I with Column II and mark the correct option from the codes given below.
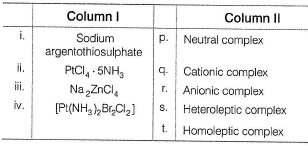
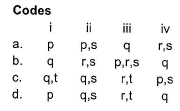
Detailed Solution: Question 18
Match the column I with column II and mark the correct option from the codes given below.
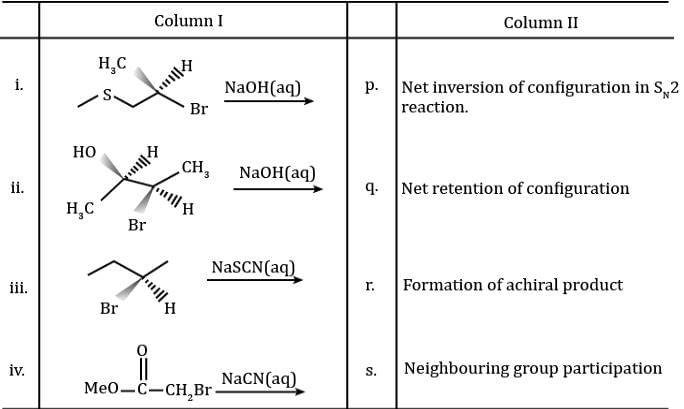

Detailed Solution: Question 19
One Integer Value Correct Type
Direction (Q. Nos. 20-24) This section contains 5 questions. When worked out will result in an integer from 0 to 9 (both inclusive).
Q. Ethylenediamminetetraacetate ion is a polydentate ligand and negatively charged. The magnitude of negative charge is
Detailed Solution: Question 20
The oxidation state of the central metai in the complex ion [Fe(EDTA)]- .
Detailed Solution: Question 21
The complex potassium dicyanodioxalatonickelate (II) in solution produce....... ions.
Detailed Solution: Question 22
Number of EDTA molecules required to form an octahedral complex.
Detailed Solution: Question 23
IUPAC name of [Pt(NH3)2Cl(NO2)] is
Detailed Solution: Question 24
Statement Type
Direction (Q. No. 25) This section is based on Statement I and Statement II. Select the correct answer from the codes given below.
Q.
Statement l : IUPAC name of the complex [(NH3)5—Cr—OH—Cr (NH3)5]CI5 is pentammine chromium-μ-hydroxopentammine chromium (III) chloride.
Statement II : IUPAC name of the complex [Ni(dmg)2] is bis (dimethylglyoximato) nickel (III).
Detailed Solution: Question 25
54 videos|290 docs|74 tests |