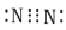Carbon & its Compounds - 1 - Free MCQ Test with solutions for UPSC Sci
MCQ Practice Test & Solutions: Carbon & its Compounds - 1 (10 Questions)
You can prepare effectively for UPSC Science & Technology for UPSC CSE with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Carbon & its Compounds - 1". These 10 questions have been designed by the experts with the latest curriculum of UPSC 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 10 minutes
- - Number of Questions: 10
Sign up on EduRev for free to attempt this test and track your preparation progress.
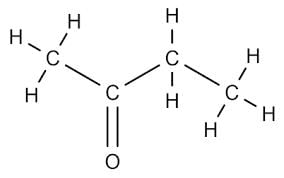
Butanone is a four-carbon compound with the functional group:
Detailed Solution: Question 1
Detailed Solution: Question 2
An organic compound X with molecular formula C2H4O2 turns blue litmus red and gives brisk effervescence with sodium bicarbonate. Identify the compound?
Detailed Solution: Question 3
Chlorine reacts with saturated hydrocarbons at room temperature in the:
Detailed Solution: Question 4
Detailed Solution: Question 5
Which of the following is the correct representation of electron dot structure of nitrogen?
Detailed Solution: Question 6
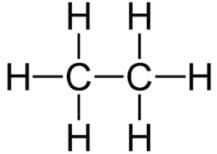
Which of the following organic compounds does not have the same chemical properties as methanol?
Detailed Solution: Question 7
Oils on treating with hydrogen in the presence of palladium or nickel catalyst form fats. This is an example of:
Detailed Solution: Question 8
Detailed Solution: Question 9
CH3CH2OH 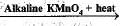 CH3COOH
CH3COOH
In the above given reaction, alkaline KMnO4 acts as
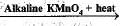 CH3COOH
CH3COOH Detailed Solution: Question 10
67 videos|481 docs|179 tests |