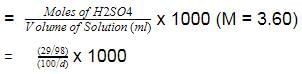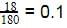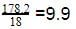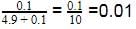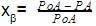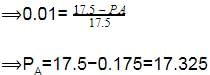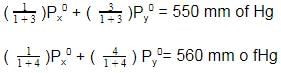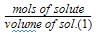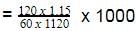JEE Previous Year Questions- Solutions with - Free MCQ Test Attempt
MCQ Practice Test & Solutions: Test: JEE Previous Year Questions- Solutions (26 Questions)
You can prepare effectively for JEE Chapter-wise Tests for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: JEE Previous Year Questions- Solutions". These 26 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 35 minutes
- - Number of Questions: 26
Sign up on EduRev for free to attempt this test and track your preparation progress.
Which of the following concentration factor is affected by change in temperature ?
[AIEEE-2002]
Detailed Solution: Question 1
For an aqueous solution, freezing point is _0.186ºC . Boiling point of the same solution is
(Kƒ = 1.86º K mol-1 kg) and Kb = 0.512º K mol-1 kg)
[AIEEE-2002]
Detailed Solution: Question 2
In a mixture of A and B, components show negative deviation when [AIEEE-2002]
Detailed Solution: Question 3
A pressure cooker reduces cooking time for food because _
[AIEEE-2003]
Detailed Solution: Question 4
If liquids A and B form an ideal solution _
[AIEEE-2003]
Detailed Solution: Question 5
In a 0.2 molal aqueous solution of a weak acid HX the degree of ionization is 0.3 . Taking kƒ for water as 1.85, the freezing point of the solution will be nearest to _
[AIEEE-2003]
Detailed Solution: Question 6
If liquid A and B form ideal solution, than
[AIEEE-2003]
Detailed Solution: Question 7
Which one of the following aqueous solutions will exhibit highest boiling point ?
[AIEEE-2004]
Detailed Solution: Question 8
Osmotic pressure of 40% (wt./vol.) urea solution is 1.64 atm and that of 3.42% (wt./vol.) cane sugar is 2.46 atm. When equal volumes of the above two solutions are mixed, the osmotic pressure of the resulting solution is -
Detailed Solution: Question 9
Which of the following liquid pairs shows a positive deviation from Raoult's law ?
[AIEEE-2004]
Detailed Solution: Question 10
Which one of the following statement is false?
[AIEEE-2004]
Detailed Solution: Question 11
If a is the degree of dissociation of Na2SO4, the vant Hoff's factor (i) used for calculating the molecular mass is–
[AIEEE-2005]
Detailed Solution: Question 12
Benzene and toluene form nearly ideal solutions. At 20ºC, the vapour pressure of benzene is 75 torr and that of toluene is 22 torr. The partial pressure of benzene at 20ºC for a solution containing 78 g of benzene and 46 g of toluene in torr is
[AIEEE-2005]
Detailed Solution: Question 13
Two solutions of a substance (non electrolyte) are mixed in the following manner. 480 ml of 1.5 M first solution + 520 mL of 1.2 M second solution. What is the molarity of the final mixture ?
[AIEEE-2005]
Detailed Solution: Question 14
Which one of the following aqueous solutions will exhibit highest boiling point?
Detailed Solution: Question 15
18 g of glucose (C6H12O6) is added to 178.2 g of water. The vapour pressure of water for this aqueous solution at 100º C is -
[AIEEE 2006]
Detailed Solution: Question 16
Density of a 2.05 M solution of acetic acid in water is 1.02 g/mL. The molality of the solution is -
[AIEEE 2006]
Detailed Solution: Question 17
A mixture of ethyl alcohol and propyl alcohol has a vapour pressure of 290 mm at 300 K. The vapour pressure of propyl alcohol is 200 mm. If the mole fraction of ethyl alcohol is 0.6, its vapour pressure (in mm) at the same temperature will be -
[AIEEE 2007]
Detailed Solution: Question 18
A 5.25% solution of a substance is isotonic with a 1.5% solution of urea (molar mass = 60 g mol-1) in the same solvent. If the densities of both the solutions are assumed to be equal to 1.0 gcm-3, molar mass of the substance will be-
[AIEEE 2007]
Detailed Solution: Question 19
The density (in g mL-1) of a 3.60 M sulphuric acid solution that is 29% H2SO4(Molar mass = 98 g mol-1) by mass will be -
[AIEEE 2007]
Detailed Solution: Question 20
The vapour pressure of water at 20º C is 17.5 mm Hg. If 18g of glucose (C6H12O6) is added to 178.2 g of water at 20° C, the vapour pressure of the resulting solution will be –
[AIEEE 2008]
Detailed Solution: Question 21
At 80º C , the vapour pressure of pure liquid `A' is 520 mm Hg and that of pure liquid `B' is 1000 mm Hg. If a mixture solution of `A' and `B' boils at 80º C and 1 atm pressure, the amount of `A' in the mixture is (1 atm = 760 mm Hg)
[AIEEE 2008]
Detailed Solution: Question 22
A binary liquid solution is prepared by mixing n-heptane and ethanol. Which one of the following statements is correct regarding the behaviour of the solution ?
[AIEEE 2009]
Detailed Solution: Question 23
Two liquids X and Y form an ideal solution At 300 K, vapour pressure of the solution containing 1 mol of X and 3 mol of Y is 550 mmHg. At the same temperature, if 1 mol of Y is further added to this solution, vapour pressure of the solution increases by 10 mmHg. Vapour pressure (in mmHg) of X and Y in their pure states will be, respectively -
[AIEEE 2009]
Detailed Solution: Question 24
Kf for water is 1.86 K kg mol-1. If your automobile radiator holds 1.0 kg of water, how many grams of ethylene glycol (C2H6O2) must you add to get the freezing point of the solution lowered to – 2.8°C ?
[AIEEE-2012]
Detailed Solution: Question 25
The density of a solution prepared by dissolving 120 g of urea (mol. mass = 60 u) in 1000g of water is 1.15 g/mL. The molarity of this solution is :
[AIEEE-2012]
Detailed Solution: Question 26
446 docs|929 tests |




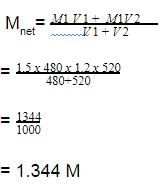
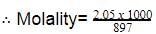
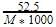 The molar concentration of urea solution is
The molar concentration of urea solution is 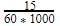
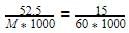
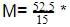 60 = 210.0 g/mol.
60 = 210.0 g/mol.