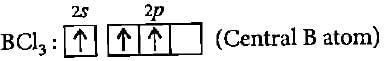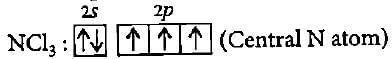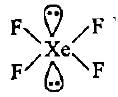The Valence Shell Electron Pair Repulsion Theory (NCERT) - Free MCQ Practice
MCQ Practice Test & Solutions: Test: The Valence Shell Electron Pair Repulsion Theory (NCERT) (15 Questions)
You can prepare effectively for NEET Chemistry Class 11 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: The Valence Shell Electron Pair Repulsion Theory (NCERT)". These 15 questions have been designed by the experts with the latest curriculum of NEET 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 15 minutes
- - Number of Questions: 15
Sign up on EduRev for free to attempt this test and track your preparation progress.
According to VSEPR theory
Detailed Solution: Question 1
In a bonded molecule, the order of repulsion between the bonded and non-bonded electrons is
Detailed Solution: Question 2
The shape of water molecule which should be tetrahedral has a bent or distorted tetrahedral shape with a bond angle 104.5°. What could be the reason for this?
Detailed Solution: Question 3
Which of the following shapes of SF4 is more stable and why?
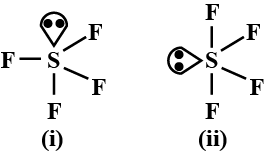
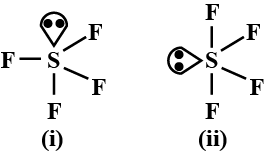
Detailed Solution: Question 4
The most stable shape of ClF3 is shown by
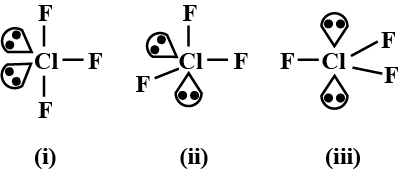
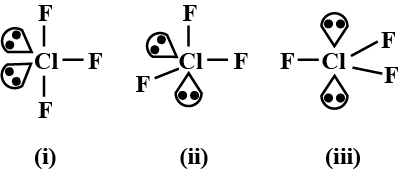
Detailed Solution: Question 5
In which of the following molecules the central atomdoes not retain any lone pair of electrons?
Detailed Solution: Question 6
Few examples of the compounds formed by chemical bonding are given below. Mark the incorrect example.
Detailed Solution: Question 7
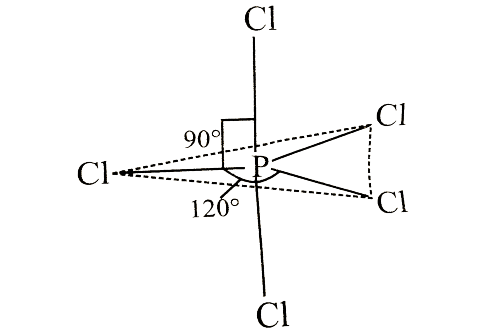
Which of the following statements is correct regarding the structure of PCI5?
Detailed Solution: Question 8
Match the molecules given in column I with their shapes given in column II and mark the appropriate choice
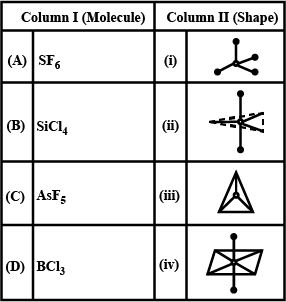

Detailed Solution: Question 9
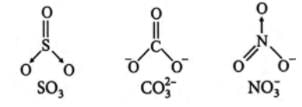
What is common between the following molecules
SO3, CO2-3, No-3
Detailed Solution: Question 10
Which molecule is depicted by the given ball and stick models?
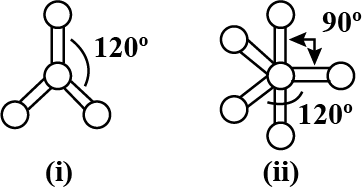

Detailed Solution: Question 11
Which of the following does not show octahedral geometry?
Detailed Solution: Question 12
Given below is the table showing shapes of some molecules having lone pairs of electrons. Fill up the blanks left in it.
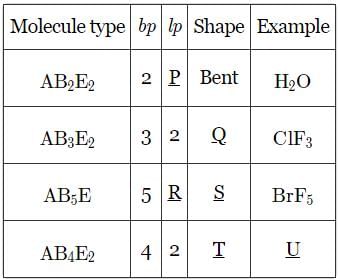
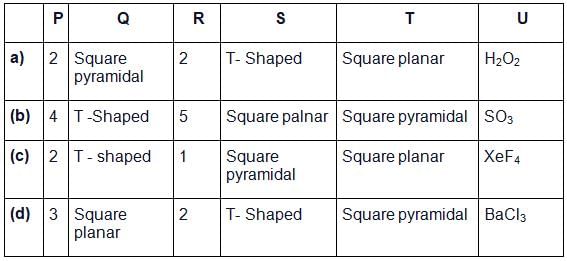


The BCI3 is a planar molecule whereas NCI3 is pyramidal, because
Detailed Solution: Question 14
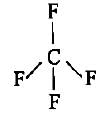
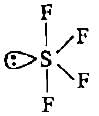
CF4, SF4 and XeF4 contain the following electronicstructure on their central atoms. Which one iscorrect option?
Detailed Solution: Question 15
98 videos|243 docs|71 tests |