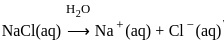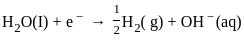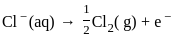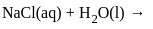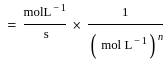CUET UG Chemistry: Mock Test - 2 Free Online Test 2026
Full Mock Test & Solutions: Chemistry: CUET Mock Test - 2 (49 Questions)
You can boost your CUET 2026 exam preparation with this Chemistry: CUET Mock Test - 2 (available with detailed solutions).. This mock test has been designed with the analysis of important topics, recent trends of the exam, and previous year questions of the last 3-years. All the questions have been designed to mirror the official pattern of CUET 2026 exam, helping you build speed, accuracy as per the actual exam.
Mock Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 60 minutes
- - Total Questions: 49
- - Analysis: Detailed Solutions & Performance Insights
Sign up on EduRev for free and get access to these mock tests, get your All India Rank, and identify your weak areas to improve your marks & rank in the actual exam.
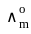 for NaCl, HCl and NaOAc are 126.4, 425.9 and 91.0 S cm2 mol-1 respectively. Calculate ∧° for HOAc
for NaCl, HCl and NaOAc are 126.4, 425.9 and 91.0 S cm2 mol-1 respectively. Calculate ∧° for HOAc
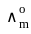 for NaCl, HCl and NaOAc are 126.4, 425.9 and 91.0 S cm2 mol-1 respectively. Calculate ∧° for HOAc
for NaCl, HCl and NaOAc are 126.4, 425.9 and 91.0 S cm2 mol-1 respectively. Calculate ∧° for HOAcDetailed Solution: Question 1
Detailed Solution: Question 2
The products formed at cathode and anode by electrolysis of aqueous NaCl solution respectively are
Detailed Solution: Question 3
Rate constant 'k' for a certain reaction is k = 2.3 × 10-5 L mol-1s-1. Order of the reaction is:
Detailed Solution: Question 4
Detailed Solution: Question 5
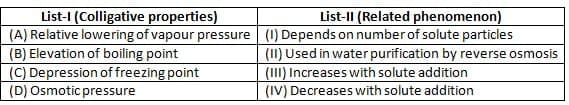
Why do colligative properties not significantly change with the addition of a small amount of a weak electrolyte to a solution?
Detailed Solution: Question 6
What accurately describes how boiling point elevation and freezing point depression are related to solute concentration?
Detailed Solution: Question 7
In the context of freezing point depression, why do roads get treated with salt in winter?
Detailed Solution: Question 8
What is the effect of a non-volatile solute on the vapor pressure of a solution?
Detailed Solution: Question 9
Osmotic pressure is a colligative property that demonstrates:
Detailed Solution: Question 10
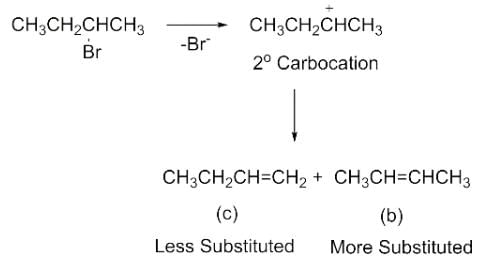
Elimination of bromine from 2-bromobutane results in the formation of
Detailed Solution: Question 11
Detailed Solution: Question 12
0.002 molal aqueous solution of an ionic compound [Co(NH3)5(N02)]CI freezes at - 0.00732° C. [Kf (H20) = 1.86° mol-1kg]. How many moles of ions does 1.0 mole of the salt produce on being dissolved in water?
Detailed Solution: Question 13
Detailed Solution: Question 14
Nitro compounds are reduced by iron scrap and hydrochloric acid to yield one of the following compounds.
Detailed Solution: Question 15
Which one of the following is used to increase blood pressure
Detailed Solution: Question 16
Which of the following monosaccharides is a pentose-
Detailed Solution: Question 17
Ring structure of glucose is due to formation of hemiacetal and ring formation between-
Detailed Solution: Question 18
Detailed Solution: Question 19
The inner transition elements are the elements in which the added electrons go to:
Detailed Solution: Question 20
If zeise’s salt has the formula [Pt(C2H4)CI3]-. In this, platinum primary and secondary valency are
Detailed Solution: Question 21
The two complexes PtCI4 . 2NH3 and PtCI4 . 2KCI do not give precipitate of AgCI when treate with AgNO3. The structures of these complexes are
Detailed Solution: Question 22
Which of the following isomeric alcohols is the most soluble in water?
Detailed Solution: Question 23
Which of the following is necessary for the bromination of phenol?
Detailed Solution: Question 24
Phenols are least soluble in which of the following?
Detailed Solution: Question 25
Four compounds A, B, C and D having similar molecular masses were tested for their boiling points. It was found that compound C has the highest boiling point of all four. What is the compound C most likely to be?
Detailed Solution: Question 26
Detailed Solution: Question 27
Detailed Solution: Question 28
Arrange the following 0.1 m aqueous solutions in increasing order ofelevation of boiling point (ΔTᵇ):
(A) KCl
(B) Urea
(C) BaCl2
(D) K3[Fe(CN)6]
Choose the correct order:
(A) KCl
(B) Urea
(C) BaCl2
(D) K3[Fe(CN)6]
Choose the correct order:
Detailed Solution: Question 29
Arrange the following metals in decreasing order of their standard reduction potential (E° values):
(A) Zn2+/Zn
(B) Fe2+/Fe
(C) Cu2+/Cu
(D) Ag+/Ag
Choose the correct order:
(A) Zn2+/Zn
(B) Fe2+/Fe
(C) Cu2+/Cu
(D) Ag+/Ag
Choose the correct order:
Detailed Solution: Question 30
41 docs|160 tests |


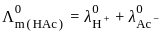
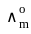 for NaCl, HCl and NaOAc are 126.4, 425.9 and 91.0 S cm2 mol-1
for NaCl, HCl and NaOAc are 126.4, 425.9 and 91.0 S cm2 mol-1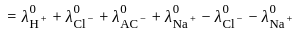
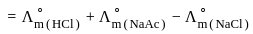
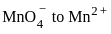 ?
?