Conformational Analysis - 2 - Free MCQ Practice Test with solutions, JEE
MCQ Practice Test & Solutions: Test: Conformational Analysis - 2 (14 Questions)
You can prepare effectively for JEE Chemistry for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: Conformational Analysis - 2". These 14 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 20 minutes
- - Number of Questions: 14
Sign up on EduRev for free to attempt this test and track your preparation progress.
Direction (Q. Nos. 1- 8) This section contains 8 multiple choice questions. Each question has four
choices (a), (b), (c) and (d), out of which ONE or MORE THANT ONE is correct.
Q.
Among the butane conformers, when viewed about C2-C3 bond, which occur at energy minima on a graph of potential energy versus dihedral angle?
choices (a), (b), (c) and (d), out of which ONE or MORE THANT ONE is correct.
The correct statement regarding cyclopropane is/are
Detailed Solution: Question 2
Select the correct statement(s) concerning conformers of ethane and hexachloroethane?
The correct statement concerning various conformers of 2-fluoroethanol is/are
Consider the various conformers possible for meso form and enantiomeric 1,2-dibromo-1,2-dichloroethane and select the correct statement(s).
Select the correct statement(s) regarding conformers of 1,2-dimethyl cyclohexane.
The correct statement regarding conformation in butane is/are
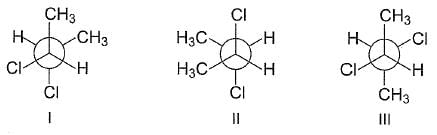
the correct statement(s) concerning the following Newmann's projection is/are

Direction (Q. Nos. 9-14) This section contains 2 paragraph, wach describing theory, experiments, data etc. three Questions related to paragraph have been given.Each question have only one correct answer among the four given ptions (a),(b),(c),(d)
Passage I
The two major contributors of conformers of 1,2-dichloroethane are anti and gauche. At 32°C in gas phase, the measured dipole moment of 1,2-dichloroethane is 1.12 D. The dipole moment of a mixture of X and Y is given by the relationship
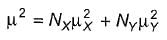
Here, N = mole fraction of each kind of molecule. From bond moment measurement, it has been estimated that gauche conformer of 1,2-dichloroethane should have a dipole moment of about 3.2 D.
Q.
What per cent of conformers of 1,2-dichloroethane is anti, at 32°C?
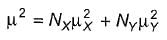
Detailed Solution: Question 9
Passage I
The two major contributors of conformers of 1,2-dichloroethane are anti and gauche. At 32°C in gas phase, the measured dipole moment of 1,2-dichloroethane is 1.12 D. The dipole moment of a mixture of X and Y is given by the relationship
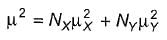
Here, N = mole fraction of each kind of molecule. From bond moment measurement, it has been estimated that gauche conformer of 1,2-dichloroethane should have a dipole moment of about 3.2 D.
Q.
What is true about percentage of gauche conformer?
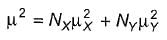
What is true about percentage of gauche conformer?
Passage I
The two major contributors of conformers of 1,2-dichloroethane are anti and gauche. At 32°C in gas phase, the measured dipole moment of 1,2-dichloroethane is 1.12 D. The dipole moment of a mixture of X and Y is given by the relationship
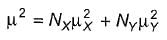
Here, N = mole fraction of each kind of molecule. From bond moment measurement, it has been estimated that gauche conformer of 1,2-dichloroethane should have a dipole moment of about 3.2 D.
Q.
What happens if temperature if increased to 52°C?
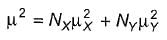
Passage II
Among conformers, the barrier to rotation originate mainly from torsional strain when atoms or groups eclipse one another. Followings are the costs of energy required to cross the rotational barrier for the indicated pair of groups eclipsing
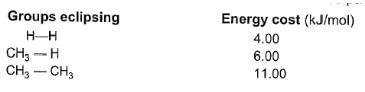
Q.
What is the energy barrier to rotation in neopentane ?
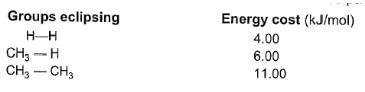
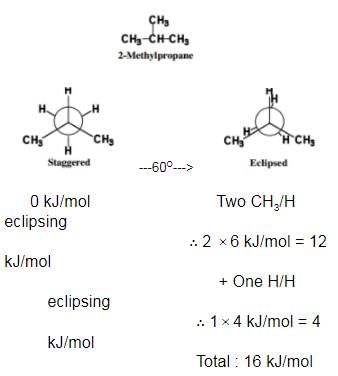
Passage II
Among conformers, the barrier to rotation originate mainly from torsional strain when atoms or groups eclipse one another. Followings are the costs of energy required to cross the rotational barrier for the indicated pair of groups eclipsing
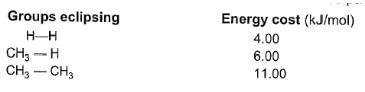
Q.
What is the energy barrier to rotation in 2-methyl propane ?
Detailed Solution: Question 13
Passage II
Among conformers, the barrier to rotation originate mainly from torsional strain when atoms or groups eclipse one another. Followings are the costs of energy required to cross the rotational barrier for the indicated pair of groups eclipsing

Q.
What is the energy difference between two eclipsed conformers of butane when viewed between C2-C3 bond ?

335 videos|699 docs|300 tests |