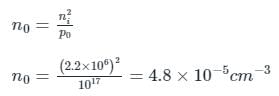GATE ECE (Electronics) Test: P-Type Semiconductor Free Online Test 2026
MCQ Practice Test & Solutions: Test: P-Type Semiconductor (5 Questions)
You can prepare effectively for Electronics and Communication Engineering (ECE) GATE ECE (Electronics) Mock Test Series 2027 with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: P-Type Semiconductor". These 5 questions have been designed by the experts with the latest curriculum of Electronics and Communication Engineering (ECE) 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 15 minutes
- - Number of Questions: 5
Sign up on EduRev for free to attempt this test and track your preparation progress.
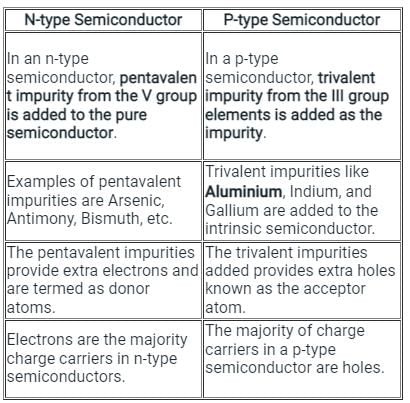
Which of the following can be used in the fabrication of p-type semiconductor?
Detailed Solution: Question 1
Detailed Solution: Question 2
Detailed Solution: Question 3
A sample of GaAs doped with NA = 1017 cm-3. For GaAs intrinsic concentration is n = 2.2 × 106 cm-3, mobility of electron is μn = 5300 cm2/V-sec, and mobility of hole is μp = 230 cm2/V sec.
If the sample is illuminated such that the excess electron concentration is 1016 cm-3. What will the conductivity [in (Ω-cm)-1] of this sample, when the light is ON?
Detailed Solution: Question 4
Detailed Solution: Question 5
26 docs|263 tests |